Advanced Synthesis of Vitamin A Intermediate 4-Acetoxy-2-methyl-2-butene-1-aldehyde via Efficient Rearrangement
The global demand for Vitamin A and its derivatives, particularly carboxylate esters like retinyl acetate and palmitate, continues to drive innovation in fine chemical manufacturing. As a critical node in the supply chain for pharmaceuticals, food additives, and feed supplements, the efficient production of key intermediates is paramount. Patent CN102311339B introduces a transformative methodology for synthesizing 4-acetoxy-2-methyl-2-butene-1-aldehyde, often referred to in the industry as the "C5 aldehyde." This compound serves as the essential five-carbon building block in the Wittig reaction-based C15+C5 route for Vitamin A ester synthesis. The disclosed technology addresses long-standing bottlenecks in yield and operational complexity, offering a robust alternative to legacy industrial processes.
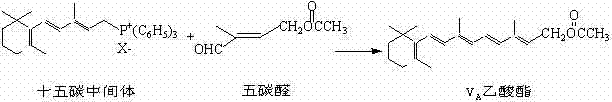
Traditionally, the synthesis of this vital intermediate has relied on pathways established decades ago, which, while effective, suffer from inherent inefficiencies. The conventional industrial route, famously reported by BASF in the mid-1970s, involves a multi-step sequence starting from acetonaldehyde dimethyl acetal. This legacy process requires condensation with acetylene to form a tertiary alcohol, followed by a reduction of the triple bond to a double bond, subsequent esterification, rearrangement to a dimethyl acetal, and finally, a hydrolysis step to reveal the target aldehyde. While this method laid the foundation for the industry, its length and the handling of hazardous gases like acetylene present significant logistical and safety challenges for modern manufacturing facilities seeking optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The historical reliance on the acetylene-based route imposes substantial burdens on both capital expenditure and operational safety. The necessity to handle acetylene gas requires specialized infrastructure and rigorous safety protocols, increasing the barrier to entry for production. Furthermore, the multi-step nature of the traditional synthesis—spanning condensation, reduction, esterification, and multiple protection/deprotection sequences—inevitably leads to cumulative yield losses. Each additional unit operation introduces potential points of failure, increases solvent consumption, and generates a larger volume of waste streams. For procurement managers and supply chain heads, these factors translate into higher variable costs and increased vulnerability to supply disruptions caused by complex processing requirements.
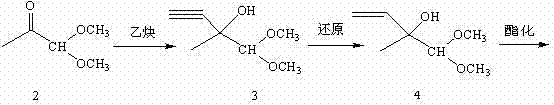
The Novel Approach
In stark contrast, the methodology outlined in CN102311339B streamlines the production workflow by leveraging a direct hydrolysis and rearrangement strategy. Instead of building the carbon skeleton from small gaseous molecules, this innovative approach utilizes 2-methyl-2-acetoxy-1,1-dimethoxy-3-butene (Acetal 5) as the starting material. The process bypasses the need for acetylene condensation and triple-bond reduction entirely. By focusing on the transformation of a pre-functionalized acetal directly into the target aldehyde through a controlled hydrolysis followed by a catalytic rearrangement, the route achieves a dramatic reduction in step count. This simplification not only accelerates the production cycle but also enhances the overall atom economy of the process.
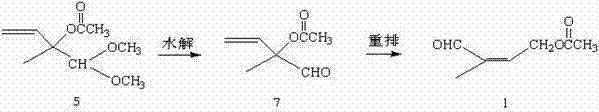
Mechanistic Insights into Acid-Catalyzed Hydrolysis and Pd-Mediated Rearrangement
The core of this technological advancement lies in the precise control of two distinct chemical transformations. The first stage involves the acid-catalyzed hydrolysis of the dimethyl acetal moiety. Under optimized conditions using dilute mineral acids such as hydrochloric acid or sulfuric acid at concentrations between 0.5N and 2.5N, the acetal protecting group is selectively removed. This reaction proceeds efficiently at temperatures ranging from 50°C to 100°C, with optimal results observed between 70°C and 80°C. The mechanism ensures the clean generation of the intermediate 2-methyl-2-acetoxy-3-butene-1-aldehyde (Aldehyde 7) without compromising the sensitive allylic acetate functionality, a critical factor in maintaining high purity levels.
Following hydrolysis, the second stage employs a transition metal-catalyzed allylic rearrangement to shift the double bond and establish the conjugated system required for the final Wittig coupling. The patent specifies the use of catalysts such as bisacetonitrile palladium chloride or cuprous chloride, typically loaded at 0.5% to 2% by mass relative to the substrate. Conducted in polar aprotic solvents like acetonitrile or DMF at moderate temperatures (55°C to 75°C), this rearrangement is highly selective. The catalytic cycle facilitates the migration of the acetate group and the double bond, converting the non-conjugated intermediate directly into the conjugated 4-acetoxy-2-methyl-2-butene-1-aldehyde. This mechanistic elegance minimizes side reactions and impurity formation, ensuring a product profile that meets stringent pharmaceutical specifications.
How to Synthesize 4-Acetoxy-2-methyl-2-butene-1-aldehyde Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and purity. The process begins with the preparation of the intermediate aldehyde via hydrolysis, followed immediately by the rearrangement step. Operators should monitor reaction progress using gas chromatography to determine the optimal endpoint for each stage, preventing over-reaction or degradation. The workup procedure involves standard extraction and vacuum distillation techniques, which are well-suited for existing chemical infrastructure. For detailed operational parameters, stoichiometry, and specific isolation techniques, please refer to the standardized guide below.
- Hydrolyze 2-methyl-2-acetoxy-1,1-dimethoxy-3-butene using dilute hydrochloric or sulfuric acid (0.5-2.5N) at 50-100°C for 1-5 hours to obtain 2-methyl-2-acetoxy-3-butene-1-aldehyde.
- Perform a rearrangement reaction on the resulting aldehyde using bisacetonitrile palladium chloride or cuprous chloride (0.5-2% catalyst loading) in acetonitrile or DMF solvent at 50-100°C.
- Isolate the final product, 4-acetoxy-2-methyl-2-butene-1-aldehyde, via vacuum distillation, collecting fractions at approximately 70-75°C under reduced pressure (1.5mmHg).
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the shift to this novel synthetic route offers compelling economic and logistical benefits. By eliminating the need for hazardous acetylene gas and reducing the total number of synthetic steps, the process inherently lowers the operational risk profile and reduces the consumption of utilities and solvents. The simplified workflow allows for faster batch turnover times, enabling manufacturers to respond more agilely to market fluctuations in Vitamin A demand. Furthermore, the use of readily available starting materials and common catalysts mitigates the risk of raw material shortages that often plague more exotic synthetic pathways.
- Cost Reduction in Manufacturing: The streamlined nature of this process directly correlates to significant cost savings. By removing the acetylene condensation and reduction steps, manufacturers eliminate the associated equipment costs, safety monitoring systems, and energy inputs required for high-pressure hydrogenation. Additionally, the high yields reported in the patent examples (exceeding 90% for the hydrolysis step and over 80% for the rearrangement) imply a substantial reduction in raw material waste per kilogram of final product. The lower catalyst loading further contributes to a reduced bill of materials, making the cost of goods sold (COGS) highly competitive compared to legacy methods.
- Enhanced Supply Chain Reliability: Dependence on complex, multi-step routes often introduces fragility into the supply chain. This new method consolidates the synthesis into fewer, more robust unit operations. The starting material, 2-methyl-2-acetoxy-1,1-dimethoxy-3-butene, is stable and easier to transport and store than gaseous acetylene. This stability ensures a consistent feedstock supply, reducing the likelihood of production stoppages. Moreover, the mild reaction conditions reduce wear and tear on reactor vessels, leading to lower maintenance downtime and more predictable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial emissions intensify, the environmental footprint of chemical manufacturing becomes a critical metric. This process generates significantly less waste due to higher atom efficiency and fewer purification steps. The absence of heavy metal reduction catalysts (like those used in alkyne hydrogenation) and the use of recoverable solvents simplify wastewater treatment protocols. The scalability is further enhanced by the fact that the reaction exotherms are manageable and do not require extreme cryogenic or high-pressure conditions, allowing for safe scale-up from pilot plants to multi-ton commercial production facilities.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for successful technology transfer and quality assurance. The following questions address common inquiries regarding the practical application of this patent, focusing on yield optimization, catalyst selection, and product purity. These insights are derived directly from the experimental data and technical disclosures within the patent documentation to provide clarity for R&D and technical procurement teams.
Q: What are the primary advantages of this new synthesis route over the traditional BASF method?
A: The novel route significantly shortens the synthetic sequence by eliminating the need for acetylene condensation and triple-bond reduction steps found in the conventional method. It utilizes a direct hydrolysis and rearrangement strategy that operates under milder conditions, resulting in higher overall yields and reduced operational complexity.
Q: What specific catalysts are required for the rearrangement step?
A: The rearrangement transformation from the intermediate aldehyde to the final product effectively utilizes transition metal catalysts such as bisacetonitrile palladium chloride or cuprous chloride. These catalysts are employed in low loadings (0.5-2% by mass), ensuring cost-efficiency while maintaining high conversion rates.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly scalable due to its use of common solvents like acetonitrile and DMF, and moderate temperature ranges (50-100°C). The elimination of hazardous reagents like acetylene gas and the simplification of purification steps make it ideal for commercial scale-up with improved safety profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Acetoxy-2-methyl-2-butene-1-aldehyde Supplier
The transition to more efficient synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of fine chemical production, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art reactors capable of handling the specific temperature and pressure requirements of the hydrolysis and rearrangement steps described in CN102311339B. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of 4-acetoxy-2-methyl-2-butene-1-aldehyde meets the exacting standards required for Vitamin A synthesis.
We invite global pharmaceutical and agrochemical companies to collaborate with us to leverage this advanced technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on the most efficient and reliable foundation available in the market.
