Advanced Manufacturing of Linagliptin Intermediates via Efficient Copper-Catalyzed Cyclization for Global Pharma Supply Chains
Advanced Manufacturing of Linagliptin Intermediates via Efficient Copper-Catalyzed Cyclization for Global Pharma Supply Chains
The escalating global demand for effective Type 2 diabetes treatments has placed immense pressure on pharmaceutical supply chains to deliver high-quality active pharmaceutical ingredients (APIs) and their precursors with greater efficiency and lower costs. Patent CN110590780B introduces a transformative preparation method for Linagliptin, a potent DPP-IV inhibitor, addressing critical bottlenecks in traditional synthesis routes. This technology leverages a novel copper-catalyzed oxidative cyclization strategy that streamlines the construction of the quinazoline core, a pivotal structural motif in the final drug molecule. By utilizing molecular oxygen as a green oxidant and optimizing leaving group chemistry, this process not only enhances reaction yields but also simplifies downstream purification, presenting a compelling value proposition for reliable pharmaceutical intermediate supplier partnerships aiming to optimize their manufacturing portfolios.
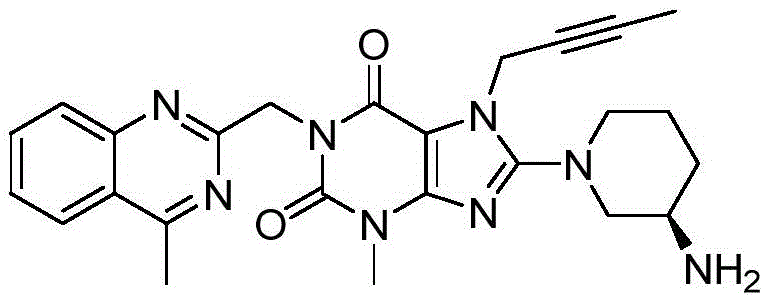
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key intermediates such as 2-chloromethyl-4-methylquinazoline has been plagued by inefficient multi-step sequences involving condensation, cyclization, and reduction under苛刻 conditions. Prior art methods, such as those cited in J Org Chem (2004), often suffer from lengthy reaction times, the requirement for expensive or hazardous reagents, and suboptimal total yields that drive up the cost of goods sold (COGS). Furthermore, three-component one-pot methods reported in literature frequently encounter issues with reaction complexity and selectivity, leading to difficult-to-remove impurities that compromise the purity profile required for regulatory compliance. These inefficiencies create significant vulnerabilities in the supply chain, resulting in extended lead times and reduced flexibility for procurement managers seeking cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN110590780B offers a streamlined two-component oxidative cyclization that directly constructs the quinazoline scaffold from o-aminoacetophenone and substituted acetamides. A standout innovation in this approach is the strategic incorporation of a tosylate (p-toluenesulfonyl) leaving group, which exhibits superior leaving ability compared to traditional halogens like chlorine or bromine. This modification facilitates smoother nucleophilic substitution in subsequent coupling steps, thereby boosting the overall yield of the coupling product. The reaction pathway is significantly shortened, operating under milder conditions with fewer by-products, which translates directly into substantial cost savings and a more robust production workflow for commercial scale-up of complex pharmaceutical intermediates.
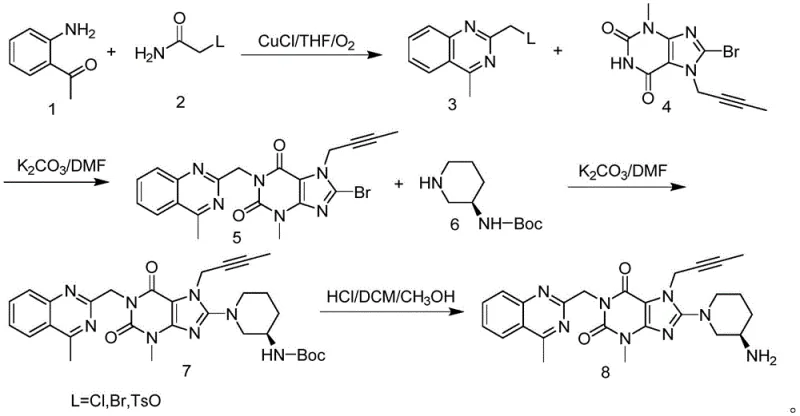
Mechanistic Insights into CuCl-Catalyzed Oxidative Cyclization
The core of this technological breakthrough lies in the copper-catalyzed oxidative cyclization mechanism, where cuprous chloride (CuCl) acts as the primary catalyst in the presence of molecular oxygen. In this system, o-aminoacetophenone reacts with the acetamide derivative in a tetrahydrofuran (THF) solvent medium, where oxygen serves as the terminal oxidant to drive the cyclization forward. The catalytic cycle likely involves the coordination of the copper species to the nitrogen and oxygen atoms of the substrates, facilitating the formation of the C-N bonds necessary to close the quinazoline ring. This aerobic oxidation process is inherently greener and more atom-economical than stoichiometric oxidants, reducing the generation of heavy metal waste and simplifying the environmental compliance burden for manufacturing facilities.
From an impurity control perspective, the choice of the leaving group plays a critical role in minimizing side reactions during the subsequent alkylation steps. The patent data indicates that replacing halogen atoms with the p-toluenesulfonate group significantly enhances the reactivity of the intermediate towards nucleophilic attack by the xanthine derivative. This increased reactivity allows the reaction to proceed to completion more rapidly and selectively, reducing the formation of unreacted starting materials and oligomeric by-products. Consequently, the final crude product exhibits a cleaner impurity profile, which eases the burden on purification units and ensures that the high-purity pharmaceutical intermediate specifications are met with greater consistency and reliability.
How to Synthesize Linagliptin Intermediates Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible roadmap for producing Linagliptin with high efficiency. The process begins with the copper-catalyzed cyclization to form the quinazoline intermediate, followed by sequential coupling reactions in DMF solvent using potassium carbonate as a base. The final step involves acidic deprotection and recrystallization to achieve the desired purity levels. This standardized approach minimizes operational variability and ensures batch-to-batch consistency, which is paramount for GMP manufacturing environments. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Perform copper-catalyzed oxidative cyclization of o-aminoacetophenone and substituted acetamide in THF with oxygen to form the quinazoline core.
- Execute nucleophilic substitution in DMF using potassium carbonate to couple the quinazoline intermediate with the xanthine derivative.
- Conduct final deprotection using hydrochloric acid in a DCM/methanol mixture followed by pH adjustment and recrystallization to obtain pure Linagliptin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible strategic advantages that extend beyond simple technical metrics. By shortening the synthetic sequence and eliminating the need for harsh reduction steps, the process drastically reduces the consumption of raw materials, solvents, and energy, leading to a leaner cost structure. The use of readily available starting materials like o-aminoacetophenone mitigates the risk of raw material shortages, ensuring a stable and continuous supply of critical intermediates. Furthermore, the simplified workup procedures, which often involve straightforward filtration and crystallization rather than complex chromatographic separations, accelerate production cycles and enhance overall throughput capacity.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts in later stages and the use of efficient leaving groups like tosylate significantly lower the cost of reagents and waste disposal. The shorter reaction route reduces labor hours and utility consumption per kilogram of product, driving down the overall manufacturing cost without compromising quality. This economic efficiency allows for more competitive pricing strategies in the global market for diabetes therapeutics.
- Enhanced Supply Chain Reliability: The robustness of the copper-catalyzed system and the availability of commodity chemicals as feedstocks ensure that production schedules are less susceptible to external supply shocks. The high yields reported in the patent examples suggest that less starting material is required to produce the same amount of product, effectively increasing the capacity of existing manufacturing assets and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process utilizes common industrial solvents such as THF and DMF, which are well-understood in large-scale operations, facilitating seamless technology transfer from pilot to commercial scale. Additionally, the use of oxygen as an oxidant and the reduction of heavy metal waste align with increasingly stringent environmental regulations, minimizing the ecological footprint and avoiding potential regulatory hurdles associated with toxic waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a transparent view of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing production lines.
Q: What is the primary advantage of using a tosylate leaving group in this synthesis?
A: The patent highlights that the sulfonyl group (tosylate) is a superior leaving group compared to halogens, facilitating easier substitution in subsequent coupling reactions and significantly improving overall yield.
Q: How does the copper-catalyzed method improve upon conventional Linagliptin synthesis?
A: Unlike traditional multi-step routes requiring harsh conditions, this method utilizes a direct one-pot oxidative cyclization with oxygen as the oxidant, reducing reaction steps, minimizing by-products, and lowering production costs.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process employs readily available starting materials like o-aminoacetophenone and uses standard solvents (THF, DMF), making it highly scalable and robust for industrial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Linagliptin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in meeting the growing global demand for diabetes medications. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Linagliptin intermediate adheres to the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this advanced copper-catalyzed technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us today to request specific COA data and comprehensive route feasibility assessments, and let us help you optimize your supply chain for the future of pharmaceutical manufacturing.
