Revolutionizing Carfilzomib Production: A High-Yield N-to-C Synthetic Strategy for Commercial Scale
Revolutionizing Carfilzomib Production: A High-Yield N-to-C Synthetic Strategy for Commercial Scale
The pharmaceutical industry continuously seeks robust synthetic pathways for complex oncology therapeutics, and Carfilzomib stands as a prime example of a challenging target due to its sensitive epoxyketone warhead and peptide backbone. Patent CN113024637B introduces a groundbreaking methodology that fundamentally shifts the synthetic paradigm from the traditional C-terminal to N-terminal extension to a more efficient N-to-C direction. This innovation leverages a specialized water-soluble alkyne amide as a condensing agent, addressing the critical pain points of racemization and purification that have long plagued peptide drug manufacturing. By starting from inexpensive chloroacetic acid and sequentially coupling protected amino acids, this route achieves a remarkable total yield while maintaining stringent stereochemical integrity. For R&D leaders and supply chain strategists, this represents a significant leap forward in process chemistry, offering a viable path toward cost-effective and scalable production of this vital proteasome inhibitor.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Carfilzomib has predominantly relied on C-to-N peptide elongation strategies, which inherently suffer from severe technical drawbacks when applied to this specific molecular architecture. In traditional approaches, the alpha-position of the polypeptide carboxylic acid is adjacent to an amide bond rather than a standard alkoxycarbonyl protecting group, making it highly susceptible to racemization during activation with conventional coupling reagents. This stereochemical erosion not only complicates the isolation of the desired enantiomer but also drastically reduces overall yield, often resulting in totals below 45 percent as noted in prior art. Furthermore, the instability of the C-terminal epoxy functional group necessitates extremely mild conditions that many standard coupling agents cannot provide, leading to hydrolysis side reactions and difficult purification scenarios that frequently require expensive preparative HPLC.
The Novel Approach
In stark contrast, the methodology disclosed in CN113024637B employs a strategic N-to-C chain extension that circumvents these stereochemical pitfalls entirely. By utilizing chloroacetic acid as the anchor and building the peptide chain towards the C-terminus, the process avoids the formation of racemization-prone intermediates associated with traditional C-terminal activation. The core innovation lies in the use of a water-soluble alkyne amide condensing agent, which facilitates activation under exceptionally mild conditions compatible with the sensitive epoxyketone moiety. This approach not only ensures complete avoidance of racemization but also simplifies the downstream processing significantly, as the water-soluble nature of the reagent's by-products allows for clean separation via simple aqueous extraction and recrystallization, bypassing the need for column chromatography entirely.
Mechanistic Insights into Water-Soluble Alkyne Amide Mediated Coupling
The mechanistic elegance of this synthesis rests on the unique reactivity profile of the water-soluble alkyne amide, specifically N-(2,2-dimethyl-1,3-dioxolan-4-ylmethyl)-N-ethynyl methanesulfonamide. Unlike carbodiimides or phosphonium salts that can induce harsh acidic or basic environments, this reagent activates carboxylic acids through a gentle mechanism that preserves the chirality of the alpha-carbon. The activation step generates a reactive intermediate that readily undergoes nucleophilic attack by the amine component of the incoming amino acid ester without forming the oxazolone species responsible for epimerization. This is particularly crucial when coupling sterically hindered residues or when the carboxyl component already contains an amide bond, a scenario where traditional reagents typically fail to maintain optical purity. The result is a coupling efficiency that supports high-fidelity peptide bond formation essential for biological activity.
Furthermore, the "three-step one-pot" operational design integrates activation, condensation, and deprotection into a seamless sequence within a single reactor vessel, minimizing material handling and exposure to potential degradants. After the coupling event, the tert-butyl protecting groups are removed using acidic conditions such as trifluoroacetic acid, and the resulting free amine is immediately ready for the next cycle of activation. The water solubility of the sulfonamide by-product generated during this cycle is the key to the simplified work-up; upon addition of water, these impurities partition exclusively into the aqueous phase, leaving the product in the organic layer with high purity. This mechanism effectively decouples the complexity of peptide synthesis from the burden of complex purification, enabling a streamlined workflow that is both chemically robust and operationally simple.
How to Synthesize Carfilzomib Efficiently
The practical execution of this synthesis involves a sequential assembly of amino acid fragments starting from chloroacetic acid, proceeding through intermediates A, B, and C, before finalizing with the epoxyketone warhead and morpholine cap. Each stage utilizes the water-soluble alkyne amide for activation followed by coupling with the respective tert-butyl protected amino acid, such as L-homophenylalanine, L-leucine, and L-phenylalanine derivatives. The process is characterized by mild temperatures ranging from 0 to 60 degrees Celsius and standard organic solvents like dichloromethane, ensuring safety and ease of control. Detailed standardized synthetic steps for implementing this high-yield route are provided in the guide below.
- Activate chloroacetic acid with water-soluble alkyne amide, condense with L-homophenylalanine tert-butyl ester, and remove protection to obtain Compound A.
- Activate Compound A, condense with L-leucine tert-butyl ester, and deprotect to form Compound B.
- Activate Compound B, condense with L-phenylalanine tert-butyl ester, and deprotect to yield Compound C.
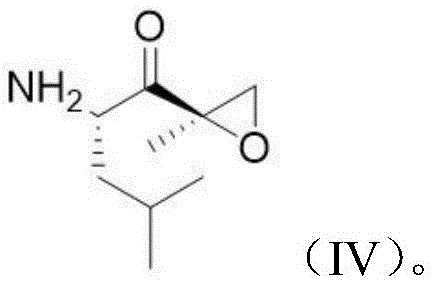
- Activate Compound C and condense with the epoxyketone warhead ((2S)-2-amino-4-methyl-1-[(2R)-2-methyloxiranyl]-1-pentanone) to form Intermediate II.
- React Intermediate II with morpholine in the presence of a catalyst (e.g., KI) to finalize the Carfilzomib structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers transformative benefits that directly impact the bottom line and operational resilience. The elimination of column chromatography is perhaps the most significant cost driver, as preparative purification often accounts for a substantial portion of manufacturing expenses in peptide chemistry. By replacing this with simple extraction and recrystallization, the process drastically reduces solvent consumption, silica gel waste, and labor hours, leading to substantial cost savings in API manufacturing. Additionally, the high atom economy and the use of readily available starting materials like chloroacetic acid and common amino acid esters ensure a stable and predictable raw material supply chain, mitigating risks associated with specialty reagent shortages.
- Cost Reduction in Manufacturing: The economic advantage of this method is derived primarily from the simplification of the purification workflow. Traditional peptide synthesis often requires multiple chromatographic separations to remove racemic impurities and coupling by-products, which is both time-consuming and expensive. This new protocol generates water-soluble by-products that are easily washed away, allowing for high-purity isolation through crystallization alone. This reduction in unit operations translates to lower utility costs, reduced waste disposal fees, and a smaller physical footprint for production, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing without compromising quality.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the reliance on commodity chemicals rather than exotic or proprietary coupling agents that may have limited suppliers. Chloroacetic acid, morpholine, and standard protected amino acids are produced globally at massive scales, ensuring that raw material availability remains robust even during market fluctuations. Furthermore, the robustness of the reaction conditions means that the process is less sensitive to minor variations in temperature or reagent quality, reducing the likelihood of batch failures. This reliability is critical for maintaining consistent inventory levels and meeting the rigorous delivery schedules demanded by downstream drug product manufacturers.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the "three-step one-pot" strategy minimizes the number of isolation steps, thereby reducing the total volume of organic solvents required per kilogram of product. This aligns with green chemistry principles by lowering the E-factor (mass of waste per mass of product) and simplifying solvent recovery processes. The mild reaction conditions also reduce energy consumption compared to processes requiring cryogenic temperatures or extended heating. These factors collectively enhance the scalability of the process from pilot plant to commercial tonnage, ensuring that the technology can meet growing market demand for Carfilzomib while adhering to increasingly strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this novel synthesis method, based on the detailed specifications and experimental data provided in the patent literature. Understanding these aspects is crucial for evaluating the feasibility of technology transfer and integration into existing manufacturing pipelines. The answers reflect the specific advantages of the water-soluble alkyne amide approach over legacy methods.
Q: How does this new method prevent racemization during peptide coupling?
A: Traditional C-to-N synthesis often causes racemization at the alpha-position of amide bonds due to oxazolone formation. This novel N-to-C approach utilizes a water-soluble alkyne amide condensing agent that activates chiral carboxylic acids under mild conditions, completely avoiding the formation of racemization-prone intermediates.
Q: What are the purification advantages of using water-soluble alkyne amide?
A: The by-products generated from the water-soluble alkyne amide condensing agent are highly soluble in water. This unique property allows for simple work-up procedures involving only extraction and recrystallization, eliminating the need for costly and time-consuming column chromatography or preparative HPLC.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It features mild reaction conditions, a 'three-step one-pot' strategy that reduces unit operations, and achieves a high total yield of approximately 70%. The elimination of complex purification steps significantly lowers production costs and environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carfilzomib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to remain competitive in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the pathway described in CN113024637B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this efficient route to life. We are committed to delivering high-purity Carfilzomib intermediates and APIs that meet stringent purity specifications, leveraging our rigorous QC labs to ensure every batch conforms to the highest international standards. Our facility is equipped to handle the specific solvent systems and crystallization protocols necessary to maximize the yield benefits of this water-soluble condensing agent technology.
We invite you to collaborate with us to optimize your supply chain and reduce your cost of goods sold through this superior manufacturing process. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can improve your margins. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to secure a reliable, high-quality supply of Carfilzomib for your oncology portfolio.
