Scalable Synthesis of 6,7-Dimethoxy-3,4-Dihydroisoquinoline for Commercial API Production
Scalable Synthesis of 6,7-Dimethoxy-3,4-Dihydroisoquinoline for Commercial API Production
The pharmaceutical industry continuously seeks robust synthetic routes for complex neurological agents, and patent CN110698397A presents a transformative approach to producing 6,7-dimethoxy-3,4-dihydroisoquinoline, a critical intermediate for Tetrabenazine. This specific compound serves as a foundational building block in the manufacture of drugs targeting Huntington's disease, where supply consistency and chemical purity are paramount for regulatory approval. The disclosed methodology leverages a multi-step sequence starting from 3,4-dihydroxyphenylethanol, effectively bypassing the economic and technical bottlenecks associated with legacy synthesis pathways. By optimizing reaction conditions such as temperature control, molar ratios, and solvent systems, this process delivers a product with exceptional chemical integrity suitable for sensitive downstream applications. For global procurement teams, understanding the technical nuances of this patent is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent quality standards. The innovation lies not just in the chemical transformation but in the holistic design of a process that balances high yield with operational simplicity, ensuring that the supply chain remains resilient against market fluctuations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 6,7-dimethoxy-3,4-dihydroisoquinoline has been plagued by significant inefficiencies that hinder large-scale commercial viability. Traditional methods often rely on 1,2,3,4-tetrahydropapaverine as a starting material, a substance that is not only difficult to source industrially but also commands a prohibitively high price point due to its complex extraction or synthesis requirements. Furthermore, literature indicates that reactions involving concentrated sulfuric acid on such substrates typically suffer from low yields, often ranging between 30% and 42%, which generates substantial chemical waste and drives up the cost per kilogram of the final active ingredient. Alternative routes utilizing urotropine and 3,4-dimethoxyphenethylamine introduce severe purification challenges, where byproducts like 6,7-dimethoxy-2-methyl-3,4-dihydro-1H-isoquinoline are structurally similar and difficult to separate, resulting in product purity levels as low as 35%. These technical deficiencies create unacceptable risks for supply chain heads who require consistent batch-to-batch quality and for procurement managers tasked with maintaining budget discipline in API manufacturing. The reliance on harsh conditions and scarce precursors fundamentally limits the ability to scale production to meet the growing global demand for neurological therapeutics.
The Novel Approach
In stark contrast, the novel methodology detailed in the patent data utilizes 3,4-dihydroxyphenylethanol, a commercially abundant and cost-effective raw material that eliminates the dependency on scarce natural alkaloids. This strategic shift in starting material allows for a streamlined synthetic sequence that avoids the formation of intractable impurities, thereby simplifying the downstream purification process significantly. The process employs a series of controlled reactions including methylation, selective ether cleavage, and cyclization, each optimized to maximize atom economy and minimize waste generation. By implementing precise temperature controls and specific molar ratios, the reaction system maintains high stability, preventing the degradation of sensitive intermediates that often plagues conventional batch processes. This approach directly addresses the need for cost reduction in pharmaceutical intermediate manufacturing by reducing the number of purification steps and increasing the overall throughput of the production line. For R&D directors, this represents a viable pathway to achieve high-purity OLED material or pharmaceutical grade intermediates without the technical headaches associated with legacy chemistry, ensuring a smoother technology transfer from lab to plant.
Mechanistic Insights into the Multi-Step Synthetic Route
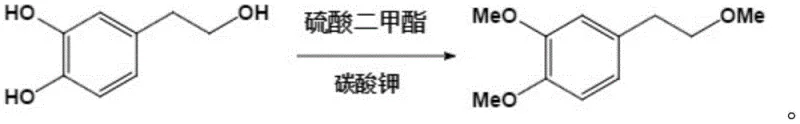
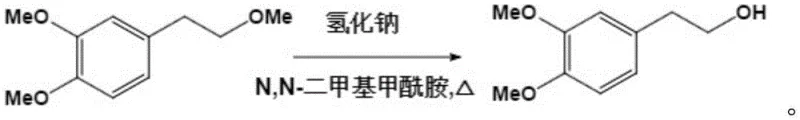
The core of this synthetic innovation lies in a meticulously designed four-step reaction sequence that transforms a simple phenolic alcohol into a complex heterocyclic system with high fidelity. The initial phase involves the exhaustive methylation of 3,4-dihydroxyphenylethanol using dimethyl sulfate in the presence of anhydrous potassium carbonate and acetone, which protects the phenolic hydroxyl groups while simultaneously methylating the aliphatic chain. This is followed by a highly selective cleavage step where sodium hydride and ethanethiol in DMF are utilized to specifically target the aliphatic ether bond, regenerating the terminal hydroxyl group without affecting the aromatic methoxy groups. This selectivity is crucial for setting up the subsequent amination reaction, where the regenerated alcohol is converted into an amine using thionyl chloride and methylamine solution under strictly controlled low-temperature conditions to prevent side reactions. The precision required in these steps underscores the importance of working with a partner who possesses deep expertise in fine chemical synthesis and process safety.
The final and most critical transformation is the cyclization step, which constructs the isoquinoline core through a Pictet-Spengler type condensation using paraformaldehyde and Eaton's reagent. This step requires careful management of exothermic activity and pH levels, utilizing isopropyl acetate for extraction and sodium hydroxide for neutralization to isolate the final yellow oil product. The mechanistic pathway ensures that the nitrogen atom is correctly positioned within the ring system, which is vital for the biological activity of the final Tetrabenazine drug substance. Impurity control is maintained throughout this sequence by monitoring reaction progress and adjusting stoichiometry, ensuring that the final product meets the rigorous specifications required for human therapeutic use. The ability to consistently achieve purity levels above 90% demonstrates the robustness of this chemical design, offering a significant advantage over methods that struggle with byproduct separation. This level of mechanistic control is what distinguishes a laboratory curiosity from a commercially viable industrial process capable of supporting global supply chains.

How to Synthesize 6,7-Dimethoxy-3,4-Dihydroisoquinoline Efficiently
Implementing this synthesis route requires strict adherence to the operational parameters outlined in the patent to ensure safety and reproducibility on a commercial scale. The process begins with the preparation of the reaction vessel under a nitrogen atmosphere to prevent oxidation of sensitive intermediates, followed by the sequential addition of reagents at controlled rates to manage heat generation. Detailed standard operating procedures dictate specific distillation volumes, cooling temperatures, and pH adjustments during the workup phase to maximize recovery and purity. While the general chemistry is straightforward, the devil is in the details regarding mixing efficiency, heat transfer, and filtration techniques which are critical for maintaining product quality. For those seeking to implement this technology, it is essential to consult the full technical documentation to understand the nuances of scaling these unit operations from grams to tons. The following guide summarizes the critical stages involved in this transformation.
- Methylation of 3,4-dihydroxyphenylethanol using dimethyl sulfate and potassium carbonate in acetone to form the dimethoxy ether intermediate.
- Selective cleavage of the aliphatic ether bond using sodium hydride and ethanethiol in DMF to regenerate the terminal hydroxyl group.
- Conversion of the alcohol to an amine followed by Pictet-Spengler cyclization with paraformaldehyde and Eaton's reagent to close the isoquinoline ring.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis route offers compelling advantages that directly impact the bottom line and operational reliability of pharmaceutical manufacturing. By shifting to a starting material that is inexpensive and readily available in the global chemical market, manufacturers can decouple their production costs from the volatility associated with scarce natural extracts. The elimination of complex purification steps required to remove structurally similar byproducts translates into shorter cycle times and reduced consumption of solvents and energy, contributing to substantial cost savings in the overall manufacturing budget. Furthermore, the high yield and purity profile of this method reduce the risk of batch failures, ensuring a more predictable supply of critical intermediates for downstream API production. This reliability is invaluable for supply chain heads who must guarantee continuity of supply to meet patient demand without incurring the costs of emergency sourcing or expedited shipping. The process is inherently designed for scalability, allowing for seamless transition from pilot plant to full commercial production without the need for major process re-engineering.
- Cost Reduction in Manufacturing: The utilization of 3,4-dihydroxyphenylethanol as a feedstock significantly lowers the raw material cost base compared to traditional alkaloid-derived starting materials. By avoiding the use of expensive catalysts and reducing the number of purification stages, the overall processing cost is drastically simplified, leading to a more competitive pricing structure for the final intermediate. The high atom economy of the reaction sequence minimizes waste disposal costs, further enhancing the economic efficiency of the production line. These factors combine to create a robust economic model that supports long-term profitability even in a competitive generic drug market.
- Enhanced Supply Chain Reliability: Sourcing raw materials that are commodity chemicals rather than specialty extracts reduces the risk of supply disruptions caused by agricultural variability or geopolitical constraints. The robustness of the synthetic route ensures that production can be maintained consistently across different batches, providing procurement managers with the confidence to plan long-term contracts. This stability is crucial for maintaining the production schedules of finished dosage forms, preventing costly delays in the delivery of life-saving medications to patients. A reliable supply of high-quality intermediates is the backbone of a resilient pharmaceutical supply chain.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing common solvents and reagents that are easy to handle and recycle in a large-scale facility. The reduction in hazardous byproducts and the ability to manage waste streams effectively align with modern environmental regulations and sustainability goals. This compliance reduces the regulatory burden on manufacturing sites and facilitates faster approvals for process changes or capacity expansions. The ease of scale-up ensures that production can be ramped up quickly to meet surges in market demand without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specific pharmaceutical intermediate. These answers are derived directly from the technical specifications and beneficial effects described in the underlying patent documentation. Understanding these details helps stakeholders make informed decisions regarding sourcing and process adoption. The information provided here reflects the current state of the art in synthetic chemistry for this class of compounds.
Q: What are the purity levels achievable with this synthesis method?
A: According to patent CN110698397A, the optimized process achieves chemical purity exceeding 90.09%, which is critical for downstream API synthesis.
Q: Why is 3,4-dihydroxyphenylethanol preferred as a starting material?
A: Unlike traditional routes using expensive 1,2,3,4-tetrahydropapaverine, this method utilizes inexpensive and readily available 3,4-dihydroxyphenylethanol, significantly lowering raw material costs.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method features short reaction times, easy byproduct handling, and high yields (over 84%), making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6,7-Dimethoxy-3,4-Dihydroisoquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and commercialization of new neurological therapies. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from clinical trials to market launch. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 6,7-dimethoxy-3,4-dihydroisoquinoline meets the highest industry standards. We understand the complexities of managing impurity profiles and the importance of documentation for regulatory filings, providing our partners with complete transparency and support throughout the product lifecycle. Our commitment to technical excellence makes us the preferred choice for companies seeking a dependable partner in the fine chemical sector.
We invite you to engage with our technical procurement team to discuss how our capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized synthesis routes can improve your project economics. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments tailored to your unique development timelines. Let us collaborate to bring innovative treatments to patients faster and more efficiently through superior chemical manufacturing solutions.
