Revolutionizing 2-Chloromethyl-3-Trifluoromethylpyridine Production via Selective Radical Chlorination for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access critical heterocyclic building blocks, and the recent disclosure in patent CN112279802B marks a significant advancement in the synthesis of 2-chloromethyl-3-trifluoromethylpyridine. This specific pyridine derivative serves as a pivotal intermediate in the development of potent C5a receptor modulators, which are increasingly relevant in treating innate immune responses and inflammatory diseases. The traditional reliance on multi-step functionalization strategies has long been a bottleneck for R&D teams aiming to accelerate drug discovery timelines. By introducing a novel radical-mediated chlorination protocol, this technology offers a direct, high-yielding route that bypasses the cumbersome oxidation and hydroxylation steps previously required. For procurement and supply chain leaders, this shift represents not just a chemical optimization, but a strategic opportunity to secure a more reliable pharmaceutical intermediate supplier capable of delivering high-purity materials with reduced lead times and lower environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of 2-chloromethyl-3-trifluoromethylpyridine was predominantly achieved through a laborious multi-step sequence that severely impacted overall process efficiency and cost-effectiveness. As illustrated in the historical synthetic route, the conventional method typically initiates with the nitrogen oxidation of the starting pyridine, followed by a separate hydroxylation step to install the oxygen functionality, and finally a chlorination reaction to generate the desired chloromethyl group. 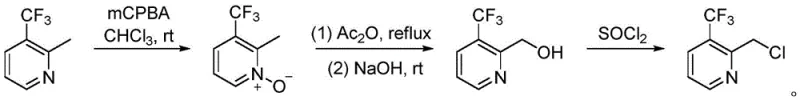 This fragmented approach inherently suffers from low total yield due to the accumulation of losses at each isolation and purification stage. Furthermore, the operational complexity involves handling multiple distinct reaction conditions, varying solvents, and potentially hazardous oxidizing agents, which complicates waste management and increases the burden on quality control laboratories. The fussy operation and high cost associated with these legacy methods make them less attractive for modern, lean manufacturing environments where speed and atom economy are paramount.
This fragmented approach inherently suffers from low total yield due to the accumulation of losses at each isolation and purification stage. Furthermore, the operational complexity involves handling multiple distinct reaction conditions, varying solvents, and potentially hazardous oxidizing agents, which complicates waste management and increases the burden on quality control laboratories. The fussy operation and high cost associated with these legacy methods make them less attractive for modern, lean manufacturing environments where speed and atom economy are paramount.
The Novel Approach
In stark contrast, the methodology disclosed in CN112279802B streamlines the entire transformation into a single, elegant pot reaction that directly converts 2-methyl-3-trifluoromethylpyridine into the target chloride. This novel approach leverages a sophisticated radical mechanism wherein an electrophilic chlorinating reagent, such as N-chlorosuccinimide (NCS), reacts in the presence of a radical initiator and a specific nitroxide radical precursor. By eliminating the need for pre-oxidation to an alcohol or N-oxide, the process drastically reduces the number of unit operations, thereby minimizing solvent consumption and energy usage. The ability to achieve high selectivity in a single step means that manufacturers can significantly reduce the footprint of their production facilities while simultaneously improving the throughput of high-purity OLED material or pharmaceutical intermediates. This transition from a linear, multi-step sequence to a convergent, one-pot strategy is a hallmark of green chemistry principles applied to industrial scale-up.
Mechanistic Insights into Nitroxide-Mediated Radical Chlorination
The core innovation of this technology lies in its precise control over radical reactivity, which is often a challenge in benzylic chlorination due to the risk of over-chlorination or ring substitution. The reaction mechanism initiates with the thermal decomposition of a radical initiator, such as azobisisobutyronitrile (AIBN), which generates free radicals that abstract a hydrogen atom from the methyl group of the pyridine substrate. 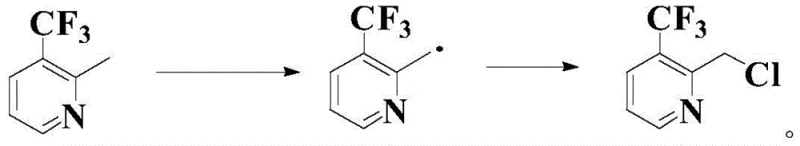 Crucially, the presence of the nitroxide radical precursor, such as N-hydroxysuccinimide or N-hydroxyisoindole-1,3-dione, acts as a mediator that stabilizes the transient carbon-centered radical intermediate. This stabilization prevents uncontrolled radical chain reactions that could lead to polymerization or degradation, ensuring that the radical species remains available for the subsequent chlorination step. The electrophilic chlorinating agent then efficiently traps this activated intermediate to form the carbon-chlorine bond, regenerating the catalytic cycle. This delicate balance allows the reaction to proceed under relatively mild thermal conditions, typically between 50-100°C, avoiding the extreme temperatures that often degrade sensitive fluorinated heterocycles.
Crucially, the presence of the nitroxide radical precursor, such as N-hydroxysuccinimide or N-hydroxyisoindole-1,3-dione, acts as a mediator that stabilizes the transient carbon-centered radical intermediate. This stabilization prevents uncontrolled radical chain reactions that could lead to polymerization or degradation, ensuring that the radical species remains available for the subsequent chlorination step. The electrophilic chlorinating agent then efficiently traps this activated intermediate to form the carbon-chlorine bond, regenerating the catalytic cycle. This delicate balance allows the reaction to proceed under relatively mild thermal conditions, typically between 50-100°C, avoiding the extreme temperatures that often degrade sensitive fluorinated heterocycles.
From an impurity control perspective, this mechanistic pathway offers superior selectivity compared to traditional ionic chlorination methods. In conventional acid-catalyzed chlorinations, there is a significant risk of electrophilic aromatic substitution on the electron-deficient pyridine ring, leading to difficult-to-remove regioisomers. However, the radical nature of this new process targets the benzylic C-H bond exclusively, as evidenced by the high purity profiles reported in the patent examples, where purities of 96.6% to 97.0% were achieved without extensive recrystallization. The nitroxide species effectively suppresses side reactions by modulating the concentration of free radicals in the solution, acting as a 'radical buffer' that maintains the reaction within the optimal kinetic window. For R&D directors, this implies a much cleaner crude profile, which simplifies downstream processing and reduces the load on purification columns, ultimately enhancing the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 2-Chloromethyl-3-Trifluoromethylpyridine Efficiently
Implementing this synthesis requires careful attention to the stoichiometric balance of the radical mediators to ensure optimal conversion. The patent outlines a robust protocol where the electrophilic chlorinating agent, substrate, radical initiator, and nitroxide precursor are dissolved in a polar aprotic solvent such as acetonitrile or dichloroethane. The detailed standardized synthesis steps below provide a clear roadmap for replicating the high yields observed in the patent examples, ensuring that technical teams can validate the process rapidly in pilot plant settings.
- Dissolve the electrophilic chlorinating agent (e.g., NCS), radical initiator (e.g., AIBN), substrate (2-methyl-3-trifluoromethylpyridine), and nitroxide precursor (e.g., N-hydroxysuccinimide) in an organic solvent like acetonitrile.
- Heat the reaction mixture to a controlled temperature range of 50-100°C, preferably around 80°C, and maintain stirring for 3 to 6 hours to ensure complete conversion.
- Upon completion, remove the solvent via rotary evaporation and purify the crude product using silica gel column chromatography to obtain the target chloride with >96% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this radical chlorination technology translates into tangible strategic advantages that go beyond simple chemical yield improvements. By collapsing a three-step sequence into a single operation, the manufacturing process inherently becomes less resource-intensive, requiring fewer reactor vessels, less solvent inventory, and reduced labor hours per kilogram of output. This consolidation of steps directly addresses the industry-wide challenge of cost reduction in API manufacturing, as it eliminates the cumulative losses and overhead costs associated with intermediate isolations. Furthermore, the use of stable, solid reagents like N-chlorosuccinimide and N-hydroxysuccinimide enhances supply chain reliability, as these materials are widely available from global chemical suppliers, reducing the risk of bottlenecks associated with specialized or hazardous gaseous reagents.
- Cost Reduction in Manufacturing: The elimination of the oxidation and hydroxylation steps removes the need for expensive oxidizing agents and the associated waste treatment costs, leading to substantial cost savings. By avoiding the isolation of unstable alcohol intermediates, the process also reduces material loss during transfer and filtration, optimizing the overall mass balance. The simplified workflow means that existing general-purpose reactors can be utilized without the need for specialized corrosion-resistant equipment often required for harsh chlorination conditions, further lowering capital expenditure requirements for production scale-up.
- Enhanced Supply Chain Reliability: The reliance on common organic solvents like acetonitrile and dichloroethane, combined with shelf-stable solid reagents, ensures that production schedules are not disrupted by the logistics of handling hazardous gases or cryogenic conditions. The robustness of the reaction across a temperature range of 50-100°C provides operational flexibility, allowing manufacturing partners to adapt to varying utility constraints without compromising product quality. This flexibility is critical for reducing lead time for high-purity pharmaceutical intermediates, enabling faster response to market demands for C5a receptor modulator precursors.
- Scalability and Environmental Compliance: The mild reaction conditions and high selectivity result in a cleaner waste stream with fewer byproducts, simplifying effluent treatment and ensuring compliance with increasingly stringent environmental regulations. The process avoids the generation of heavy metal waste or toxic gas emissions, aligning with the sustainability goals of modern chemical enterprises. Additionally, the short reaction time of 3-6 hours allows for higher batch turnover rates, facilitating the rapid scaling from kilogram-level R&D batches to multi-ton commercial production campaigns without significant process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this radical chlorination technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering a transparent view of the process capabilities and limitations for potential partners.
Q: Why is the nitroxide radical precursor critical in this chlorination process?
A: According to patent CN112279802B, the nitroxide radical precursor (such as N-hydroxysuccinimide) is essential for mediating the radical reaction. Without it, comparative examples show yields dropping drastically to as low as 4%, indicating that the precursor stabilizes the methyl radical intermediate and ensures selective chlorination at the benzylic position rather than ring chlorination or decomposition.
Q: What are the typical yield and purity metrics for this new method?
A: The patented method demonstrates robust performance with isolated yields reaching up to 75% and purity levels exceeding 97.0% after standard column chromatography purification. This represents a significant improvement over traditional multi-step oxidation routes which often suffer from cumulative yield losses.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for scalability. It utilizes commercially available reagents like N-chlorosuccinimide and AIBN, operates at moderate temperatures (50-100°C), and avoids hazardous gaseous chlorine, making it safer and easier to implement in standard chemical reactors compared to conventional chlorination methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloromethyl-3-Trifluoromethylpyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic routes play in the competitiveness of the global pharmaceutical market. Our team of expert chemists has extensively evaluated the radical chlorination methodology described in CN112279802B and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of materials that meet stringent purity specifications. Our rigorous QC labs are equipped to verify the absence of regioisomeric impurities and residual solvents, guaranteeing that every batch of 2-chloromethyl-3-trifluoromethylpyridine delivered is ready for immediate use in downstream coupling reactions.
We invite forward-thinking organizations to collaborate with us to leverage this advanced technology for their pipeline projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this streamlined route for your specific volume requirements. We encourage you to contact us today to discuss your project needs,索取 specific COA data, and review our comprehensive route feasibility assessments to ensure your supply chain is optimized for the future.
