Advanced Radical Chlorination Strategy for Commercial Scale-Up of 2-Chloromethyl-3-Trifluoromethylpyridine
Advanced Radical Chlorination Strategy for Commercial Scale-Up of 2-Chloromethyl-3-Trifluoromethylpyridine
The pharmaceutical industry constantly seeks more efficient pathways to access critical heterocyclic building blocks, and patent CN112279802B presents a transformative approach to synthesizing 2-chloromethyl-3-trifluoromethylpyridine. This compound serves as a pivotal intermediate in the development of C5a receptor modulators, which are essential for treating innate immune responses, as well as ion channel blockers and psychotropic drugs. Traditionally, accessing this chloromethyl pyridine derivative required a cumbersome multi-step sequence involving nitrogen oxidation and subsequent functional group interconversions. However, the disclosed invention introduces a novel free-radical chlorination strategy that bypasses these inefficiencies. By leveraging a specific combination of electrophilic chlorinating reagents, radical initiators, and nitroxide radical precursors, the process achieves direct benzylic chlorination under mild thermal conditions. This technological leap not only simplifies the molecular construction but also addresses critical pain points regarding yield, selectivity, and operational safety, positioning it as a superior candidate for reliable pharmaceutical intermediate supplier networks aiming to optimize their supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of 2-chloromethyl-3-trifluoromethylpyridine was plagued by operational complexity and low atom economy. As illustrated in the conventional pathway, the process typically commenced with the oxidation of the pyridine nitrogen using meta-chloroperoxybenzoic acid (mCPBA), followed by a rearrangement or hydroxylation step using acetic anhydride and base, and finally, a chlorination step employing thionyl chloride (SOCl2). 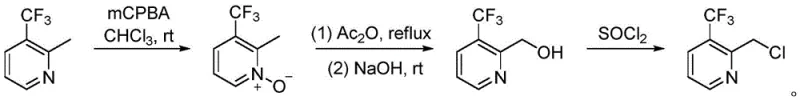 This three-step sequence inherently suffers from cumulative yield losses, where the efficiency of each individual step compounds to result in a significantly depressed overall output. Furthermore, the use of strong oxidants and corrosive chlorinating agents like thionyl chloride generates substantial hazardous waste, complicating environmental compliance and increasing disposal costs. The requirement for distinct reaction conditions for each step—ranging from room temperature oxidation to refluxing chlorination—demands extensive equipment turnover and energy consumption, making the conventional route economically unviable for cost reduction in pharmaceutical intermediates manufacturing on a large scale.
This three-step sequence inherently suffers from cumulative yield losses, where the efficiency of each individual step compounds to result in a significantly depressed overall output. Furthermore, the use of strong oxidants and corrosive chlorinating agents like thionyl chloride generates substantial hazardous waste, complicating environmental compliance and increasing disposal costs. The requirement for distinct reaction conditions for each step—ranging from room temperature oxidation to refluxing chlorination—demands extensive equipment turnover and energy consumption, making the conventional route economically unviable for cost reduction in pharmaceutical intermediates manufacturing on a large scale.
The Novel Approach
In stark contrast, the methodology outlined in patent CN112279802B collapses this entire sequence into a single, elegant transformation. The novel approach utilizes a radical-mediated mechanism where 2-methyl-3-trifluoromethylpyridine reacts directly with an electrophilic chlorinating source, such as N-chlorosuccinimide (NCS), in the presence of a radical initiator like AIBN. Crucially, the inclusion of a nitroxide radical precursor, such as N-hydroxysuccinimide, acts as a selectivity controller, guiding the radical species to the benzylic position while suppressing unwanted side reactions on the electron-deficient pyridine ring. This one-pot procedure operates effectively in common organic solvents like acetonitrile or dichloromethane at moderate temperatures between 50°C and 100°C. By eliminating the need for isolation of intermediates and avoiding the use of hazardous gases or aggressive reagents, this method drastically simplifies the workflow. The result is a process that is not only chemically superior in terms of selectivity but also logistically streamlined, offering a robust solution for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Radical-Mediated Benzylic Chlorination
The core of this technological advancement lies in the precise control of free radical chemistry to achieve site-selective functionalization. The reaction initiates when the thermal decomposition of the radical initiator, such as Azobisisobutyronitrile (AIBN), generates primary radical species. These radicals abstract a hydrogen atom from the methyl group at the 2-position of the pyridine ring, generating a stabilized benzylic radical intermediate. 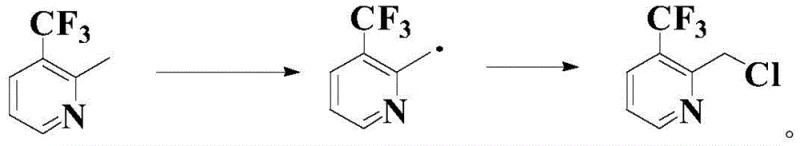 This specific intermediate is key to the process success; without the nitroxide radical precursor, the highly reactive benzylic radical might undergo uncontrolled polymerization or non-selective chlorination. The nitroxide precursor interacts with the radical intermediate, likely forming a transient adduct that moderates its reactivity, allowing for a controlled transfer of the chlorine atom from the electrophilic source (e.g., NCS). This mechanism ensures that the chlorination occurs exclusively at the methyl group rather than on the aromatic ring or the nitrogen atom. The electron-withdrawing nature of the trifluoromethyl group and the pyridine nitrogen usually deactivates the ring towards electrophilic attack, but the radical pathway bypasses these electronic constraints, targeting the weaker C-H bond of the methyl group. This mechanistic understanding allows R&D teams to fine-tune reaction parameters, such as the molar ratio of the nitroxide precursor (optimized between 0.01 to 0.2 equivalents), to maximize the formation of the desired 2-chloromethyl product while minimizing impurities.
This specific intermediate is key to the process success; without the nitroxide radical precursor, the highly reactive benzylic radical might undergo uncontrolled polymerization or non-selective chlorination. The nitroxide precursor interacts with the radical intermediate, likely forming a transient adduct that moderates its reactivity, allowing for a controlled transfer of the chlorine atom from the electrophilic source (e.g., NCS). This mechanism ensures that the chlorination occurs exclusively at the methyl group rather than on the aromatic ring or the nitrogen atom. The electron-withdrawing nature of the trifluoromethyl group and the pyridine nitrogen usually deactivates the ring towards electrophilic attack, but the radical pathway bypasses these electronic constraints, targeting the weaker C-H bond of the methyl group. This mechanistic understanding allows R&D teams to fine-tune reaction parameters, such as the molar ratio of the nitroxide precursor (optimized between 0.01 to 0.2 equivalents), to maximize the formation of the desired 2-chloromethyl product while minimizing impurities.
Furthermore, the choice of solvent and temperature plays a pivotal role in managing the radical chain propagation. Solvents like acetonitrile provide an ideal polarity balance to dissolve both the polar succinimide byproducts and the organic substrate, facilitating efficient collision frequency between reactants. The reaction temperature window of 70-90°C is critical; it is high enough to ensure the steady decomposition of the initiator and sustain the radical chain but low enough to prevent thermal degradation of the sensitive pyridine scaffold or the chlorinated product. From an impurity control perspective, this mechanism inherently limits the formation of poly-chlorinated byproducts because the introduction of the first chlorine atom strengthens the remaining C-H bonds and alters the electronic environment, making subsequent abstraction energetically less favorable. This self-limiting characteristic contributes to the high purity profiles observed in the examples, often exceeding 96% purity after simple workup, thereby reducing the burden on downstream purification processes.
How to Synthesize 2-Chloromethyl-3-Trifluoromethylpyridine Efficiently
The implementation of this synthesis route requires careful attention to reagent stoichiometry and thermal management to replicate the high yields reported in the patent data. The standard protocol involves dissolving the substrate, chlorinating agent, initiator, and precursor in the chosen solvent, followed by a controlled heating phase. The detailed standardized synthesis steps for replicating this high-efficiency route are provided in the guide below.
- Dissolve the electrophilic chlorinating agent (e.g., N-chlorosuccinimide), radical initiator (e.g., AIBN), substrate (2-methyl-3-trifluoromethylpyridine), and nitroxide radical precursor (e.g., N-hydroxysuccinimide) in an organic solvent such as acetonitrile.
- Heat the reaction mixture to a temperature range of 50-100°C, preferably around 80°C, and maintain stirring for 3 to 6 hours to ensure complete conversion.
- Upon completion, remove the solvent via rotary evaporation and purify the crude product using silica gel column chromatography to obtain high-purity 2-chloromethyl-3-trifluoromethylpyridine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from the conventional multi-step oxidation route to this direct radical chlorination represents a significant opportunity for value engineering. The primary driver of value is the drastic reduction in unit operations; moving from three distinct chemical transformations to a single reactor run fundamentally alters the cost structure of the molecule. This consolidation eliminates the need for intermediate isolation, drying, and quality control testing between steps, which are often hidden cost drivers in fine chemical manufacturing. Additionally, the replacement of expensive and hazardous reagents like mCPBA and thionyl chloride with more stable and manageable solids like N-chlorosuccinimide reduces raw material volatility and storage costs. The mild reaction conditions also imply lower energy consumption for heating and cooling cycles, contributing to a greener and more cost-effective production profile that aligns with modern sustainability goals.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived principally from the simplification of the synthetic tree. By removing two entire reaction steps, the manufacturer saves on labor, solvent usage, and waste disposal fees associated with those skipped stages. The use of catalytic amounts of radical initiators and precursors means that the bulk of the cost is driven by the commodity starting material, 2-methyl-3-trifluoromethylpyridine, rather than expensive proprietary reagents. Furthermore, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, which reduces the loss of material during purification and increases the overall mass balance efficiency. This leads to substantial cost savings without compromising the quality of the final API intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available, commodity-grade reagents. N-chlorosuccinimide, AIBN, and acetonitrile are standard inventory items for most chemical manufacturers, reducing the risk of supply bottlenecks associated with specialized oxidants. The robustness of the reaction conditions—tolerating a broad temperature range and various solvent systems—means that production is less susceptible to minor fluctuations in utility availability or equipment performance. This reliability ensures consistent delivery schedules for downstream pharmaceutical customers who depend on this intermediate for critical drug synthesis programs, thereby strengthening the partnership between the supplier and the end-user.
- Scalability and Environmental Compliance: Scaling this process from laboratory to pilot and eventually to commercial tonnage is straightforward due to the absence of exothermic hazards typical of peroxide oxidations or gas-evolving chlorinations. The reaction generates solid succinimide byproducts which can be easily filtered or washed away, simplifying the effluent treatment process. This ease of waste management facilitates compliance with increasingly stringent environmental regulations regarding halogenated waste and heavy metal residues (since no metal catalysts are used). The ability to run the reaction in standard glass-lined or stainless steel reactors without specialized pressure vessels further lowers the barrier to entry for large-scale production, ensuring a continuous and scalable supply of high-purity materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this radical chlorination technology. These answers are derived directly from the experimental data and claims within patent CN112279802B, providing a factual basis for evaluating the process feasibility.
Q: What are the key advantages of the radical chlorination method over traditional oxidation routes?
A: The radical chlorination method described in patent CN112279802B significantly reduces the synthetic route from three steps (oxidation, hydroxylation, chlorination) to a single direct substitution step. This eliminates the need for harsh reagents like thionyl chloride and mCPBA, resulting in higher overall yields, improved selectivity, and substantially lower production costs.
Q: Which radical initiators and precursors are most effective for this transformation?
A: The patent identifies Azobisisobutyronitrile (AIBN) as a highly effective radical initiator. For the nitroxide radical precursor, N-hydroxysuccinimide (NHS), N-hydroxyisoindole-1,3-dione, and 1-hydroxy-benzotriazole are preferred, with NHS demonstrating excellent selectivity in controlling the radical intermediate to prevent over-chlorination or side reactions.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for industrial scalability. It operates under mild conditions (50-100°C) using commercially available solvents like acetonitrile or dichloromethane. The short reaction time (3-6 hours) and simplified post-treatment (solvent removal and chromatography) make it highly amenable to commercial scale-up compared to multi-step conventional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloromethyl-3-Trifluoromethylpyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the global pharmaceutical supply chain. Our technical team has thoroughly analyzed the potential of the radical chlorination pathway described in CN112279802B and is prepared to leverage this knowledge for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-chloromethyl-3-trifluoromethylpyridine meets the exacting standards required for GMP API synthesis.
We invite you to collaborate with us to unlock the full commercial potential of this advanced synthetic route. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to discuss specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both cost and continuity in the competitive landscape of pharmaceutical intermediates.
