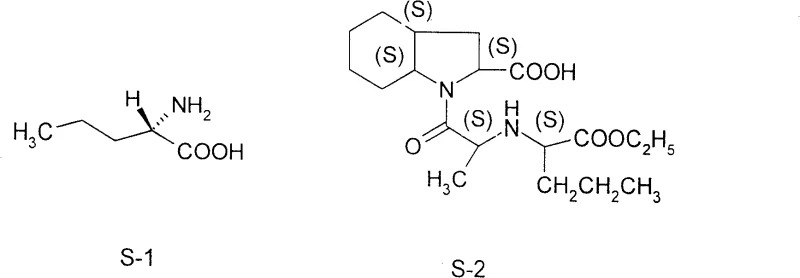
Scalable Synthesis of L-Norvaline: A Strategic Breakthrough for API Manufacturing
Introduction to Patent CN100427460C Technology
The pharmaceutical industry continuously seeks robust, scalable pathways for non-proteinogenic amino acids, which serve as critical chiral building blocks for complex drug molecules. Patent CN100427460C, published in October 2008, discloses a highly efficient synthetic method for L-norvaline, a key intermediate in the production of antihypertensive agents such as perindopril. This technology represents a significant departure from traditional biological fermentation or hazardous chemical routes, offering a streamlined six-step process that begins with the readily available commodity chemical, n-valeric acid. By leveraging standard unit operations such as acylation, halogenation, and chiral resolution, this method addresses the longstanding challenges of yield and scalability associated with amino acid manufacturing. For R&D directors and process chemists, understanding the nuances of this patent is essential for developing supply chains that are both resilient and cost-effective.
The strategic value of this synthesis lies in its ability to bypass the limitations of earlier methodologies. Historically, the production of L-norvaline has been bottlenecked by either the low productivity of fermentation strains or the severe safety hazards of cyanide-based chemistry. The approach detailed in CN100427460C utilizes a logical progression of functional group transformations that are well-understood in fine chemical manufacturing, thereby reducing technical risk. As a reliable pharmaceutical intermediate supplier, analyzing this patent reveals a pathway that balances chemical elegance with industrial pragmatism, ensuring that high-purity materials can be delivered consistently to meet the rigorous demands of global API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations described in this patent, the landscape for L-norvaline synthesis was fraught with significant technical and economic inefficiencies. One prominent method, documented in patent JP7553587, relied on microbial fermentation. While fermentation is often praised for its stereoselectivity, in this specific case, the reported titer was merely approximately 3.7g/L. Such low productivity is economically unsustainable for large-scale industrial applications, as it necessitates massive bioreactor volumes and extensive downstream processing to isolate the product, leading to prohibitive operational expenditures. Furthermore, fermentation processes are inherently sensitive to contamination and strain degeneration, introducing variability that supply chain managers cannot afford when supporting multi-ton API campaigns.
Alternatively, existing chemical synthesis routes, such as that described in Chinese patent CN1651400, presented severe safety and environmental liabilities. These methods frequently employed acetone cyanohydrin as a starting material. Acetone cyanohydrin is a highly toxic compound that poses acute risks to personnel and requires specialized, costly infrastructure for containment and waste treatment. The use of such hazardous reagents not only increases the capital expenditure for plant safety systems but also complicates regulatory compliance and environmental permitting. For procurement teams, reliance on processes involving restricted or dangerous chemicals introduces supply volatility, as raw material availability can be impacted by shifting global safety regulations and transportation restrictions.
The Novel Approach
The methodology outlined in CN100427460C offers a transformative solution by utilizing n-valeric acid as the primary feedstock. This starting material is a bulk commodity chemical, widely available from petrochemical sources, which immediately stabilizes the supply chain and reduces raw material costs. The process initiates with the conversion of n-valeric acid to n-valeryl chloride using thionyl chloride, followed by alpha-bromination. This sequence effectively activates the carbon chain for subsequent nucleophilic substitution without introducing toxic cyanide groups. The transition from hazardous cyanohydrins to a halogenated acid chloride pathway represents a fundamental shift towards greener chemistry, aligning with modern ESG (Environmental, Social, and Governance) goals prevalent in the pharmaceutical sector.
Furthermore, the novel approach integrates a classical resolution strategy using L-tartaric acid, a renewable and inexpensive chiral pool material. This step allows for the precise isolation of the L-enantiomer from the racemic mixture generated during ammonolysis. By combining robust chemical transformations with a proven resolution technique, the process achieves a balance between stereochemical control and operational simplicity. The elimination of complex biocatalytic steps and toxic reagents results in a shorter production cycle and a simplified workflow. This streamlining is crucial for cost reduction in API manufacturing, as it minimizes the number of isolation steps and reduces the overall solvent consumption, directly impacting the bottom line for commercial production.
Mechanistic Insights into Alpha-Halo Acid Chloride Synthesis and Resolution
The core of this synthetic route relies on the precise manipulation of the alpha-carbon of the valeric acid chain. The process begins with acyl chlorination, where thionyl chloride reacts with the carboxylic acid group. This activation is critical because the resulting acid chloride is significantly more electrophilic than the parent acid, facilitating the subsequent alpha-bromination. In the bromination step, liquid bromine reacts with the enolizable alpha-position of the acid chloride. The reaction conditions, typically maintained between 50°C and 80°C, are optimized to ensure mono-bromination while minimizing poly-halogenation side products. The generation of alpha-bromo-n-valeryl chloride is a pivotal moment in the synthesis, as it installs the leaving group necessary for the introduction of the nitrogen atom.
Following bromination, the mechanism shifts to nucleophilic substitution via ammonolysis. Liquid ammonia acts as a potent nucleophile, displacing the bromide ion to form the primary amine functionality, yielding racemic alpha-aminovaleramide. The use of liquid ammonia under pressure ensures high concentration and reaction rates, driving the equilibrium towards the amide product. However, since this step produces a racemate, the subsequent resolution is mechanistically vital. The process exploits the difference in solubility between diastereomeric salts. By reacting the racemic amide with L-tartaric acid in methanol, the L-aminovaleramide-L-tartrate salt precipitates preferentially. This thermodynamic control allows for the enrichment of the desired L-isomer, which is further purified through recrystallization to achieve the stringent optical purity required for pharmaceutical applications.
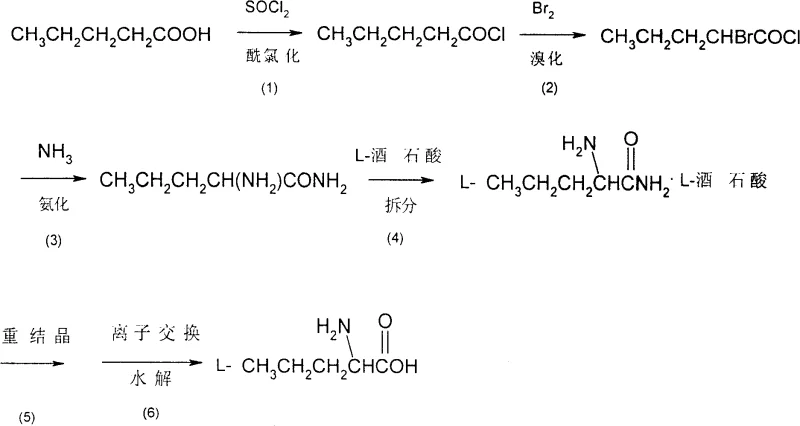
Impurity control is managed through the physical properties of the intermediates. The crude alpha-bromo-n-valeryl chloride is purified via vacuum distillation, removing unreacted starting materials and poly-brominated byproducts before they can propagate through the synthesis. Similarly, the resolution step acts as a powerful purification gate; impurities that do not fit the crystal lattice of the tartrate salt remain in the mother liquor. The final hydrolysis step utilizes a cation exchange resin, which not only cleaves the amide bond to release the free amino acid but also serves to trap residual metal ions or organic impurities. This dual function of the resin simplifies the workup, eliminating the need for extensive extraction sequences and ensuring that the final L-norvaline meets high purity specifications without requiring chromatographic purification.
How to Synthesize L-Norvaline Efficiently
The synthesis of L-norvaline via this patented route involves a sequence of six distinct operational stages, each requiring precise control of temperature, stoichiometry, and reaction time to maximize yield and optical purity. The process transforms simple n-valeric acid into a high-value chiral amino acid through activation, functionalization, and stereochemical selection. Understanding the critical parameters at each stage—such as the molar ratio of thionyl chloride in the acylation step or the temperature profile during tartaric acid resolution—is essential for successful technology transfer from the laboratory to the pilot plant. The following guide outlines the standardized workflow derived from the patent examples, providing a roadmap for process engineers to replicate the reported efficiencies.
- Acylate n-valeric acid with thionyl chloride to form n-valeryl chloride.
- Brominate the acid chloride with liquid bromine to obtain alpha-bromo-n-valeryl chloride.
- Perform ammonolysis with liquid ammonia to generate racemic alpha-aminovaleramide.
- Resolve the racemate using L-tartaric acid to isolate the L-enantiomer salt.
- Hydrolyze the resolved salt using cation exchange resin to yield pure L-norvaline.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the CN100427460C synthesis route offers tangible strategic benefits that extend beyond mere chemical feasibility. The primary advantage lies in the decoupling of production from agricultural or biological constraints. Unlike fermentation, which is susceptible to seasonal variations in feedstock quality and biological contamination, this chemical synthesis relies on petrochemical-derived n-valeric acid. This ensures a consistent, year-round supply of starting materials, drastically reducing the risk of production stoppages. The stability of the raw material supply chain is a critical factor for long-term contracts with API manufacturers, who require guaranteed continuity to support their own regulatory filings and market commitments.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the use of low-cost, commodity-grade reagents. By avoiding expensive chiral catalysts or specialized enzymes, the variable cost of goods sold (COGS) is significantly minimized. Furthermore, the elimination of toxic acetone cyanohydrin removes the substantial overhead costs associated with hazardous waste disposal and specialized safety monitoring. The process utilizes standard glass-lined or stainless steel reactors common in fine chemical plants, meaning no exotic metallurgy or high-pressure bioreactors are required. This compatibility with existing infrastructure allows for rapid deployment without heavy capital investment, translating to lower amortization costs per kilogram of product.
- Enhanced Supply Chain Reliability: The robustness of the chemical steps ensures high predictability in lead times. The reaction conditions, such as the reflux temperatures and atmospheric or moderate pressure requirements, are easily controllable on a multi-ton scale. The resolution step using L-tartaric acid is a mature technology with well-established supply chains for the resolving agent itself. This redundancy in the supply of key reagents means that single-source bottlenecks are minimized. For supply chain planners, this translates to a more resilient vendor network capable of absorbing demand shocks without compromising delivery schedules, a crucial attribute in the post-pandemic pharmaceutical landscape.
- Scalability and Environmental Compliance: Scaling this process is straightforward due to the linear nature of the reaction sequence. Each step, from distillation to crystallization, is a unit operation that scales predictably from liters to cubic meters. Moreover, the environmental footprint is markedly lower than alternative routes. The absence of cyanide waste streams simplifies effluent treatment, reducing the load on wastewater treatment plants and lowering compliance costs. The ability to recover and recycle solvents like chloroform or methanol, as implied by the concentration steps, further enhances the sustainability profile. This alignment with green chemistry principles facilitates smoother regulatory approvals and enhances the brand reputation of the final drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this L-norvaline synthesis technology. These insights are derived directly from the experimental data and claims within patent CN100427460C, providing clarity on yield expectations, purity profiles, and operational safety. Understanding these details is vital for technical teams evaluating the feasibility of integrating this intermediate into their broader manufacturing workflows.
Q: What are the advantages of this chemical synthesis over fermentation?
A: Unlike fermentation methods which suffer from low titers (approx. 3.7g/L) and long cycles, this chemical route uses commodity starting materials (n-valeric acid) allowing for significantly higher throughput and shorter production cycles suitable for industrial scale.
Q: How is chirality controlled in this process?
A: Chirality is introduced via classical resolution using L-tartaric acid. The process forms a diastereomeric salt (L-aminovaleramide tartrate) which is purified through recrystallization, ensuring high optical purity before final hydrolysis.
Q: Does this method avoid toxic reagents found in prior art?
A: Yes. Unlike previous chemical methods (e.g., CN1651400) that utilized highly toxic acetone cyanohydrin, this protocol relies on safer reagents like thionyl chloride and liquid ammonia, reducing hazardous waste disposal costs and improving operator safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Norvaline Supplier
The synthesis of L-norvaline described in CN100427460C exemplifies the type of innovative process chemistry that drives value in the modern pharmaceutical supply chain. At NINGBO INNO PHARMCHEM, we possess the technical expertise to translate such patented methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory success is faithfully reproduced at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of L-norvaline meets the exacting standards required for GMP API synthesis.
We invite global partners to collaborate with us to optimize their supply chains for perindopril and other related therapeutics. By leveraging our process development capabilities, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can secure a sustainable, high-quality supply of this critical chiral intermediate, ensuring your production timelines are met with precision and reliability.
