Advanced Chemical Synthesis of L-Norvaline: A Scalable Route for Pharmaceutical Intermediates
Advanced Chemical Synthesis of L-Norvaline: A Scalable Route for Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks robust, scalable, and cost-effective methods for producing chiral amino acids, which serve as critical building blocks for complex active pharmaceutical ingredients (APIs). Patent CN1651400A introduces a groundbreaking chemical synthesis method for L-norvaline, a non-proteinogenic amino acid that acts as a pivotal intermediate in the manufacture of Perindopril, a widely prescribed antihypertensive agent. Unlike traditional biological fermentation routes that often struggle with low volumetric productivity and complex purification requirements, this novel chemical approach leverages readily available petrochemical derivatives to achieve high efficiency. The process transforms n-butyraldehyde and acetone cyanohydrin into high-purity L-norvaline through a sequence of cyanation, ammonification, and chiral resolution steps. This technological shift represents a significant advancement for reliable L-norvaline suppliers aiming to secure the supply chain for cardiovascular medications. By moving away from the biological variability of fermentation, manufacturers can ensure consistent quality and batch-to-batch reproducibility, which are paramount for regulatory compliance in global markets.
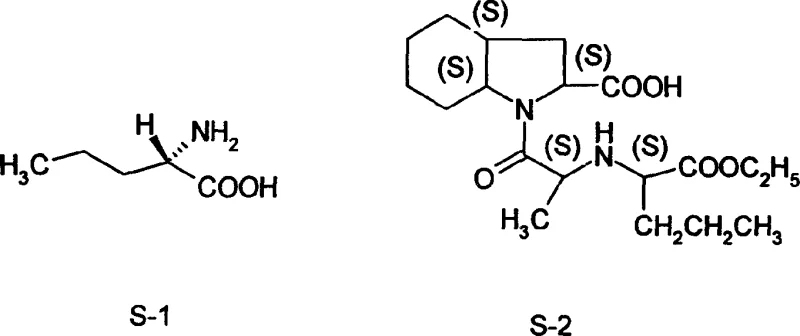
The strategic importance of L-norvaline extends beyond its immediate chemical utility; it is the structural cornerstone for Perindopril, as illustrated in the molecular relationship between structure S-1 and S-2. The ability to synthesize this moiety chemically rather than biologically opens new avenues for cost reduction in pharmaceutical intermediates manufacturing. Traditional fermentation processes for amino acids often require extensive nutrient media, sterile conditions, and energy-intensive separation techniques to isolate the product from the broth. In contrast, the chemical route described in the patent utilizes standard organic synthesis unit operations that are easily integrated into existing fine chemical infrastructure. This compatibility reduces the barrier to entry for production and allows for rapid scaling to meet surging global demand for ACE inhibitors. Furthermore, the process avoids the use of genetically modified organisms (GMOs), simplifying regulatory filings and environmental impact assessments in jurisdictions with strict biosafety laws.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of L-norvaline has been heavily reliant on fermentation technologies, a method that presents inherent bottlenecks for industrial-scale manufacturing. As noted in the background art of the patent, fermentation yields for L-norvaline are notoriously low, often hovering around 3.7g/L, which is substantially below the productivity levels seen in other amino acid fermentations. This low titer necessitates the processing of massive volumes of fermentation broth to recover small quantities of the product, leading to exorbitant costs in water usage, energy consumption for evaporation, and waste treatment. Additionally, fermentation processes are susceptible to biological contamination and strain degeneration, which can halt production lines and compromise supply continuity. The downstream processing is equally challenging, requiring complex ion-exchange chromatography and crystallization steps to remove cellular debris, proteins, and other metabolic byproducts that co-elute with the target amino acid. These factors combined make fermentation an economically fragile strategy for meeting the high-volume demands of the generic pharmaceutical market.
The Novel Approach
The chemical synthesis route disclosed in patent CN1651400A effectively circumvents these biological limitations by employing a deterministic, step-wise organic transformation. By starting with n-butyraldehyde and acetone cyanohydrin, the process establishes a linear and predictable reaction pathway that is independent of biological variables. The initial cyanation step creates the carbon skeleton with high atom economy, while the subsequent ammonification introduces the nitrogen functionality under controlled pressure and temperature conditions. This chemical precision allows for the optimization of each reaction step independently, maximizing overall yield without the cross-talk issues common in metabolic engineering. Moreover, the use of L-tartaric acid for chiral resolution provides a highly selective method for isolating the desired L-enantiomer, ensuring that the final product meets the stringent optical purity requirements necessary for API synthesis. This approach not only enhances commercial scale-up of complex amino acids but also significantly shortens the production cycle time, enabling manufacturers to respond more agilely to market fluctuations.
Mechanistic Insights into Chemical Synthesis and Chiral Resolution
The core of this synthesis lies in the efficient construction of the norvaline carbon backbone followed by the precise installation of chirality. The process begins with a base-catalyzed cyanation where n-butyraldehyde reacts with acetone cyanohydrin. In this step, the cyanide group acts as a nucleophile, attacking the carbonyl carbon of the aldehyde to form a cyanohydrin intermediate. The reaction conditions are meticulously controlled, typically maintaining a pH between 7 and 9 using alkaline catalysts like potassium hydroxide or sodium hydroxide in methanol. This specific pH range is critical to prevent the polymerization of the aldehyde while ensuring sufficient nucleophilicity of the cyanide species. Following this, the cyanohydrin undergoes ammonification with liquid ammonia. This high-pressure reaction replaces the hydroxyl group with an amine, generating aminopentylonitrile. The use of liquid ammonia in excess drives the equilibrium forward and minimizes side reactions, resulting in a crude nitrile that is ready for hydrolysis without extensive purification.
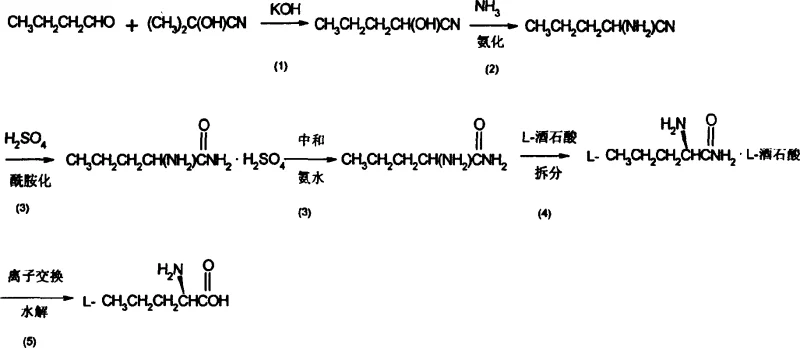
The final and perhaps most critical stage involves the hydrolysis of the nitrile and the subsequent chiral resolution. The aminopentylonitrile is hydrolyzed in concentrated sulfuric acid to form the racemic amino valeramide sulfate salt. Neutralization with ammonia liberates the free racemic amide, which is then subjected to resolution using L-tartaric acid. This diastereomeric salt formation is a classic yet powerful technique for enantiomeric separation; the L-amino valeramide-L-tartrate salt exhibits significantly lower solubility in methanol compared to its D-counterpart, allowing it to crystallize out selectively. The patent specifies recrystallization from a water-methanol mixture to further enhance optical purity. Finally, the resolved amide is hydrolyzed on a cationic exchange resin column. This solid-phase hydrolysis not only converts the amide to the free acid (L-norvaline) but also simultaneously captures the tartaric acid for recycling, demonstrating a clever integration of reaction and separation that minimizes waste and maximizes resource efficiency.
How to Synthesize L-Norvaline Efficiently
The synthesis of L-norvaline via this chemical route requires careful attention to reaction parameters, particularly temperature control during the exothermic cyanation and pressure management during ammonification. The process is designed to be telescoped where possible, minimizing the isolation of unstable intermediates like the cyanohydrin. Operators must ensure strict stoichiometric control of the starting materials to prevent the formation of oligomeric byproducts. The resolution step is sensitive to cooling rates and solvent composition; slow addition of the resolving agent and controlled crystallization temperatures are essential for achieving high enantiomeric excess. For detailed operational parameters, safety guidelines, and specific workup procedures, please refer to the standardized protocol below.
- Perform cyanation of n-butyraldehyde with acetone cyanohydrin under alkaline catalysis to form butyraldehyde cyanohydrin.
- React the cyanohydrin with liquid ammonia to produce aminopentylonitrile, followed by dehydration.
- Hydrolyze the nitrile in sulfuric acid to obtain racemic amino valeramide, then resolve using L-tartaric acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from fermentation to this chemical synthesis route offers compelling economic and logistical benefits. The primary advantage lies in the raw material profile; n-butyraldehyde and acetone cyanohydrin are bulk commodity chemicals produced on a multi-million-ton scale globally. This ensures a stable, diversified supply base that is immune to the agricultural or biological disruptions that can affect fermentation substrates. Furthermore, the elimination of fermentation tanks and the associated sterilization infrastructure drastically reduces capital expenditure (CAPEX) for new production facilities. The process relies on standard glass-lined or stainless steel reactors found in any typical fine chemical plant, allowing for immediate technology transfer without the need for specialized bioreactor installations. This flexibility translates directly into reducing lead time for high-purity amino acid intermediates, as production slots can be secured more easily in multi-purpose facilities.
- Cost Reduction in Manufacturing: The chemical route eliminates the high operational costs associated with maintaining sterile fermentation environments and processing large volumes of dilute broth. By concentrating the reaction mass through efficient organic synthesis steps, the energy required for solvent recovery and product drying is significantly lower than the thermal energy needed to evaporate water from fermentation broths. Additionally, the recovery and recycling of L-tartaric acid from the ion-exchange mother liquor further lowers the net cost of goods sold (COGS). The simplified downstream processing, which avoids complex chromatographic separations typical of bioprocessing, reduces both consumable costs and labor hours, driving substantial overall cost efficiency.
- Enhanced Supply Chain Reliability: Relying on petrochemical feedstocks rather than biological strains mitigates the risk of production failure due to phage attacks or strain mutations. The chemical process is robust and reproducible, ensuring that delivery schedules are met consistently. The modular nature of the synthesis steps allows for parallel processing or campaign production, providing the flexibility to ramp up output quickly in response to sudden increases in demand for Perindopril. This reliability is crucial for long-term supply agreements with major pharmaceutical companies who prioritize continuity of supply above all else.
- Scalability and Environmental Compliance: The process generates less aqueous waste compared to fermentation, as the reaction volumes are smaller and solvent recovery rates are high. The use of ion-exchange resins for hydrolysis minimizes the use of harsh liquid acids and bases in the final steps, simplifying wastewater treatment. The scalability is linear; moving from pilot scale to multi-ton production simply requires increasing reactor size without re-optimizing biological parameters. This ease of scale-up ensures that the technology remains viable and cost-effective even at very high production volumes, supporting the growing global market for cardiovascular drugs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of L-norvaline using this patented chemical method. These insights are derived directly from the experimental data and process descriptions within patent CN1651400A, providing clarity on the feasibility and advantages of this synthetic route for industry stakeholders.
Q: Why is chemical synthesis preferred over fermentation for L-norvaline?
A: Chemical synthesis offers significantly higher yields and shorter production cycles compared to fermentation, which typically suffers from low output (around 3.7g/L) and complex downstream processing.
Q: What are the key starting materials for this process?
A: The process utilizes readily available commodity chemicals: n-butyraldehyde and acetone cyanohydrin, ensuring stable supply chains and cost efficiency.
Q: How is chirality controlled in this synthesis?
A: Chirality is introduced through a classical resolution step using L-tartaric acid to separate the L-enantiomer from the racemic amino valeramide intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Norvaline Supplier
The synthesis of L-norvaline via the chemical route described in CN1651400A represents a mature, industrially viable technology that aligns perfectly with the needs of modern pharmaceutical manufacturing. NINGBO INNO PHARMCHEM stands at the forefront of adopting such innovative processes, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with the necessary corrosion-resistant reactors and high-pressure equipment required for the ammonification and hydrolysis steps, ensuring that we can deliver this critical intermediate with consistent quality. We maintain stringent purity specifications and operate rigorous QC labs to verify the optical rotation and chemical purity of every batch, guaranteeing that our L-norvaline meets the exacting standards required for Perindopril synthesis.
We invite pharmaceutical partners to collaborate with us to optimize their supply chains for cardiovascular APIs. By choosing our chemically synthesized L-norvaline, you gain access to a more stable and cost-efficient supply source. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term production goals.
