Revolutionizing PCMX Production: A Catalyst-Free, Water-Based Industrial Approach
Revolutionizing PCMX Production: A Catalyst-Free, Water-Based Industrial Approach
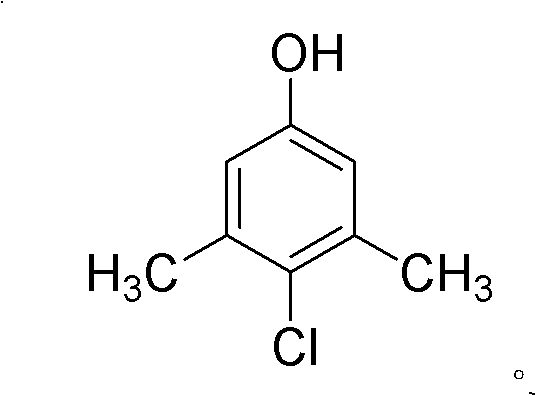
The global demand for high-purity antimicrobial agents continues to surge, driven by heightened health awareness and stringent regulatory standards in the pharmaceutical and personal care sectors. At the forefront of this evolution is the green industrialized preparation method for 1-hydroxy-3,5-dimethyl-4-chlorobenzene, commonly known as PCMX, as detailed in patent CN101823941A. This groundbreaking technology represents a paradigm shift from traditional halogenation processes, moving away from hazardous organic solvents and expensive, difficult-to-recover catalysts towards a sustainable, water-based system. By leveraging a unique multi-stage temperature control strategy, this method achieves exceptional para-selectivity and conversion rates while drastically minimizing environmental footprint. For R&D directors and procurement leaders, understanding the mechanistic underpinnings and commercial implications of this patent is critical for securing a competitive edge in the supply of high-value fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-hydroxy-3,5-dimethyl-4-chlorobenzene has relied heavily on electrophilic substitution reactions utilizing aggressive catalyst systems and toxic solvent environments. Prior art, such as US Patent 3,920,757 and US Patent 4,245,127, describes methods employing co-catalysts like diphenyl sulfide and metal halides (e.g., ferric chloride) within inert organic solvent systems like tetrachloroethylene. These conventional approaches suffer from significant drawbacks that impact both operational expenditure and environmental compliance. The use of diphenyl sulfide introduces a costly reagent that is notoriously difficult to recover and recycle, leading to substantial material loss and increased waste disposal costs. Furthermore, the reliance on chlorinated organic solvents like tetrachloroethylene poses severe environmental hazards due to volatile organic compound (VOC) emissions and the generation of hazardous waste streams that require complex treatment protocols. Additionally, these methods often struggle with selectivity issues, producing unwanted ortho-chlorinated by-products and dichlorinated impurities that necessitate energy-intensive purification steps to meet pharmaceutical-grade specifications.
The Novel Approach
In stark contrast, the methodology outlined in CN101823941A introduces a remarkably elegant solution by utilizing water as the sole reaction medium and completely eliminating the need for external catalysts. This novel approach capitalizes on the intrinsic physicochemical properties of the reactants and the solvent to drive selectivity, rather than relying on expensive additives. The process employs sulfuryl chloride or chlorine gas as the chlorinating agent, reacting with 1-hydroxy-3,5-dimethylbenzene in an aqueous suspension. The true innovation lies in the sophisticated multi-stage temperature control profile, which dynamically manages the physical state of the reaction mixture to overcome mass transfer limitations. By operating initially at lower temperatures (0-70°C) and subsequently cycling through a melting phase (75-100°C), the process ensures that unreacted substrate trapped within product crystals is released for further conversion. This eliminates the "wrapping" effect common in solid-liquid reactions, ensuring near-complete substrate consumption and maximizing the yield of the desired para-isomer without the baggage of toxic solvents or catalyst residues.
Mechanistic Insights into Water-Mediated Para-Selective Chlorination
The exceptional selectivity observed in this water-based system is not merely a result of solubility differences but stems from a profound molecular interaction between the solvent and the substrate. The inventors discovered that water molecules form strong intermolecular hydrogen bonds with the hydroxyl group of the 1-hydroxy-3,5-dimethylbenzene substrate. This hydrogen bonding network effectively increases the steric hindrance around the ortho-positions of the aromatic ring, physically blocking the approach of the chlorinating agent to these sites. Consequently, the electrophilic attack is directed almost exclusively to the para-position, which remains sterically accessible. This phenomenon is negatively correlated with temperature; at lower temperatures, the hydrogen bonds are more stable and the steric shielding effect is more pronounced, thereby suppressing the formation of ortho-chlorinated by-products. This intrinsic selectivity mechanism allows the process to achieve high purity levels directly from the reaction, reducing the burden on downstream purification units and ensuring a cleaner impurity profile suitable for sensitive applications in cosmetics and pharmaceuticals.
Beyond selectivity, the thermal management strategy addresses a critical kinetic bottleneck inherent in heterogeneous reactions involving melting solids. The substrate, 1-hydroxy-3,5-dimethylbenzene, has a melting point between 63-65°C, while the chlorinated product has a significantly higher melting point. In traditional low-temperature reactions, the solid product tends to crystallize on the surface of the remaining solid substrate, creating a physical barrier that prevents the chlorinating agent from reaching the unreacted core. This "encapsulation" leads to incomplete conversion and forces the reaction towards polychlorination of the exposed product surface. The patented multi-stage temperature control resolves this by periodically heating the reaction mass to 75-100°C. This thermal spike melts the product shell, liberating the trapped substrate into the aqueous phase where it can react efficiently. Subsequent cooling allows the product to recrystallize, ideally leaving the remaining substrate exposed for the next cycle of chlorination. This dynamic cycling ensures that the reaction proceeds to >99.5% conversion while maintaining high selectivity for the mono-chlorinated species.
How to Synthesize 4-Chloro-3,5-Dimethylphenol Efficiently
Implementing this green synthesis route requires precise adherence to the thermal cycling protocol to maximize the benefits of the water-mediated mechanism. The process begins with the suspension of the phenolic substrate in water, followed by the controlled addition of the chlorinating agent under mild conditions to establish initial selectivity. As the reaction progresses and conversion reaches the 50-80% range, the system undergoes a deliberate thermal excursion to disrupt product crystallization and expose fresh substrate surfaces.
- Mix water and 1-hydroxy-3,5-dimethylbenzene substrate, maintaining temperature between 15-55°C while adding sulfuryl chloride.
- Stop reaction at 50-80% conversion, then heat the solution to 75-100°C to melt product crystals and release trapped substrate.
- Cool the solution back to 15-55°C and resume chlorination, repeating the heating cycle until substrate conversion exceeds 99.5%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this catalyst-free, aqueous process offers transformative economic and logistical benefits that extend far beyond simple raw material costs. The elimination of expensive and difficult-to-recover catalysts like diphenyl sulfide and metal halides removes a significant line item from the bill of materials, while simultaneously simplifying the downstream processing workflow. Without the need to strip heavy metals or sulfur residues from the final product, manufacturers can bypass complex purification stages such as activated carbon treatment or extensive washing protocols, leading to substantial reductions in utility consumption and processing time. Furthermore, the removal of toxic organic solvents like tetrachloroethylene eradicates the costs associated with solvent recovery distillation columns and hazardous waste disposal fees, resulting in a leaner, more cost-effective manufacturing operation that is resilient to fluctuating solvent markets.
- Cost Reduction in Manufacturing: The complete absence of catalysts and organic solvents fundamentally alters the cost structure of PCMX production. By removing the requirement for diphenyl sulfide and ferric chloride, the process eliminates the capital and operational expenditures linked to catalyst recovery systems and the purchase of these high-value reagents. Additionally, the simplified work-up procedure, which relies on basic filtration and drying rather than complex solvent exchanges or chromatographic separations, drastically reduces labor and energy inputs. This streamlined workflow translates directly into lower unit costs, allowing suppliers to offer more competitive pricing structures while maintaining healthy margins in a price-sensitive market.
- Enhanced Supply Chain Reliability: Relying on water as a solvent and readily available chlorinating agents like sulfuryl chloride or chlorine gas significantly de-risks the supply chain compared to methods dependent on specialized organic solvents. Tetrachloroethylene and similar chlorinated solvents are subject to strict environmental regulations and supply volatility, which can lead to production stoppages. By shifting to a water-based system, manufacturers insulate themselves from these external supply shocks, ensuring consistent production continuity. Moreover, the robustness of the multi-stage temperature control allows for scalable operations that can adapt to varying batch sizes without compromising quality, ensuring reliable delivery schedules for downstream customers in the pharmaceutical and agrochemical sectors.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this technology offers a clear pathway to sustainable manufacturing. The avoidance of VOC emissions and the generation of non-hazardous aqueous waste streams simplify regulatory compliance and reduce the liability associated with environmental permits. The process inherently produces less solid waste since there are no spent catalysts to dispose of, aligning perfectly with modern green chemistry principles. This environmental superiority not only future-proofs the manufacturing site against tightening global regulations but also enhances the brand value of the final product, appealing to end-users who prioritize sustainably sourced ingredients in their formulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green chlorination technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a factual basis for evaluating the feasibility of adopting this method for large-scale production.
Q: Why is water used as a solvent instead of organic solvents in this PCMX synthesis?
A: Water forms intermolecular hydrogen bonds with the hydroxyl group of the substrate, increasing steric hindrance at the ortho-position. This naturally suppresses ortho-chlorination by-products, achieving high para-selectivity without needing toxic organic solvents like tetrachloroethylene.
Q: How does the multi-stage temperature control improve yield?
A: The product has a higher melting point than the substrate. During reaction, solid product can wrap around unreacted substrate, blocking further chlorination. Heating to 75-100°C melts the product shell, releasing the substrate for further reaction, thereby pushing conversion rates above 99.5%.
Q: What are the advantages of eliminating metal catalysts in this process?
A: Traditional methods use expensive catalysts like diphenyl sulfide and ferric chloride which are difficult to recover. Eliminating them removes the need for complex purification steps to remove heavy metals and sulfur residues, significantly lowering production costs and environmental waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Chloro-3,5-Dimethylphenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced, sustainable manufacturing technologies to meet the evolving needs of the global pharmaceutical and fine chemical industries. Our expertise extends beyond simple execution; we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory concepts like the water-based PCMX synthesis are successfully translated into robust industrial realities. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 4-chloro-3,5-dimethylphenol meets the highest standards of quality and consistency required for sensitive antimicrobial applications.
We invite potential partners to engage with our technical team to explore how this green synthesis route can be integrated into your supply chain to drive efficiency and sustainability. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the specific economic benefits tailored to your volume requirements. We encourage you to contact our technical procurement team today to obtain specific COA data and route feasibility assessments, ensuring that your sourcing strategy is built on a foundation of scientific excellence and commercial reliability.
