Advanced Synthesis of Vitamin A Key Intermediate: Streamlining C5 Aldehyde Production for Global Markets
The global demand for Vitamin A and its derivatives, particularly esters like Vitamin A acetate, continues to drive innovation in the fine chemical sector, necessitating more efficient routes for key intermediates. Patent CN102311339A introduces a transformative preparation method for 4-acetoxy-2-methyl-2-butene-1-aldehyde, widely known in the industry as the C5 aldehyde. This compound serves as the critical C5 building block in the Wittig reaction synthesis of Vitamin A esters, coupling with the C15 phosphonium salt to form the final vitamin structure. The disclosed technology addresses long-standing inefficiencies in traditional manufacturing by proposing a concise, two-step sequence that begins with the hydrolysis of a specific acetal precursor followed by a catalytic rearrangement. This approach not only simplifies the operational workflow but also enhances the purity profile of the final product, making it a highly attractive option for large-scale industrial application.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of this vital C5 aldehyde has relied on complex multi-step sequences, most notably the route developed by BASF AG in the mid-1970s. As illustrated in the reaction scheme below, the conventional process initiates with pyruvic aldehyde dimethyl acetal, which undergoes condensation with acetylene to form a tertiary alcohol. This intermediate must then be subjected to a reduction step to convert the triple bond into a double bond, followed by esterification and a subsequent rearrangement before finally undergoing acetal hydrolysis to yield the target aldehyde. 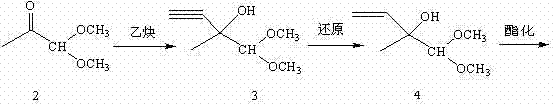
This traditional pathway presents significant logistical and economic challenges for modern procurement and supply chain teams. The requirement for acetylene gas introduces severe safety hazards and necessitates specialized handling infrastructure, thereby increasing capital expenditure and operational risk. Furthermore, the sheer length of the synthetic route, involving at least five distinct chemical transformations, inherently compounds yield losses at each stage. The accumulation of impurities across such a long sequence complicates downstream purification, often requiring extensive chromatography or distillation efforts that drive up production costs and extend lead times for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to the cumbersome legacy methods, the novel approach detailed in the patent leverages a strategically designed shortcut that bypasses the need for alkyne chemistry entirely. The new methodology utilizes 2-methyl-2-acetoxy-1,1-dimethoxy-3-butene (referred to as Acetal 5) as the starting material, which is commercially accessible or easily synthesized via alternative stable routes. The process flows directly through a hydrolysis step to generate an intermediate aldehyde, which then undergoes a seamless allylic rearrangement to afford the final 4-acetoxy-2-methyl-2-butene-1-aldehyde. 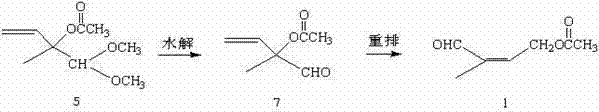
This streamlined architecture offers profound advantages for cost reduction in pharmaceutical intermediates manufacturing. By collapsing the synthesis into just two main reaction vessels, the process drastically reduces solvent consumption, energy usage for heating and cooling cycles, and labor hours required for batch processing. The elimination of the acetylene condensation and reduction steps removes the most hazardous and equipment-intensive portions of the legacy workflow. Consequently, this allows manufacturers to achieve a more robust and reliable supply chain for Vitamin A precursors, with significantly reduced exposure to raw material volatility and processing bottlenecks.
Mechanistic Insights into Acid-Catalyzed Hydrolysis and Allylic Rearrangement
The core of this technological breakthrough lies in the precise control of two distinct chemical transformations: the acidic hydrolysis of the dimethyl acetal moiety and the subsequent metal-catalyzed [3,3]-sigmatropic rearrangement. In the first stage, the acetal protecting group is cleaved under mild acidic conditions, typically utilizing dilute sulfuric acid or hydrochloric acid with a concentration range of 0.5N to 2.5N. The reaction is conducted at elevated temperatures between 50°C and 100°C, optimally around 70°C to 80°C, to ensure complete conversion to the intermediate 2-methyl-2-acetoxy-3-butene-1-aldehyde without degrading the sensitive allylic acetate functionality. This step is critical because it generates the free aldehyde necessary to trigger the subsequent rearrangement, and the use of aqueous acid media simplifies the workup procedure compared to anhydrous Lewis acid methods.
Following hydrolysis, the intermediate aldehyde undergoes a fascinating allylic rearrangement driven by transition metal catalysis. The patent highlights the efficacy of bis(acetonitrile)palladium(II) chloride or cuprous chloride as catalysts, employed at loading levels of 0.5% to 2% by mass relative to the substrate. In solvents such as acetonitrile or dimethylformamide (DMF), the metal center coordinates with the alkene and the carbonyl oxygen, facilitating the migration of the acetoxy group from the C2 position to the C4 position while shifting the double bond. This mechanistic pathway is highly selective, minimizing the formation of regioisomers or polymerization byproducts that often plague thermal rearrangements. The ability to tune the reaction temperature between 55°C and 75°C allows operators to balance reaction rate against selectivity, ensuring a high-purity output suitable for direct use in downstream Vitamin A synthesis.
How to Synthesize 4-Acetoxy-2-Methyl-2-Butene-1-Aldehyde Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and minimize impurity formation. The process begins with the controlled addition of the acetal starting material into the heated acidic aqueous phase, followed by extraction and drying to isolate the intermediate aldehyde. This intermediate is then immediately subjected to the rearrangement conditions in an anhydrous organic solvent containing the palladium or copper catalyst.
- Hydrolyze 2-methyl-2-acetoxy-1,1-dimethoxy-3-butene using dilute sulfuric or hydrochloric acid at 50-100°C to obtain the intermediate aldehyde.
- Perform an allylic rearrangement on the intermediate aldehyde using a palladium or copper catalyst in acetonitrile or DMF solvent.
- Isolate the final 4-acetoxy-2-methyl-2-butene-1-aldehyde product via vacuum distillation after reaction completion monitoring by GC.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology represents a strategic opportunity to optimize the cost structure and reliability of Vitamin A intermediate sourcing. The fundamental shift away from hazardous gaseous reagents and multi-step sequences translates directly into lower operational expenditures and reduced regulatory burden. By simplifying the manufacturing footprint, suppliers can offer more competitive pricing structures while maintaining rigorous quality standards required by global pharmacopeias. The following points detail the specific commercial benefits derived from this technical innovation.
- Cost Reduction in Manufacturing: The elimination of the acetylene condensation and hydrogenation steps removes the need for expensive high-pressure reactors and specialized gas handling infrastructure. This reduction in capital intensity allows for significant savings in fixed asset depreciation and maintenance costs. Furthermore, the shorter synthetic route inherently improves the overall mass balance, meaning less raw material is wasted in side reactions or lost during multiple isolation steps. The use of common, commodity-grade solvents like acetonitrile and DMF, rather than exotic reagents, further stabilizes the variable cost profile against market fluctuations.
- Enhanced Supply Chain Reliability: Relying on a starting material that is a stable liquid acetal rather than a compressed gas significantly mitigates supply risks associated with transportation and storage. The simplified two-step process reduces the probability of batch failures caused by cumulative errors in long synthesis chains, leading to more consistent delivery schedules. This reliability is crucial for downstream Vitamin A manufacturers who operate on tight production timelines and cannot afford interruptions in their C5 aldehyde supply. The robustness of the catalytic system also ensures that production can be scaled up or down rapidly in response to market demand without compromising product quality.
- Scalability and Environmental Compliance: The mild reaction conditions, operating well below 100°C, reduce the energy load required for heating and cooling, contributing to a lower carbon footprint for the manufacturing facility. The reduction in the number of chemical steps directly correlates to a decrease in the volume of wastewater and organic waste generated per kilogram of product. This aligns with increasingly stringent environmental regulations globally, reducing the risk of production shutdowns due to compliance issues. The process is inherently safer, minimizing the potential for runaway reactions or hazardous exposures, which simplifies insurance and safety auditing requirements for large-scale commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims presented in the patent documentation to provide clarity for potential partners.
Q: What are the primary advantages of this new synthesis route over the traditional BASF method?
A: The new route significantly shortens the synthetic pathway by eliminating the need for acetylene condensation and reduction steps found in the traditional method. It utilizes milder reaction conditions and readily available starting materials, resulting in higher overall yields and reduced operational complexity.
Q: Which catalysts are preferred for the rearrangement step in this process?
A: The patent specifies the use of bis(acetonitrile)palladium(II) chloride or cuprous chloride as effective catalysts. These catalysts facilitate the allylic rearrangement efficiently at moderate temperatures between 50°C and 100°C, ensuring high conversion rates.
Q: How does this method impact environmental compliance and waste generation?
A: By reducing the number of reaction steps and avoiding hazardous reagents like acetylene gas, the process inherently generates less three-waste discharge. The use of standard solvents like acetonitrile and DMF allows for established recycling protocols, enhancing the environmental profile of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Acetoxy-2-Methyl-2-Butene-1-Aldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that high-quality intermediates play in the efficient production of essential vitamins and nutraceuticals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless. We are committed to delivering 4-acetoxy-2-methyl-2-butene-1-aldehyde with stringent purity specifications, supported by our rigorous QC labs that utilize advanced GC-MS and NMR instrumentation to verify every batch. Our facility is equipped to handle the specific catalytic requirements of this novel route, guaranteeing a consistent supply of this critical building block.
We invite global partners to collaborate with us to leverage these technological advancements for their Vitamin A supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your production goals are met with the highest standards of quality and efficiency.
