Revolutionizing Porphyrin Functionalization via DDQ-Mediated Oxidative Coupling for Commercial Scale
Introduction to Next-Generation Porphyrin Synthesis
The landscape of organic synthesis for complex macrocycles is undergoing a significant transformation, driven by the urgent need for more efficient and environmentally benign methodologies in the production of high-value pharmaceutical intermediates. Patent CN110698487B introduces a groundbreaking preparation method for porphyrin derivatives featuring DDQ-mediated carbon-oxygen and carbon-carbon coupling at the meso-position. This technology addresses a critical bottleneck in the field of photodynamic therapy agents, where the structural diversification of porphyrin cores has historically been limited by harsh reaction conditions and low yields. By leveraging a non-metal oxidative strategy, this innovation enables the direct modification of 5,15-diaryl substituted metalloporphyrins without the prerequisite of functional group mediation, thereby streamlining the supply chain for advanced photosensitizers used in treating various cancers including breast and colorectal tumors.
For research and development directors overseeing the pipeline of novel therapeutic agents, the ability to rapidly access diverse porphyrin scaffolds is paramount. The traditional reliance on multi-step functionalization sequences often introduces impurities that are difficult to remove, complicating the regulatory approval process for new drug applications. The disclosed method offers a robust alternative that not only enhances the conjugated system of the porphyrin through the introduction of polar oxygen atoms but also improves water solubility, a key physicochemical property for biological efficacy. As a reliable porphyrin derivative supplier, understanding these mechanistic advancements allows us to offer clients superior intermediates that align with modern green chemistry principles while maintaining the rigorous purity standards required for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the derivatization of porphyrin at the meso-position has been a formidable challenge due to the inherent stability of the aromatic macrocycle and the lack of reactive sites for direct substitution. Conventional strategies typically involve a two-step sequence beginning with the bromination of the porphyrin core followed by a transition metal-catalyzed cross-coupling reaction, often utilizing expensive palladium complexes. This approach suffers from several intrinsic drawbacks, including the high cost of noble metal catalysts, the generation of toxic heavy metal waste that requires extensive downstream processing to meet pharmaceutical specifications, and the sensitivity of the reaction to moisture and oxygen. Furthermore, the pre-functionalization step adds significant time and material costs to the overall manufacturing process, reducing the atom economy and increasing the environmental footprint of producing these specialized fine chemicals.
In addition to economic inefficiencies, the conventional palladium-catalyzed routes often exhibit limited substrate scope, particularly when attempting to introduce bulky or electron-rich groups which can lead to catalyst deactivation or poor regioselectivity. The formation of homocoupling byproducts is another persistent issue that compromises the yield and purity of the final product, necessitating complex chromatographic separations that are difficult to scale. For procurement managers focused on cost reduction in photosensitizer manufacturing, these inefficiencies translate directly into higher raw material expenses and longer lead times. The dependency on scarce transition metals also introduces supply chain volatility, as the availability and price of palladium can fluctuate wildly based on geopolitical factors, posing a risk to the continuity of production for critical medical intermediates.
The Novel Approach
The innovative methodology described in the patent data circumvents these traditional hurdles by employing 2,3-dichloro-5,6-dicyano-p-benzoquinone (DDQ) as a potent non-metal oxidant to drive the cross-coupling reaction directly. This single-step oxidative coupling strategy allows for the direct formation of carbon-oxygen or carbon-carbon bonds between the porphyrin meso-position and phenolic substrates under remarkably mild conditions. For carbon-oxygen bond formation, the process utilizes scandium trifluoromethanesulfonate as a Lewis acid catalyst in 1,2-dichloroethane at elevated temperatures, achieving high conversion rates without the need for pre-halogenation. This represents a paradigm shift in porphyrin chemistry, enabling the efficient synthesis of meso-O substituted derivatives that were previously inaccessible or economically unviable to produce.
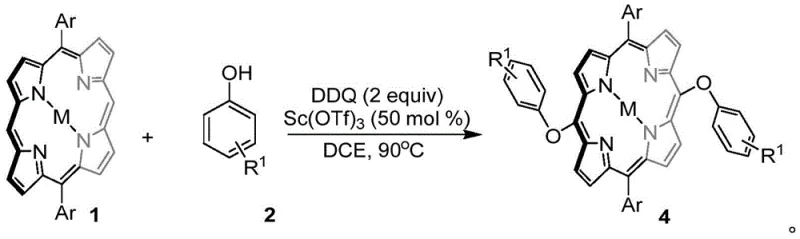
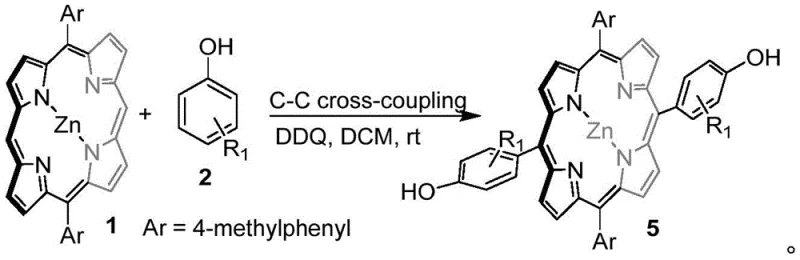
Moreover, the versatility of this DDQ-mediated system extends to carbon-carbon bond formation when specific steric conditions are met, such as using phenol derivatives with large steric hindrance around the hydroxyl group or naphthol substrates. In these instances, the reaction proceeds smoothly in dichloromethane at room temperature, demonstrating exceptional chemoselectivity that avoids self-coupling of the porphyrin or unwanted side reactions between the nucleophile and the oxidant. This dual capability to toggle between C-O and C-C bond formation simply by adjusting the substrate and solvent conditions provides a powerful tool for medicinal chemists to explore new chemical space. The elimination of transition metal catalysts not only reduces the cost of goods sold but also simplifies the purification workflow, ensuring that the final high-purity porphyrin derivatives are free from toxic metal residues that could compromise patient safety.
Mechanistic Insights into DDQ-Mediated Oxidative Cross-Coupling
The underlying mechanism of this transformation involves a sophisticated interplay of single electron transfer (SET) processes and radical stabilization dynamics that distinguish it from classical ionic reactions. DDQ acts as a strong electron acceptor, facilitating the oxidation of the electron-rich phenol substrate to generate a phenoxy radical intermediate. In the presence of the metalloporphyrin, which serves as a nucleophilic partner, this radical species attacks the meso-position of the porphyrin ring. The use of scandium triflate in the C-O coupling pathway is critical, as the Lewis acidic scandium center coordinates with the phenolic oxygen, enhancing its electrophilicity and stabilizing the transition state for oxygen attack, thereby favoring the formation of the ether linkage over the carbon-carbon bond. This precise control over the reaction trajectory ensures high regioselectivity, directing the substitution exclusively to the meso-positions of the 5,15-diaryl substituted porphyrin framework.
Conversely, when sterically hindered phenols are employed, the stability of the phenoxy radical shifts, and the reaction pathway favors radical coupling at the ortho-carbon position of the phenol ring with the porphyrin meso-carbon, resulting in a direct C-C bond. This mechanistic divergence highlights the subtle balance between steric and electronic factors that govern the outcome of the oxidative coupling. The ability to harness these radical pathways without the need for external light sources or harsh initiators underscores the robustness of the DDQ system. For technical teams evaluating process feasibility, understanding that the reaction avoids the formation of unstable intermediates common in metal-catalyzed cycles means fewer side products and a cleaner impurity profile. This mechanistic clarity allows for better prediction of reaction outcomes when scaling up to commercial batches, ensuring consistent quality across different production runs.
How to Synthesize Meso-O Substituted Porphyrin Efficiently
The practical implementation of this synthesis route is designed for operational simplicity, requiring standard laboratory equipment and readily available reagents that are compatible with existing manufacturing infrastructure. The process begins by dissolving the 5,15-diaryl substituted metalloporphyrin and the chosen phenolic compound in 1,2-dichloroethane, followed by the addition of the scandium catalyst and DDQ oxidant. The reaction mixture is then heated to 90°C and stirred for approximately three hours, a duration sufficient to ensure complete consumption of the starting porphyrin material as monitored by thin-layer chromatography. Upon completion, the solvent is removed under reduced pressure, and the resulting crude solid is subjected to column chromatography to isolate the pure meso-O substituted product with high fidelity.
- Dissolve 5,15-diaryl substituted metalloporphyrin and phenolic compounds in 1,2-dichloroethane solvent within a reaction vessel.
- Add scandium trifluoromethanesulfonate catalyst and DDQ oxidant, then heat the mixture to 90°C for approximately 3 hours.
- Remove solvent via spin-drying and purify the crude product using column chromatography to isolate the target meso-O substituted porphyrin.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this DDQ-mediated technology offers substantial benefits that extend beyond mere technical performance, directly impacting the bottom line and supply chain resilience. The most immediate advantage is the drastic simplification of the synthetic route, which eliminates the need for a separate bromination step and the subsequent palladium-catalyzed coupling. This consolidation of steps reduces the total processing time and minimizes the handling of hazardous intermediates, leading to significant operational cost savings. Furthermore, the exclusion of expensive noble metal catalysts removes a major variable cost component, shielding the production budget from the volatility of the precious metals market and ensuring more predictable pricing for long-term supply contracts.
- Cost Reduction in Manufacturing: The elimination of palladium catalysts and the associated ligand systems results in a direct reduction in raw material expenditures, while the simplified workup procedure reduces solvent consumption and waste disposal costs. By avoiding the need for specialized metal scavengers to meet residual metal limits, the downstream processing becomes more efficient and less resource-intensive. This streamlined approach allows for a more competitive pricing structure for the final porphyrin intermediates, providing a clear economic advantage over traditional synthesis methods that rely on multi-step metal-catalyzed sequences.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as DDQ, scandium triflate, and common solvents like dichloromethane ensures a stable and secure supply of reagents, mitigating the risk of production delays caused by shortages of specialized catalysts. The robustness of the reaction conditions, which tolerate a wide range of functional groups on the phenol substrate, allows for flexibility in sourcing raw materials, enabling procurement teams to qualify multiple vendors for key inputs. This diversification of the supply base strengthens the overall resilience of the manufacturing network against geopolitical disruptions or logistical bottlenecks.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of toxic heavy metals make this process inherently safer and more environmentally friendly, aligning with increasingly stringent global regulations on chemical manufacturing. The reduced generation of hazardous waste simplifies compliance with environmental standards and lowers the cost of waste treatment. Additionally, the high selectivity of the reaction minimizes the formation of byproducts, improving the overall atom economy and facilitating easier scale-up from pilot plant to full commercial production without the need for extensive process re-optimization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel porphyrin synthesis technology, providing clarity for stakeholders evaluating its potential integration into their supply chains. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What are the primary advantages of DDQ-mediated coupling over traditional palladium catalysis?
A: The DDQ-mediated method eliminates the need for expensive transition metal catalysts like palladium and avoids pre-functionalization steps such as bromination, significantly reducing raw material costs and simplifying the purification process by removing heavy metal residues.
Q: How does the reaction control selectivity between carbon-oxygen and carbon-carbon bonds?
A: Chemoselectivity is primarily governed by the steric hindrance of the phenol substrate; bulky groups around the hydroxyl favor carbon-carbon coupling, while less hindered phenols in the presence of Sc(OTf)3 favor carbon-oxygen bond formation at the porphyrin meso-position.
Q: Is this synthesis method scalable for industrial production of photosensitizers?
A: Yes, the process utilizes mild reaction conditions, commercially available oxidants like DDQ, and common solvents such as dichloromethane or dichloroethane, making it highly suitable for scale-up from laboratory to commercial manufacturing volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Porphyrin Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the DDQ-mediated coupling technology in advancing the development of next-generation photosensitizers and functional materials. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering high-purity porphyrin derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite pharmaceutical and fine chemical companies to collaborate with us to leverage this cutting-edge synthesis method for their specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, along with specific COA data and route feasibility assessments to validate the commercial viability of your target molecules. Let us help you accelerate your R&D timeline and optimize your manufacturing costs with our reliable supply of advanced porphyrin intermediates.
