Scalable Manufacturing of Tetrabenazine via Regioselective Mannich Reaction
Scalable Manufacturing of Tetrabenazine via Regioselective Mannich Reaction
The pharmaceutical landscape for treating hyperkinetic movement disorders, specifically Huntington's chorea, relies heavily on the consistent supply of high-quality Tetrabenazine. Patent CN102936246A introduces a transformative synthetic methodology that addresses critical bottlenecks in the production of this vital VMAT2 inhibitor. By shifting away from hazardous reagents like lithium aluminium hydride and toxic methyl iodide, this novel approach leverages inexpensive, commercially available starting materials such as dimethylamine and formalin. The core innovation lies in the strategic use of imine salts as aminomethylation reagents, which fundamentally alters the reaction pathway to favor the desired regioisomer. This technical breakthrough not only enhances the chemical purity of the final API intermediate but also streamlines the downstream processing requirements. For global supply chain stakeholders, this represents a significant opportunity to secure a more robust and cost-effective source of Tetrabenazine, mitigating the risks associated with complex purification protocols and hazardous waste disposal inherent in legacy manufacturing processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Tetrabenazine has been plagued by severe inefficiencies and safety hazards that compromise both economic viability and operational safety. Early methodologies, such as those reported by Osbond, relied on the use of lithium aluminium hydride, a highly pyrophoric and expensive reducing agent that necessitates stringent anhydrous conditions and specialized handling equipment. Furthermore, alternative routes described in prior art, including patent WO 080582612, utilize a Mannich reaction involving dimethylamine hydrochloride and paraformaldehyde which notoriously generates a substantial quantity of the regioisomer 1-(dimethylamino)-6-methyl-3-heptanone. As illustrated in the reaction scheme below, this isomer possesses a boiling point dangerously close to the desired product, rendering fractional distillation ineffective and forcing manufacturers to resort to preparative column chromatography. This reliance on chromatographic purification is a major bottleneck for commercial scale-up, resulting in extremely low overall yields, often cited around 16%, and generating massive volumes of silica gel waste. Additionally, the use of methyl iodide for quaternization in these older pathways introduces significant toxicity concerns and regulatory burdens regarding residual solvent limits in the final pharmaceutical product.
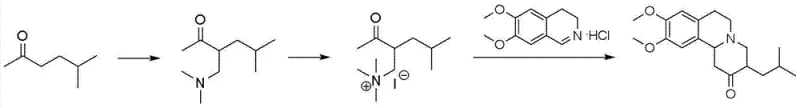
The Novel Approach
In stark contrast to these cumbersome legacy processes, the method disclosed in CN102936246A offers a streamlined, three-step sequence that prioritizes atom economy and operational simplicity. The process initiates with the condensation of dimethylamine and formaldehyde to form tetramethylmethylenediamine, a stable and easily handled intermediate. This diamine is subsequently activated with acetyl chloride to generate a reactive iminium species in situ, which then undergoes a highly regioselective Mannich reaction with 5-methyl-2-hexanone. As depicted in the following schematic, this specific activation mode effectively suppresses the formation of the problematic branched isomer, directing the aminomethyl group exclusively to the desired position on the ketone chain. The final cyclization step is performed in an aqueous medium, reacting the purified ketone intermediate with 6,7-dimethoxy-3,4-dihydroisoquinoline hydrochloride. This aqueous protocol not only eliminates the need for flammable organic solvents like acetone in the final stage but also facilitates product isolation through simple crystallization or filtration, thereby drastically reducing the environmental footprint and processing time required to obtain pharmaceutical-grade Tetrabenazine.
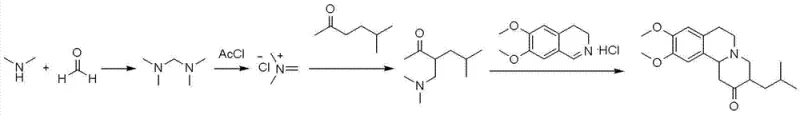
Mechanistic Insights into Imine Salt-Mediated Aminomethylation
The cornerstone of this improved synthesis is the mechanistic deviation from standard aminal-based Mannich conditions to an iminium ion-mediated pathway. In traditional aqueous Mannich reactions, the equilibrium between the amine, aldehyde, and ketone often leads to thermodynamic mixtures of products, explaining the high isomer content observed in prior art. However, by pre-forming the tetramethylmethylenediamine and subsequently treating it with acetyl chloride, the process generates a highly electrophilic N,N-dimethyliminium chloride species. This activated electrophile reacts with the enol form of 5-methyl-2-hexanone under kinetic control. The steric environment created by the bulky iminium intermediate, combined with the specific electronic activation provided by the acetyl chloride moiety, favors attack at the less hindered methyl group adjacent to the carbonyl, rather than the internal methylene position. This kinetic preference is crucial for achieving the high regioselectivity reported in the patent, ensuring that the carbon skeleton is constructed correctly from the outset. Consequently, the reaction mixture contains predominantly the linear 3-[(dimethylamino)methyl]-5-methyl-2-hexanone, minimizing the burden on downstream purification units and preserving yield throughout the synthesis.
Furthermore, the choice of solvent systems plays a pivotal role in maintaining the integrity of the reactive intermediates and facilitating efficient mass transfer. The patent specifies the use of polar aprotic solvents such as DMF, acetonitrile, or methyl tert-butyl ether during the aminomethylation step, which solubilize the ionic iminium species while allowing for effective temperature control between 0°C and 75°C. Following the reaction, the workup procedure involves a carefully controlled pH adjustment to separate the basic amine product from acidic byproducts and unreacted starting materials. The final isolation via vacuum distillation at reduced pressure (20 mmHg) allows for the removal of the product at temperatures between 95°C and 105°C, well below the decomposition threshold of the sensitive ketone functionality. This gentle purification method ensures that the intermediate retains its optical and chemical purity, which is essential for the subsequent stereoselective cyclization with the isoquinoline derivative to form the final tetrabenazine structure without introducing racemization or degradation impurities.
How to Synthesize Tetrabenazine Efficiently
The implementation of this synthetic route requires precise control over reaction parameters to maximize the benefits of the imine salt mechanism. Operators must ensure strict temperature regulation during the addition of acetyl chloride to prevent exothermic runaway, followed by a gradual warming phase to drive the aminomethylation to completion. The detailed standardized operating procedures, including specific molar ratios, stirring rates, and distillation cut points, are critical for reproducing the high yields (>70% for the intermediate and >85% for the final API) described in the patent embodiments. For a comprehensive guide on executing this process safely and effectively, please refer to the technical protocol outlined below.
- Synthesize tetramethylmethylenediamine by reacting dimethylamine aqueous solution with formalin, followed by alkaline treatment and distillation.
- Perform aminomethylation by reacting the diamine with acetyl chloride and 5-methyl-2-hexanone in an organic solvent to form the key ketone intermediate.
- React the intermediate with 6,7-dimethoxy-3,4-dihydroisoquinoline hydrochloride in water to cyclize and precipitate high-purity Tetrabenazine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic pathway offers profound strategic advantages that extend far beyond simple chemical yield improvements. By fundamentally redesigning the manufacturing process to utilize commodity chemicals and eliminate hazardous reagents, the total cost of ownership for Tetrabenazine production is significantly optimized. The removal of complex purification steps such as column chromatography translates directly into reduced cycle times and lower capital expenditure on specialized separation equipment. Moreover, the shift towards aqueous chemistry in the final step aligns perfectly with modern sustainability mandates, reducing the volume of volatile organic compound (VOC) emissions and hazardous waste streams that require costly disposal. This enhanced process efficiency ensures a more reliable supply of high-purity Tetrabenazine, mitigating the risk of production delays caused by reagent shortages or environmental compliance audits.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents such as lithium aluminium hydride and methyl iodide results in a drastic reduction in raw material expenditures. Traditional methods often incur hidden costs related to the safe handling, storage, and neutralization of these dangerous chemicals, which are completely avoided in this new protocol. Furthermore, the ability to purify the key intermediate via simple vacuum distillation rather than resource-intensive column chromatography significantly lowers solvent consumption and labor costs associated with batch processing. This streamlined approach allows for higher throughput in existing reactor vessels, effectively increasing capacity without the need for new infrastructure investment, thereby driving down the unit cost of the active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the primary starting materials—dimethylamine, formalin, and 5-methyl-2-hexanone—are bulk commodity chemicals available from multiple global suppliers. Unlike specialized reagents that may be subject to geopolitical supply constraints or single-source monopolies, these feedstocks ensure continuous production capability even during market fluctuations. The robustness of the reaction conditions, which tolerate standard industrial grade solvents and do not require ultra-dry or cryogenic environments, further reduces the risk of batch failures due to minor variations in raw material quality. This resilience ensures that procurement teams can maintain consistent inventory levels and meet delivery schedules for downstream formulation partners without interruption.
- Scalability and Environmental Compliance: The transition to water as the reaction medium for the final cyclization step represents a major advancement in green chemistry compliance, significantly reducing the facility's environmental footprint. By avoiding large volumes of organic solvents like acetone in the final stage, the load on solvent recovery distillation columns is minimized, and the generation of hazardous waste sludge is virtually eliminated. This simplification of the waste profile facilitates easier regulatory approval and reduces the long-term liability associated with environmental remediation. Additionally, the high regioselectivity of the process minimizes the formation of difficult-to-remove isomers, ensuring that the final product consistently meets stringent pharmacopeial specifications for impurities, which is critical for successful regulatory filings and market authorization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced Tetrabenazine synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on how this method outperforms historical precedents. Understanding these distinctions is vital for technical teams evaluating process transfer feasibility and for commercial teams assessing the long-term value proposition of this manufacturing technology.
Q: How does this method improve regioselectivity compared to traditional Mannich reactions?
A: By utilizing an imine salt generated in situ from tetramethylmethylenediamine and acetyl chloride, the reaction avoids the formation of the unwanted 1-(dimethylamino)-6-methyl-3-heptanone isomer, significantly simplifying purification.
Q: What are the environmental benefits of using water in the final cyclization step?
A: Using water as the solvent for the final coupling eliminates the need for large volumes of volatile organic solvents like acetone, reduces solid waste from inorganic salts, and simplifies product isolation through precipitation.
Q: Why is column chromatography avoided in this synthesis route?
A: The high regioselectivity of the aminomethylation step yields a crude product with minimal isomeric impurities, allowing for purification via simple vacuum distillation rather than costly and slow column chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrabenazine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of this patent can be realized in a GMP-compliant environment. We are equipped with rigorous QC labs and state-of-the-art processing units capable of handling the specific distillation and crystallization requirements of this route, guaranteeing stringent purity specifications for every batch of Tetrabenazine we produce. Our commitment to quality assurance means that we not only deliver the final API but also provide full traceability and documentation support necessary for your regulatory submissions.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective and environmentally sustainable synthesis technology. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can improve your margins. We encourage you to contact us today to discuss route feasibility assessments and to obtain specific COA data for our pilot batches, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
