Advanced Manufacturing of High-Purity Tadalafil via Optimized Acyl Chlorination and Cyclization
The pharmaceutical industry continuously demands more efficient and purer synthesis routes for critical active pharmaceutical ingredients (APIs), particularly for widely prescribed medications like Tadalafil. Patent CN113880837A introduces a groundbreaking preparation method that addresses longstanding challenges in purity and cost-efficiency. This innovative approach utilizes D-cis-carboline hydrochloride as the initial raw material, subjecting it to a precise acyl chlorination reaction followed by a rigorous refining process. The resulting chloroacetyl acylate intermediate is then undergoes an aminolysis cyclization reaction with methylamine to yield the final product. Unlike conventional methods that often struggle to breach the 99.5% purity threshold without complex chromatography, this patented technique consistently achieves a purity of over 99.9%. The strategic integration of activated carbon decolorization and controlled crystallization steps ensures that the impurity profile is meticulously managed, making this route highly attractive for manufacturers seeking to optimize their supply chain for high-quality erectile dysfunction treatments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for Tadalafil, such as those disclosed in earlier patents like CN110437228A, typically initiate from D-tryptophan. While effective, these legacy routes involve multiple esterification and Pictet-Spengler reaction steps that can introduce significant variability in stereochemical integrity and overall yield. A critical bottleneck in these conventional processes is the difficulty in removing trace impurities generated during the amidation and cyclization stages, often capping the final purity at approximately 99.3%. Furthermore, the reliance on sulfuric acid catalysis in the early stages necessitates rigorous neutralization and waste treatment protocols, adding to the environmental burden and operational complexity. The cumulative effect of these inefficiencies is a longer production cycle and higher manufacturing costs, which ultimately impact the competitiveness of the final API in a price-sensitive global market. Manufacturers relying on these older methodologies often face challenges in meeting the increasingly stringent regulatory requirements for impurity limits without resorting to expensive preparative HPLC purification.
The Novel Approach
In stark contrast, the novel approach detailed in patent CN113880837A streamlines the synthesis by starting directly with D-cis-carboline hydrochloride, a stable and commercially available intermediate. This strategic shift eliminates the need for the initial esterification and P-S reaction steps, significantly shortening the production timeline. The core innovation lies in the two-stage purification strategy: a first refining step for the chloroacetyl intermediate and a secondary refining step for the crude Tadalafil. By introducing activated carbon decolorization specifically during the intermediate stage, the process proactively removes colored impurities and potential side-reaction products before they can propagate through the synthesis. This proactive impurity management allows the final aminolysis cyclization to proceed with higher fidelity, resulting in a crude product that requires only simple pulping and washing to achieve pharmaceutical-grade purity. This methodology not only enhances the quality of the output but also simplifies the downstream processing, offering a robust solution for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Acyl Chlorination and Aminolysis Cyclization
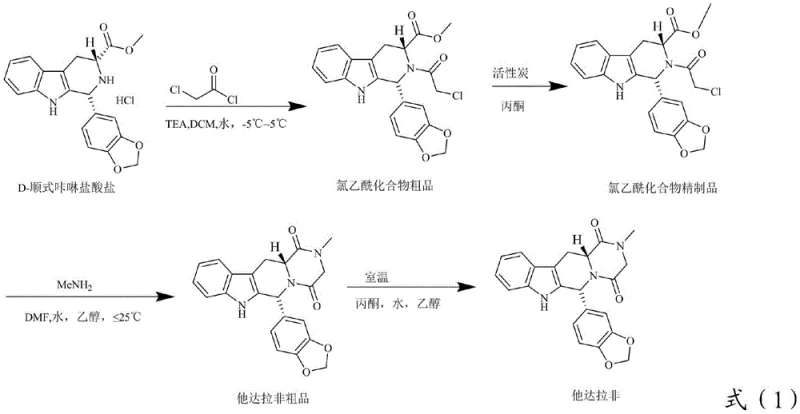
The success of this synthesis route hinges on the precise control of the acyl chlorination reaction, where D-cis-carboline hydrochloride reacts with chloroacetyl chloride. The mechanism involves the nucleophilic attack of the secondary amine nitrogen on the carbonyl carbon of the acid chloride, facilitated by an acid-binding agent such as triethylamine. Maintaining the reaction temperature between 0°C and 25°C is critical; temperatures that are too high can lead to over-acylation or degradation of the sensitive indole ring, while temperatures that are too low may result in incomplete conversion. The use of dichloromethane or tetrahydrofuran as the solvent provides an optimal medium for solubilizing the reactants while allowing for efficient heat dissipation during the exothermic acylation. The stoichiometry is carefully balanced, with a molar ratio of chloroacetyl chloride to the starting material ranging from 1.5:1 to 3.0:1, ensuring that the reaction drives to completion without excessive accumulation of unreacted acid chloride, which could complicate subsequent purification steps.
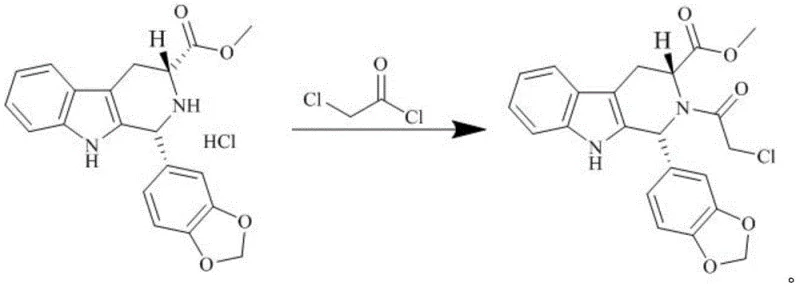
Following the formation of the chloroacetyl intermediate, the process transitions to the aminolysis cyclization reaction, which constructs the final piperazine-2,5-dione ring characteristic of Tadalafil. In this step, the chloroacetyl group acts as an electrophile, undergoing nucleophilic substitution with methylamine. The reaction is conducted in polar aprotic solvents like DMF or methanol, which stabilize the transition state and facilitate the displacement of the chloride ion. The temperature control remains paramount, kept below 25°C to prevent the formation of polymeric by-products or racemization of the chiral centers. The mechanism proceeds through a tetrahedral intermediate, which collapses to release the chloride and form the amide bond, followed by an intramolecular cyclization. The high selectivity of this reaction, combined with the high purity of the incoming intermediate, ensures that the final cyclization yields the desired cis-isomer with minimal trans-isomer contamination, thereby securing the biological activity and safety profile of the final API.
How to Synthesize Tadalafil Efficiently
The synthesis of Tadalafil via this patented route offers a streamlined pathway that balances reaction kinetics with purification efficiency. The process begins with the acylation of the carboline scaffold, followed by a critical purification of the intermediate to ensure high fidelity in the subsequent cyclization. The final steps involve a mild aminolysis and a straightforward workup that avoids harsh conditions. For process chemists looking to implement this route, attention to the crystallization parameters—specifically the addition rate of water and the cooling profile—is essential for maximizing yield and crystal quality. The detailed standardized synthesis steps, including specific solvent ratios and temperature ramps, are outlined in the guide below to ensure reproducibility and compliance with GMP standards.
- Perform acyl chlorination of D-cis-carboline hydrochloride with chloroacetyl chloride using an acid-binding agent in an organic solvent at 0-25°C.
- Refine the crude chloroacetyl intermediate via activated carbon decolorization and crystallization to remove impurities.
- Conduct aminolysis cyclization by reacting the refined intermediate with methylamine in a second organic solvent.
- Purify the crude tadalafil product through pulping, washing, and drying to obtain the final high-purity API.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route presents significant strategic advantages, primarily driven by the optimization of raw material usage and process simplicity. The shift to D-cis-carboline hydrochloride as a starting material leverages a supply chain that is already well-established for other carboline derivatives, reducing the risk of raw material shortages. Furthermore, the elimination of complex chromatographic purification steps in favor of crystallization and pulping drastically reduces the consumption of expensive silica gel and organic solvents associated with column chromatography. This simplification translates directly into lower operational expenditures and a reduced environmental footprint, aligning with modern green chemistry initiatives. The robustness of the process, demonstrated by its successful scale-up from 500mL flasks to 300L reactors, assures supply chain heads of its viability for commercial scale-up of complex pharmaceutical intermediates without the typical teething problems associated with new technology transfer.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the inherent efficiency of the reaction sequence and the avoidance of costly purification technologies. By utilizing cheap and easy-to-obtain raw materials and employing standard unit operations like filtration and drying, the overall production cost is substantially lowered. The high yield of the aminolysis step, coupled with the high recovery rate during the refining stages, minimizes material loss, ensuring that the cost per kilogram of the final API is competitive. Additionally, the reduced reaction time contributes to higher throughput in existing manufacturing facilities, allowing for better asset utilization and faster return on investment.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available starting materials mitigates the risk of supply disruptions that can plague synthetic routes dependent on custom-synthesized precursors. The process conditions are mild and do not require specialized high-pressure or cryogenic equipment, meaning that production can be easily distributed across multiple manufacturing sites with standard chemical infrastructure. This flexibility enhances supply continuity, ensuring that downstream drug product manufacturers receive consistent deliveries. The simplified workflow also reduces the lead time for high-purity pharmaceutical intermediates, enabling faster response to market demand fluctuations.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method excels by minimizing the generation of hazardous waste. The use of activated carbon for decolorization is a scalable and environmentally benign alternative to more aggressive chemical treatments. The solvent systems employed, such as acetone, ethanol, and water, are relatively easy to recover and recycle, further reducing the environmental impact. The process has been proven to scale effectively, maintaining high purity and yield even at the 100L to 300L scale, which de-risks the transition from pilot plant to full commercial production. This scalability ensures that the supply chain can grow in tandem with market needs without compromising on quality or regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Tadalafil preparation method. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on process capabilities and quality outcomes. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios.
Q: What is the primary advantage of using D-cis-carboline hydrochloride as the starting material?
A: Using D-cis-carboline hydrochloride allows for a shorter production period and lower costs compared to traditional routes starting from D-tryptophan, while enabling the achievement of purity levels exceeding 99.9%.
Q: How does the new purification method improve impurity control?
A: The process incorporates a specific refining step for the chloroacetyl intermediate involving activated carbon decolorization and controlled crystallization, which effectively removes colored impurities and by-products before the final cyclization step.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the patent demonstrates successful scale-up from laboratory (500mL) to pilot/industrial scales (100L-300L reactors) using standard equipment like glass or enamel reaction kettles, indicating high scalability and robustness.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tadalafil Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis routes like the one described in CN113880837A to maintain a competitive edge in the global pharmaceutical market. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity Tadalafil that meets stringent purity specifications, leveraging our rigorous QC labs to verify every batch against the highest international standards. Our facility is equipped to handle the specific solvent systems and temperature controls required for this acyl chlorination and cyclization process, guaranteeing consistent quality and supply reliability for our partners.
We invite pharmaceutical companies and generic drug manufacturers to collaborate with us to optimize their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to see how our implementation of this patented method can enhance your product portfolio. Let us help you secure a stable, high-quality supply of Tadalafil that supports your long-term business goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
