Advanced Synthesis of Tetrafluorobenzyl-5-Aminosalicylic Acid Salts for Neurodegenerative Therapies
The pharmaceutical landscape for treating acute and chronic neurodegenerative disorders, such as Alzheimer's disease, Parkinson's disease, and Huntington's disease, demands intermediates of exceptional purity and structural integrity. Patent CN101180263A introduces a groundbreaking preparation method for tetrafluorobenzyl-5-aminosalicylic acid derivatives and their pharmaceutically acceptable salts. These compounds have demonstrated significant therapeutic potential in preventing and treating ischemic brain disorders and epilepsy. The core innovation lies in a novel synthetic pathway that circumvents the formation of persistent dimer impurities, a longstanding challenge in the prior art. By shifting from a direct alkylation strategy to a reductive amination approach via a stable imine intermediate, this technology offers a robust solution for producing high-quality active pharmaceutical ingredients (APIs). For R&D teams and procurement specialists seeking a reliable pharmaceutical intermediates supplier, understanding the mechanistic advantages of this route is critical for ensuring supply chain continuity and product efficacy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tetrafluorobenzyl-5-aminosalicylic acid derivatives relied on the direct reaction of 5-aminosalicylic acid with tetrafluorobenzyl bromide. As illustrated in the prior art reaction scheme, this conventional nucleophilic substitution pathway is fraught with significant chemical inefficiencies. The primary drawback is the unavoidable formation of a dimer impurity, chemically defined as a bis-substituted byproduct, which often constitutes greater than 1% of the total reaction mass. This dimer arises from a secondary side reaction where the initially formed secondary amine group reacts further with the remaining tetrafluorobenzyl bromide starting material. Removing this structurally similar impurity is notoriously difficult, often requiring complex and yield-reducing purification protocols such as repeated recrystallization or preparative chromatography. Consequently, the overall production costs are inflated, and the final purity specifications required for clinical applications are harder to meet consistently.
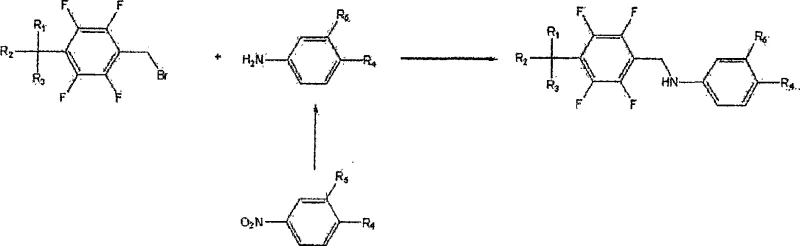
The Novel Approach
In stark contrast, the methodology disclosed in CN101180263A employs a strategic three-step sequence that fundamentally alters the reaction trajectory to suppress dimerization. Instead of direct alkylation, the process initiates with the oxidation of tetrafluorobenzyl alcohol to the corresponding aldehyde, followed by a dehydration condensation with 5-aminosalicylic acid to form a tetrafluorobenzylidene-5-aminosalicylic acid imine intermediate. This Schiff base intermediate is then subjected to catalytic hydrogenation to yield the final amine product. This indirect route effectively masks the reactivity of the amine during the coupling phase, thereby preventing the over-alkylation that leads to dimer formation. The result is a cleaner reaction profile with significantly reduced downstream processing requirements, offering a compelling value proposition for cost reduction in pharmaceutical intermediates manufacturing.
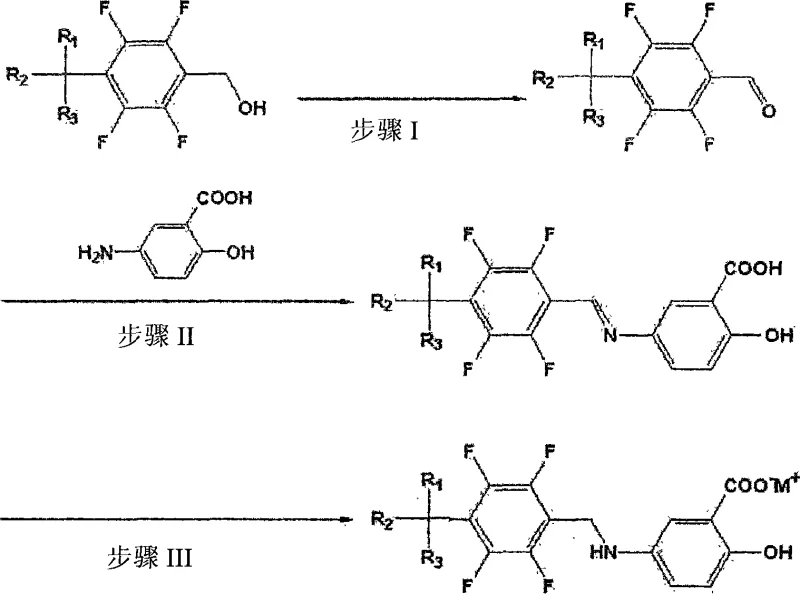
Mechanistic Insights into Reductive Amination via Imine Intermediate
The success of this synthetic strategy hinges on the precise control of the imine formation and subsequent reduction steps. In the first stage, tetrafluorobenzyl alcohol is oxidized to tetrafluorobenzaldehyde using pyridinium chlorochromate (PCC) in dichloromethane, achieving yields as high as 98%. The resulting aldehyde is highly electrophilic, facilitating a rapid condensation with the amino group of 5-aminosalicylic acid. The use of molecular sieves in this step is crucial, as they act as a dehydrating agent to drive the equilibrium towards the imine product (Formula II) by removing the water byproduct. This condensation proceeds efficiently at room temperature, yielding the imine intermediate in approximately 88% yield without the need for harsh conditions that might degrade the sensitive salicylic acid moiety.
Following the isolation of the imine, the final transformation involves a contact reduction reaction. Utilizing a platinum catalyst in an alcohol solvent, the carbon-nitrogen double bond of the imine is selectively hydrogenated to a single bond, generating the target tetrafluorobenzyl-5-aminosalicylic acid derivative. This step typically achieves yields around 85% with high purity (>99.8% by HPLC). Furthermore, the process allows for the direct formation of pharmaceutically acceptable salts, such as sodium, potassium, or lithium salts, by treating the final acid with the corresponding hydroxide. These salt forms exhibit vastly superior solubility and stability profiles compared to the free acid, with potassium salts showing solubility up to 590 mg/ml, making them ideal for injectable formulations.
How to Synthesize Tetrafluorobenzyl-5-Aminosalicylic Acid Efficiently
The operational simplicity of this three-step protocol makes it highly attractive for process chemistry teams aiming to scale up production. The synthesis begins with the oxidation of the alcohol precursor, followed by a room-temperature condensation that requires minimal energy input. The final hydrogenation step utilizes standard heterogeneous catalysis equipment, ensuring that the technology can be readily transferred from the laboratory to pilot and commercial plants. Detailed standardized operating procedures for each reaction stage, including specific reagent ratios, temperature controls, and workup protocols, are essential for maintaining the high purity standards demanded by regulatory bodies. The following guide outlines the critical operational parameters derived from the patent examples to ensure reproducible results.
- Oxidize tetrafluorobenzyl alcohol to tetrafluorobenzaldehyde using pyridinium chlorochromate in dichloromethane.
- Condense the resulting aldehyde with 5-aminosalicylic acid using molecular sieves to form the tetrafluorobenzylidene-5-aminosalicylic acid imine intermediate.
- Hydrogenate the imine intermediate using a platinum catalyst in an alcohol solvent to obtain the final tetrafluorobenzyl-5-aminosalicylic acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates into tangible operational efficiencies and risk mitigation. The elimination of the dimer impurity removes the need for expensive and time-consuming purification steps, such as complex chromatography or multiple recrystallizations, which traditionally bottleneck production throughput. This streamlining of the manufacturing process directly contributes to substantial cost savings by reducing solvent consumption, labor hours, and waste disposal costs associated with low-yield purification attempts. Moreover, the use of stable, commercially available starting materials like tetrafluorobenzyl alcohol ensures a resilient supply chain that is less susceptible to raw material volatility.
- Cost Reduction in Manufacturing: The avoidance of dimer formation significantly lowers the cost of goods sold (COGS) by improving the overall mass balance of the process. Since the impurity is prevented at the source rather than removed post-reaction, the yield loss associated with purification is minimized. This efficiency gain allows for more competitive pricing structures without compromising on the stringent quality specifications required for neurodegenerative disease therapeutics. Additionally, the ability to produce stable salt forms directly reduces formulation development costs for downstream drug manufacturers.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route enhances supply security by reducing the likelihood of batch failures due to impurity spikes. The process relies on well-established chemical transformations—oxidation, condensation, and hydrogenation—that are easily scalable and controllable. This predictability enables suppliers to commit to tighter delivery schedules and larger volume contracts, providing pharmaceutical partners with the confidence needed for long-term project planning. The improved stability of the salt forms also extends shelf-life, reducing inventory write-offs and logistics complexities.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the process is favorable as it avoids the use of highly reactive alkyl halides in the final coupling step, reducing potential hazards. The workup procedures involve standard filtration and crystallization techniques that generate manageable waste streams. The high purity achieved (>99.8%) minimizes the environmental burden of solvent-intensive purification processes. This alignment with green chemistry principles facilitates easier regulatory approval and supports corporate sustainability goals, making the technology future-proof for increasingly strict environmental regulations.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the chemical properties and manufacturing feasibility of these derivatives. The following responses are grounded in the experimental data and technical disclosures found within the patent literature, addressing key concerns about stability, toxicity, and process scalability. Understanding these nuances is vital for integrating this intermediate into your broader drug development pipeline.
Q: How does this new method prevent dimer impurity formation?
A: By utilizing a tetrafluorobenzylidene-5-aminosalicylic acid imine intermediate, the process avoids the direct nucleophilic substitution that causes secondary amine side reactions and dimerization in conventional methods.
Q: Which salt forms offer the best stability for injection formulations?
A: Experimental data indicates that potassium, sodium, and lithium salts exhibit superior solubility and stability compared to the free acid or organic salts, with potassium salts showing particularly enhanced stability in aqueous solutions.
Q: Is this synthesis route scalable for commercial production?
A: Yes, the method eliminates complex purification steps required for dimer removal and uses standard reagents like platinum catalysts and molecular sieves, making it highly suitable for industrial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrafluorobenzyl-5-Aminosalicylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving neurodegenerative therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of tetrafluorobenzyl-5-aminosalicylic acid derivatives meets the highest international standards. Our commitment to quality assurance means that you can rely on us for consistent supply and technical support throughout your product lifecycle.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this dimer-free process. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your project requirements. Let us collaborate to accelerate the delivery of effective treatments for Alzheimer's, Parkinson's, and other chronic neurological conditions.
