Advanced DDQ-Mediated Synthesis of Meso-Functionalized Porphyrins for Commercial Photosensitizer Production
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to functionalize complex macrocycles like porphyrins, which serve as critical scaffolds for next-generation photosensitizers and electronic materials. Patent CN110698487A introduces a groundbreaking DDQ-mediated preparation method for porphyrin meso-position carbon-oxygen and carbon-carbon coupled derivatives that fundamentally shifts the paradigm of porphyrin derivatization. Unlike traditional approaches that rely heavily on pre-functionalized substrates or expensive transition metal catalysts, this innovation utilizes 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) as a potent oxidant to drive direct cross-coupling reactions between 5,15-diaryl substituted metalloporphyrins and various phenolic compounds. This technical breakthrough offers a streamlined pathway to access high-value intermediates with enhanced water solubility and expanded conjugated systems, addressing long-standing challenges in the synthesis of photodynamic therapy agents and organic semiconductors. For R&D teams and procurement strategists alike, this patent represents a significant opportunity to optimize supply chains and reduce dependency on scarce noble metals while maintaining rigorous purity standards required for clinical and electronic applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of porphyrin macrocycles at the meso-position has been fraught with synthetic inefficiencies and economic bottlenecks that hinder large-scale commercialization. Conventional strategies often necessitate the use of transition metal catalysts such as palladium or nickel to facilitate Suzuki-Miyaura or Kumada cross-coupling reactions, which introduce substantial cost burdens due to the high price of these noble metals and the ligands required to stabilize them. Furthermore, these metal-catalyzed processes frequently demand harsh reaction conditions, including high temperatures and inert atmospheres, which complicate process safety and increase energy consumption in industrial settings. A critical drawback of these traditional methods is the difficulty in removing trace metal residues from the final product, a stringent requirement for pharmaceutical intermediates intended for human use, often necessitating additional purification steps that drastically lower overall yields. Additionally, many existing protocols require pre-functionalized porphyrin substrates, such as brominated derivatives, adding extra synthetic steps and generating hazardous waste streams that conflict with modern green chemistry principles and environmental compliance regulations.
The Novel Approach
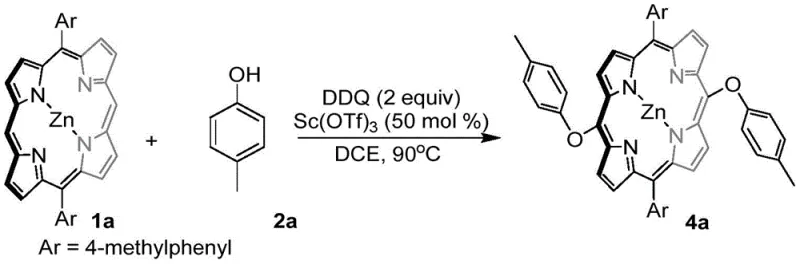
The novel DDQ-mediated oxidative cross-coupling strategy described in the patent data offers a transformative solution by enabling direct C-O and C-C bond formation without the mediation of functional groups or expensive transition metal catalysts. This approach leverages the strong oxidizing power of DDQ, potentially assisted by Lewis acids like scandium trifluoromethanesulfonate, to activate the porphyrin ring and phenolic substrates under relatively mild conditions, typically ranging from room temperature to 90°C in common solvents like dichloroethane. By bypassing the need for halogenated precursors and noble metal catalysts, this method significantly simplifies the synthetic route, reducing the number of unit operations and minimizing the generation of toxic heavy metal waste. The versatility of this system is demonstrated by its ability to accommodate a wide range of phenolic substrates, including those with varying steric hindrance and electronic properties, allowing for the precise tuning of the porphyrin's physicochemical characteristics. This flexibility not only accelerates the development of new photosensitizers with improved water solubility but also provides a cost-effective platform for the commercial scale-up of complex porphyrin derivatives essential for the agrochemical and electronic material sectors.
Mechanistic Insights into DDQ-Mediated Oxidative Cross-Coupling
The mechanistic underpinning of this transformation relies on the unique ability of DDQ to facilitate single-electron transfer (SET) processes that generate reactive radical intermediates capable of forming new bonds at the electron-rich meso-positions of the porphyrin ring. In the presence of a Lewis acid catalyst such as Sc(OTf)3, the coordination to the phenolic oxygen enhances the nucleophilicity or facilitates the formation of a phenoxyl radical species, which then attacks the oxidized porphyrin intermediate. This pathway effectively competes with homocoupling reactions, ensuring high chemoselectivity for the desired cross-coupled product over self-coupled byproducts. The reaction proceeds through a cationic or radical-cationic manifold where the aromaticity of the porphyrin is temporarily disrupted to allow for substitution, followed by rearomatization driven by the elimination of protons and the reduction of the DDQ oxidant. Understanding this mechanism is crucial for R&D directors aiming to optimize reaction parameters, as the balance between oxidant strength and substrate stability determines the final yield and purity profile of the synthesized intermediates.
Impurity control in this DDQ-mediated system is inherently superior to metal-catalyzed alternatives due to the absence of metal-ligand complexes that can degrade into hard-to-remove side products. The primary impurities typically arise from over-oxidation of the phenolic substrate or incomplete conversion of the starting porphyrin, both of which can be effectively managed by controlling the stoichiometry of the DDQ oxidant and the reaction temperature. The use of scandium triflate as a co-catalyst further refines the selectivity by directing the attack to the specific meso-carbon atoms, minimizing the formation of regioisomers that could complicate downstream purification. For quality assurance teams, this means that the resulting crude products often require fewer chromatographic passes to achieve the stringent purity specifications demanded by regulatory bodies for API intermediates. The robustness of this oxidative protocol ensures consistent batch-to-batch reproducibility, a critical factor for supply chain heads managing the inventory of high-value specialty chemicals where variability can lead to significant production delays.
How to Synthesize Meso-Functionalized Porphyrins Efficiently
The synthesis of these high-value porphyrin derivatives follows a straightforward protocol that is easily adaptable to both laboratory discovery and pilot plant operations, leveraging standard glassware and heating equipment found in most chemical facilities. The process begins with the dissolution of the 5,15-diaryl substituted metalloporphyrin and the chosen phenolic coupling partner in a chlorinated solvent, followed by the sequential addition of the Lewis acid catalyst and the DDQ oxidant under controlled thermal conditions. Reaction monitoring is typically conducted via thin-layer chromatography or HPLC to ensure complete consumption of the starting porphyrin material before proceeding to workup. Upon completion, the reaction mixture is concentrated under reduced pressure to remove the solvent, yielding a crude solid that contains the target meso-substituted porphyrin along with reduced DDQ byproducts. The final purification is achieved through standard column chromatography techniques using silica gel, which effectively separates the highly colored porphyrin product from polar impurities, delivering a material suitable for subsequent biological or electronic testing.
- Dissolve 5,15-diaryl substituted metalloporphyrin and phenolic compound in 1,2-dichloroethane solvent within a reaction vessel.
- Add scandium trifluoromethanesulfonate catalyst and DDQ oxidant, then heat the mixture to 90°C and stir for approximately 3 hours.
- Remove solvent via rotary evaporation to obtain crude product, followed by column chromatography purification to isolate the final meso-O substituted porphyrin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this DDQ-mediated synthesis route presents a compelling value proposition centered on cost reduction, supply security, and operational simplicity. By eliminating the reliance on volatile and expensive palladium or nickel catalysts, manufacturers can significantly stabilize their raw material costs and insulate their production budgets from the fluctuations of the precious metals market. The simplified workflow, which removes the need for pre-halogenation steps and complex ligand systems, translates directly into reduced processing time and lower labor costs per kilogram of finished product. Furthermore, the use of commodity chemicals like DDQ and scandium triflate, which are readily available from multiple global suppliers, mitigates the risk of supply chain disruptions that often plague specialized catalytic reagents. This resilience is critical for maintaining continuous production schedules and meeting the just-in-time delivery requirements of downstream pharmaceutical and electronic clients who cannot afford interruptions in their own manufacturing lines.
- Cost Reduction in Manufacturing: The economic benefits of this technology are driven primarily by the removal of costly transition metal catalysts and the reduction of synthetic steps required to prepare the starting materials. Without the need for expensive ligands or rigorous inert atmosphere conditions often associated with metal catalysis, the overhead costs for reactor usage and gas consumption are substantially lowered. Additionally, the high efficiency of the DDQ oxidation minimizes the loss of valuable porphyrin starting materials, improving the overall mass balance and reducing the cost of goods sold. These factors combine to create a more lean and profitable manufacturing process that allows for competitive pricing in the global market for specialty chemical intermediates.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the key reagents, including DDQ and various phenols, are produced on a massive industrial scale for diverse applications, ensuring a steady and reliable supply stream. Unlike proprietary catalyst systems that may be sourced from a single vendor, the commoditized nature of the oxidants and solvents used in this process allows procurement teams to negotiate better terms and maintain safety stocks without fear of obsolescence. This diversification of the supply base reduces the vulnerability of the production line to geopolitical tensions or logistical bottlenecks, ensuring that delivery commitments to key accounts in the pharma and agrochemical sectors are consistently met with high reliability.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is markedly improved by the avoidance of heavy metal waste, which simplifies effluent treatment and reduces the regulatory burden associated with hazardous waste disposal. The reaction conditions are mild enough to be safely scaled from gram to ton quantities without requiring exotic high-pressure equipment, facilitating a smoother technology transfer from R&D to commercial production. This scalability ensures that as demand for porphyrin-based photosensitizers grows, the manufacturing capacity can be expanded rapidly to meet market needs while adhering to increasingly strict environmental protection standards and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this DDQ-mediated porphyrin synthesis technology, providing clarity for stakeholders evaluating its integration into their existing workflows. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these nuances helps in assessing the feasibility of adopting this method for specific project requirements and anticipating potential operational adjustments needed for successful deployment.
Q: What are the primary advantages of DDQ-mediated coupling over traditional metal-catalyzed methods?
A: The DDQ-mediated method eliminates the need for expensive transition metal catalysts like palladium or nickel, significantly reducing raw material costs and simplifying the removal of toxic metal residues from the final pharmaceutical intermediate.
Q: Does this synthesis method support large-scale commercial production?
A: Yes, the process utilizes commercially available reagents and standard solvents like dichloroethane, operating under mild conditions that are highly amenable to scale-up from laboratory grams to multi-ton annual production capacities.
Q: How does this method improve the water solubility of porphyrin derivatives?
A: By introducing polar oxygen atoms directly into the porphyrin meso-position through carbon-oxygen bond formation, the conjugated system is expanded, which inherently enhances the water solubility crucial for biological applications like photodynamic therapy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Porphyrin Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the DDQ-mediated synthesis route for producing high-purity porphyrin derivatives and are fully equipped to bring this technology to commercial fruition for our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of porphyrin intermediate meets the exacting standards required for photodynamic therapy applications and advanced electronic materials. Our commitment to quality and consistency makes us the preferred choice for companies seeking a dependable source of complex macrocyclic intermediates.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be tailored to your specific product portfolio and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements and supply chain configuration. We encourage you to reach out for specific COA data and route feasibility assessments that demonstrate our capability to deliver superior value and performance. Let us collaborate to optimize your supply chain and accelerate the development of next-generation porphyrin-based solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
