Advanced Synthesis of Tetrafluorobenzyl-5-Aminosalicylic Acid Derivatives for Neurodegenerative Therapeutics
The pharmaceutical landscape for treating acute and chronic neurodegenerative diseases, such as Alzheimer's, Parkinson's, and Huntington's disease, demands intermediates of exceptional purity and structural integrity. Patent CN101180263B introduces a groundbreaking preparation method for tetrafluorobenzyl-5-aminosalicylic acid derivatives and their pharmaceutically acceptable salts, addressing critical limitations found in prior art. This technology leverages a unique tetrafluorobenzylidene-5-aminosalicylic acid intermediate to circumvent the formation of stubborn dimeric impurities that have historically plagued the synthesis of this neuroprotective scaffold. By shifting from a direct alkylation approach to a condensation-hydrogenation sequence, the invention not only enhances chemical purity but also optimizes the physicochemical properties of the final drug substance through the formation of stable alkali metal salts. For R&D teams and procurement strategists alike, this patent represents a pivotal shift towards more robust and scalable manufacturing processes for complex fluorinated pharmaceutical intermediates.
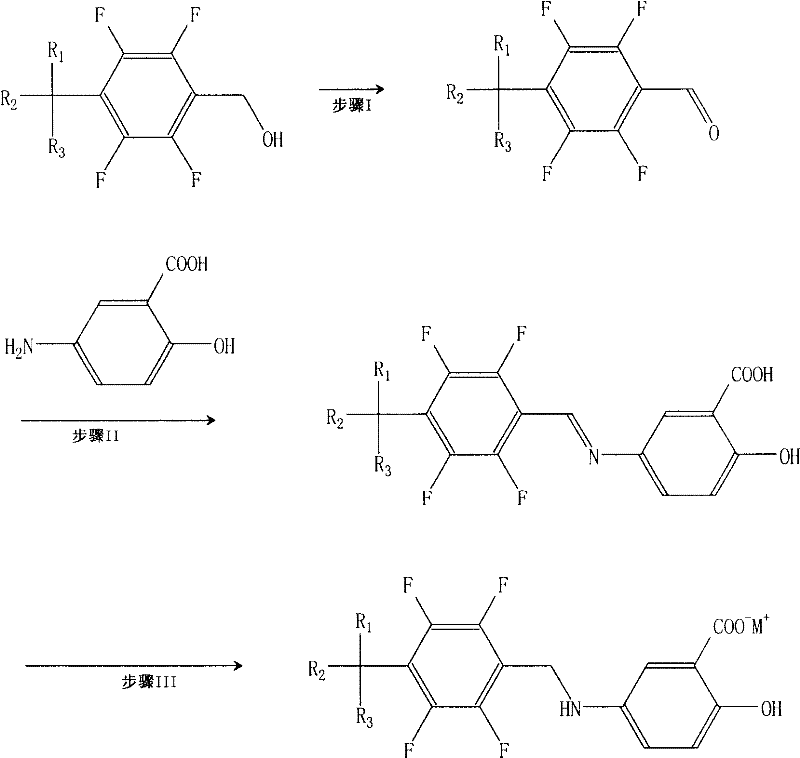
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tetrafluorobenzyl-5-aminosalicylic acid derivatives relied on the direct nucleophilic substitution of a nitrobenzene-derived aniline with tetrafluorotoluene bromide, as illustrated in older Korean patent publications. This conventional pathway, while conceptually straightforward, suffers from a severe inherent flaw: the generation of a persistent dimeric byproduct, chemically defined as a bis-benzylated species. This dimer forms because the initially produced secondary amine product retains nucleophilic character, allowing it to react further with the remaining tetrafluorobenzyl bromide starting material. The presence of this dimer, often exceeding 1% of the total crude product, creates a significant bottleneck in downstream processing because it cannot be easily removed through standard recrystallization techniques. Consequently, manufacturers are forced to employ complex and costly purification protocols to meet the stringent impurity thresholds of less than 0.1% required for pharmaceutical applications, drastically reducing overall yield and inflating production costs.
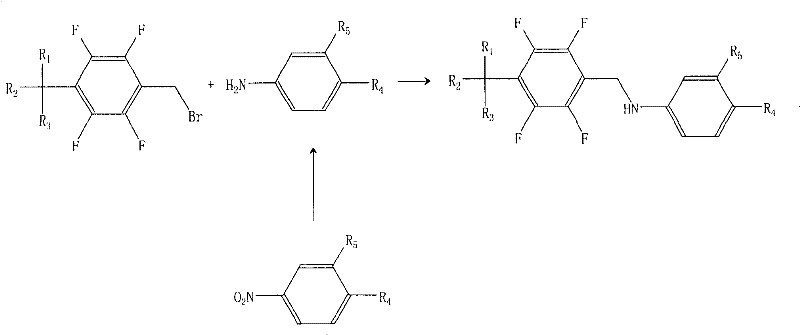
The Novel Approach
In stark contrast to the problematic direct alkylation, the methodology disclosed in CN101180263B employs a strategic detour through a tetrafluorobenzylidene-5-aminosalicylic acid imine intermediate. This innovative route begins with the oxidation of tetrafluorobenzyl alcohol to the corresponding aldehyde, followed by a dehydration condensation with 5-aminosalicylic acid. This condensation step effectively masks the amine functionality as an imine bond, thereby preventing the premature nucleophilic attacks that lead to dimerization. The final step involves the selective catalytic hydrogenation of this imine intermediate to yield the desired secondary amine. By decoupling the carbon-nitrogen bond formation from the potential over-alkylation side reactions, this novel approach virtually eliminates the formation of the dimeric impurity at the source. This fundamental change in synthetic logic allows for a much simpler workup procedure, often requiring only basic recrystallization to achieve high-purity standards suitable for clinical use.
Mechanistic Insights into Imine-Mediated Synthesis and Salt Stabilization
The core mechanistic advantage of this process lies in the kinetic control afforded by the imine intermediate. In the first stage, the oxidation of the benzyl alcohol to the aldehyde using reagents like pyridinium chlorochromate establishes the electrophilic center necessary for coupling. Subsequently, the reaction with 5-aminosalicylic acid proceeds via a nucleophilic addition-elimination mechanism to form the Schiff base (imine). Crucially, the nitrogen atom in the imine is significantly less nucleophilic than the free amine in the final product, rendering it inert towards the remaining electrophiles in the reaction mixture. This temporary deactivation is the key to suppressing the dimerization side reaction observed in Formula III. Following this, the catalytic hydrogenation using platinum on carbon or similar catalysts reduces the C=N double bond to a C-N single bond under mild conditions. This step is highly selective, preserving the sensitive phenolic hydroxyl and carboxylic acid moieties on the salicylic acid ring while efficiently generating the target tetrafluorobenzyl amine structure.
Beyond the synthetic route, the patent provides profound insights into the stabilization of the final molecule through salt formation. The free acid form of tetrafluorobenzyl-5-aminosalicylic acid exhibits poor aqueous solubility, which poses challenges for bioavailability and formulation. The inventors systematically evaluated various counterions, discovering that alkali metal salts—specifically sodium, potassium, and lithium—offer a dramatic improvement in solubility profiles. Mechanistically, the ionic nature of these salts disrupts the strong intermolecular hydrogen bonding networks present in the neutral acid crystal lattice, facilitating rapid dissolution in physiological media. Furthermore, stability studies indicate that the potassium salt, in particular, demonstrates superior resistance to degradation under accelerated storage conditions compared to the sodium and lithium analogues. This enhanced stability is likely attributed to the optimal lattice energy and hydration shell dynamics of the potassium cation, which protects the susceptible amine and phenol groups from oxidative degradation over time.
How to Synthesize Tetrafluorobenzyl-5-Aminosalicylic Acid Efficiently
The synthesis of this high-value neuroprotective intermediate is streamlined into three distinct operational stages that can be seamlessly integrated into a multi-purpose chemical reactor setup. The process begins with the oxidation of the fluorinated alcohol, followed by the crucial condensation step where molecular sieves are employed to drive the equilibrium towards imine formation by removing water. The final hydrogenation step utilizes standard heterogeneous catalysis equipment, making the technology highly transferable to existing manufacturing infrastructure. Detailed standardized synthesis steps, including specific molar ratios, solvent choices, and temperature profiles for each stage, are outlined in the technical guide below to ensure reproducibility and safety during scale-up operations.
- Oxidize tetrafluorobenzyl alcohol to tetrafluorobenzaldehyde using pyridinium chlorochromate in dichloromethane.
- Perform dehydration condensation between the resulting aldehyde and 5-aminosalicylic acid using molecular sieves to form the tetrafluorobenzylidene-5-aminosalicylic acid imine intermediate.
- Hydrogenate the imine intermediate using a platinum catalyst in an alcohol solvent to yield the final tetrafluorobenzyl-5-aminosalicylic acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond mere chemical elegance. The primary value driver is the significant simplification of the purification train. By eliminating the formation of the difficult-to-remove dimer impurity, the process removes the need for extensive chromatographic separations or multiple recrystallization cycles that typically erode yield and increase solvent consumption. This reduction in processing complexity translates directly into lower manufacturing costs and a reduced environmental footprint, aligning with modern green chemistry initiatives. Furthermore, the reliance on robust, commodity-grade reagents such as tetrafluorobenzyl alcohol and 5-aminosalicylic acid ensures a stable and resilient supply chain, minimizing the risk of raw material shortages that often plague specialized fluorinated syntheses.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the drastic reduction in waste generation and solvent usage associated with impurity removal. Since the dimeric byproduct is prevented from forming rather than removed post-reaction, the overall mass balance of the process is significantly improved, leading to higher effective yields of the active pharmaceutical ingredient. Additionally, the ability to use simple recrystallization for final polishing reduces the capital expenditure required for specialized purification equipment, allowing for more flexible and cost-effective production scheduling across different facility classes.
- Enhanced Supply Chain Reliability: The synthetic pathway utilizes widely available starting materials and standard catalytic hydrogenation techniques, which reduces dependency on exotic or single-source reagents. This commoditization of the supply base enhances the reliability of delivery timelines, as alternative vendors can be easily qualified for key inputs like the fluorinated alcohol or the platinum catalyst. Moreover, the improved stability of the potassium and sodium salt forms extends the shelf-life of the stored intermediate, providing greater flexibility in inventory management and reducing the risk of stock write-offs due to degradation during long-term storage or transit.
- Scalability and Environmental Compliance: The three-step sequence is inherently scalable, moving smoothly from laboratory benchtop to pilot plant and full commercial production without requiring fundamental changes to the reaction chemistry. The avoidance of harsh alkylation conditions and the use of catalytic hydrogenation instead of stoichiometric reducing agents minimizes the generation of hazardous waste streams. This cleaner profile simplifies regulatory compliance and waste disposal logistics, ensuring that the manufacturing process remains sustainable and compliant with increasingly stringent environmental regulations governing fluorinated organic synthesis.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on impurity control, salt selection, and process robustness. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing portfolio of neurodegenerative disease therapeutics.
Q: Why is the imine intermediate strategy superior to direct alkylation for this compound?
A: Direct alkylation of the aniline with tetrafluorobenzyl bromide leads to significant dimer formation (>1%) due to side reactions with the secondary amine product. The imine intermediate route completely bypasses this nucleophilic substitution pathway, ensuring higher purity without complex purification.
Q: Which salt forms offer the best stability and solubility for formulation?
A: The patent data indicates that alkali metal salts, specifically sodium, potassium, and lithium salts, exhibit significantly improved water solubility compared to the free acid. Among these, the potassium salt demonstrates superior stability in both solution and solid states under accelerated aging conditions.
Q: What are the critical reaction conditions for the hydrogenation step?
A: The reduction of the imine bond is effectively achieved using a platinum catalyst in an alcohol solvent under mild pressure (approximately 4 atm) and temperature (20-25°C). This contact reduction ensures selective conversion to the secondary amine without affecting other sensitive functional groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrafluorobenzyl-5-Aminosalicylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of next-generation treatments for neurodegenerative disorders. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN101180263B can be executed with precision and consistency. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect trace impurities, guaranteeing that every batch of tetrafluorobenzyl-5-aminosalicylic acid derivative meets the exacting standards required for clinical trials and commercial drug launch.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain and reduce overall project costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits of switching to this dimer-free process. We encourage potential partners to contact us directly to obtain specific COA data and comprehensive route feasibility assessments tailored to your unique production requirements and timeline constraints.
