Revolutionizing Aromatic Amide Production: A Metal-Free Radical Amidation Strategy for Commercial Scale
In the rapidly evolving landscape of fine chemical synthesis, the demand for efficient, environmentally benign, and cost-effective methodologies for constructing amide bonds has never been more critical. Amide functionalities serve as the structural backbone for a vast array of bioactive molecules, including over 25% of known pharmaceutical drugs, as well as key components in agrochemicals and advanced polymer materials. Addressing the longstanding challenges associated with traditional amidation, the groundbreaking technology disclosed in patent CN111039813B introduces a transformative approach to synthesizing aromatic amide compounds. This innovation leverages a metal-free radical catalysis system that utilizes readily available aromatic aldehydes and O-benzoyl hydroxylamines as primary substrates. By operating under atmospheric conditions with simple peroxide oxidants, this method circumvents the need for precious transition metal catalysts, offering a streamlined pathway that aligns perfectly with the rigorous purity and sustainability standards demanded by modern global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the amide bond has relied on several established but inherently flawed methodologies that pose significant hurdles for large-scale manufacturing. The classical approach involving the condensation of carboxylic acids and amines often necessitates harsh reaction conditions, including high temperatures and the use of activating agents that generate toxic by-products, creating substantial environmental disposal burdens. Alternatively, methods utilizing alcohols or nitriles frequently depend on stoichiometric amounts of expensive oxidants or noble metal catalysts, driving up raw material costs and complicating the removal of trace metal impurities—a critical failure point for pharmaceutical intermediates. Furthermore, strategies employing aldehydes and amines typically require excessive amounts of additives and transition metals to proceed efficiently, resulting in poor atom economy and complex workup procedures. Even oxime rearrangement techniques, while useful in specific contexts, generally demand strong acidic environments and heavy metal catalysts, imposing severe constraints on equipment corrosion resistance and operational safety.
The Novel Approach
In stark contrast to these legacy technologies, the novel synthetic route detailed in the patent offers a direct, one-step amidation protocol that fundamentally reshapes the economic and operational landscape of amide production. This method ingeniously couples aromatic aldehydes with O-benzoyl hydroxylamines in the presence of a peroxide catalyst, facilitating the formation of the amide bond without any metal participation. The reaction proceeds smoothly in common organic solvents such as dichloroethane or chlorobenzene at moderate temperatures ranging from 50-80°C. This shift not only drastically reduces the energy consumption associated with high-temperature reflux but also eliminates the costly and time-consuming steps required for metal scavenging. The simplicity of the protocol, which requires only filtration and standard purification post-reaction, represents a paradigm shift towards greener chemistry, enabling manufacturers to achieve high-purity aromatic amides with significantly reduced operational complexity and waste generation.
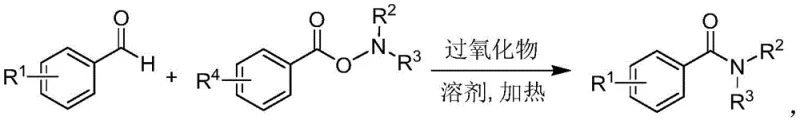
Mechanistic Insights into Peroxide-Catalyzed Radical Amidation
The efficacy of this metal-free transformation is rooted in a sophisticated radical chain mechanism that ensures high conversion rates and excellent selectivity. Under the influence of the peroxide initiator, the aromatic aldehyde substrate undergoes hydrogen abstraction at the formyl group, generating a highly reactive acyl radical species (ArCO·). This transient radical intermediate then selectively attacks the nitrogen center of the O-benzoyl hydroxylamine, effectively cleaving the weak N-O bond to forge the new C-N amide linkage. Concurrently, this step releases a benzoyloxy radical, which rapidly undergoes decarboxylation to form a phenyl radical. This phenyl radical subsequently propagates the chain reaction by interacting with other species in the mixture, thereby sustaining the catalytic cycle without the need for external metal coordination. This elegant mechanistic pathway not only explains the high efficiency observed but also highlights the robustness of the system against various electronic effects on the aromatic rings.
From an impurity control perspective, this radical mechanism offers distinct advantages over ionic pathways often plagued by side reactions such as over-oxidation or polymerization. The specific reactivity of the acyl radical towards the O-benzoyl hydroxylamine ensures that the primary reaction channel dominates, minimizing the formation of undesired by-products like carboxylic acids or esters. Furthermore, because the reaction does not involve transition metals, there is no risk of metal-induced degradation of sensitive functional groups often present in complex drug intermediates. The absence of metal catalysts also means that the final product profile is cleaner, with the primary impurities being easily removable organic residues rather than persistent metal complexes. This inherent purity profile is crucial for downstream applications in the pharmaceutical industry, where strict limits on elemental impurities are enforced by regulatory bodies globally.
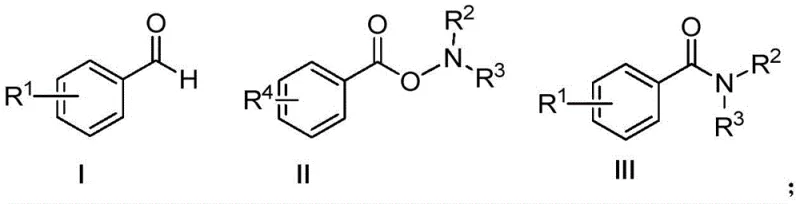
How to Synthesize Aromatic Amides Efficiently
Implementing this synthesis strategy in a laboratory or pilot plant setting requires adherence to specific procedural parameters to maximize yield and safety. The process begins with the precise weighing of the aromatic aldehyde and the O-benzoyl hydroxylamine derivative, ensuring a molar ratio between 1:1 and 1:2 to drive the equilibrium towards product formation. These substrates are dissolved in a suitable organic solvent, with dichloroethane being a preferred choice due to its solubility profile and boiling point characteristics. The addition of the peroxide oxidant, such as dibenzoyl peroxide or tert-butyl hydroperoxide, is critical and must be controlled to maintain a molar ratio of (1-5):100 relative to the aldehyde. Once the mixture is assembled, heating the reaction vessel to a temperature window of 50-80°C for a duration of 6-10 hours allows the radical cascade to proceed to completion. Detailed standardized synthetic steps see the guide below.
- Prepare the reaction mixture by combining aromatic aldehyde and O-benzoyl hydroxylamine compounds in a molar ratio of 1: (1-2) within an organic solvent such as dichloroethane.
- Add a catalytic amount of peroxide oxidant, such as dibenzoyl peroxide or ammonium persulfate, maintaining a molar ratio of (1-5): 100 relative to the aldehyde.
- Heat the reaction mixture to 50-80°C under atmospheric conditions for 6-10 hours, followed by filtration, washing, and purification to isolate the target amide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free amidation technology translates into tangible strategic benefits that extend far beyond simple chemical yield. By eliminating the dependency on scarce and volatile transition metals like palladium or platinum, manufacturers can insulate their production costs from the erratic fluctuations of the precious metals market. The simplified workflow, which removes the need for specialized metal scavenging resins and extensive filtration protocols, results in a drastically shortened production cycle time. This efficiency gain allows for faster turnover of reactor vessels, effectively increasing the overall capacity of existing manufacturing infrastructure without the need for capital-intensive expansion. Moreover, the use of inexpensive, commodity-grade oxidants and solvents ensures a stable and predictable cost structure, facilitating more accurate long-term budgeting and pricing strategies for high-volume contracts.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the complete removal of expensive transition metal catalysts from the bill of materials. In traditional amidation processes, the cost of the catalyst and the subsequent purification media can constitute a significant portion of the total production expense. By replacing these with low-cost peroxides, the direct material cost is substantially lowered. Additionally, the mild reaction conditions (50-80°C) reduce energy consumption compared to high-temperature reflux methods, further contributing to operational expenditure savings. The simplified workup procedure also reduces labor hours and solvent usage during purification, compounding the economic benefits across the entire manufacturing value chain.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of widely available, non-specialized raw materials. Unlike proprietary ligands or rare earth catalysts that may face geopolitical supply constraints or long lead times, the aldehydes, hydroxylamines, and peroxides required for this process are commodity chemicals with robust global supply networks. This accessibility minimizes the risk of production stoppages due to raw material shortages. Furthermore, the atmospheric nature of the reaction removes the need for complex inert gas handling systems, simplifying the engineering requirements for production facilities and allowing for greater flexibility in manufacturing location and scale.
- Scalability and Environmental Compliance: Scaling this process from gram-scale R&D to multi-ton commercial production is inherently safer and more straightforward due to the absence of pyrophoric metal catalysts and hazardous activating agents. The reduced generation of toxic heavy metal waste simplifies environmental compliance and lowers the costs associated with waste treatment and disposal. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile, a factor increasingly weighted in vendor selection criteria by major multinational pharmaceutical and agrochemical corporations seeking to reduce their Scope 3 emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel amidation technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these nuances is essential for R&D teams evaluating route feasibility and procurement officers assessing supplier qualifications.
Q: Why is this metal-free amidation method superior to traditional transition metal catalysis?
A: Traditional methods often require expensive transition metal catalysts like palladium or ruthenium, which necessitate complex removal steps to meet pharmaceutical purity standards. This patented method utilizes inexpensive peroxides in an atmospheric environment, eliminating heavy metal contamination risks and significantly simplifying the downstream purification process.
Q: What are the typical reaction conditions and yields for this synthesis?
A: The reaction operates under mild thermal conditions between 50-80°C and completes within 6-10 hours. Experimental data from the patent indicates that yields consistently exceed 87%, with specific examples demonstrating efficiencies up to 92% for substrates like 4-methylbenzaldehyde.
Q: Can this method be applied to substrates with diverse functional groups?
A: Yes, the method exhibits excellent substrate universality. It tolerates various substituents on the aromatic ring, including halogens, nitro groups, cyano groups, and alkyl chains, making it highly versatile for synthesizing complex intermediates for drugs and agrochemicals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Amide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of this metal-free amidation method are faithfully reproduced at an industrial level. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of aromatic amide intermediate meets the exacting standards required for API synthesis and advanced material applications. We are committed to delivering consistent quality and reliability, serving as a stable anchor in your supply chain.
We invite you to leverage our technical proficiency to optimize your specific production needs. Whether you are looking to retrofit an existing process or develop a new synthetic route from scratch, our team is ready to provide a Customized Cost-Saving Analysis tailored to your project goals. We encourage potential partners to contact our technical procurement team to request specific COA data and comprehensive route feasibility assessments. By collaborating with us, you gain access to a seamless integration of cutting-edge chemistry and world-class manufacturing, positioning your organization for success in a competitive global market.
