Advanced Oxychlorination Technology for High-Purity PCMX Commercial Production
The chemical industry is currently witnessing a significant paradigm shift towards greener synthesis methodologies, particularly in the production of high-value microbicides and disinfectant intermediates. Patent CN101624333A introduces a groundbreaking preparation method for 1-hydroxyl-3,5-dimethyl-4-chlorobenzene, widely known in the industry as PCMX. This compound serves as a critical active ingredient with potent antibacterial and antifungal properties, extensively utilized in pharmaceutical formulations, industrial sanitizers, and cosmetic preservatives. The patented technology addresses long-standing inefficiencies in traditional chlorination processes by employing a novel oxychlorination strategy. Instead of relying on hazardous chlorinating agents that produce toxic by-products, this method utilizes a catalytic system composed of divalent copper salts and lower fatty acids. 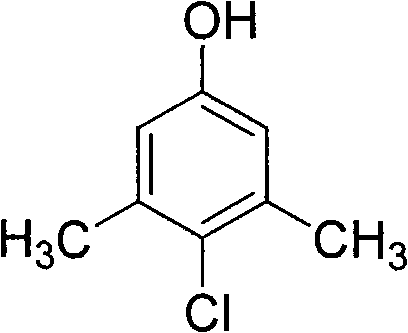 . This structural precision is achieved under mild reaction conditions ranging from 0°C to 100°C and pressures between 1 to 5 atm. For R&D directors and technical procurement officers, this patent represents a viable pathway to enhance product purity while adhering to increasingly stringent environmental regulations. The ability to synthesize this key intermediate with high atom economy positions it as a superior choice for modern supply chains seeking sustainability without compromising on yield or quality standards.
. This structural precision is achieved under mild reaction conditions ranging from 0°C to 100°C and pressures between 1 to 5 atm. For R&D directors and technical procurement officers, this patent represents a viable pathway to enhance product purity while adhering to increasingly stringent environmental regulations. The ability to synthesize this key intermediate with high atom economy positions it as a superior choice for modern supply chains seeking sustainability without compromising on yield or quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 1-hydroxyl-3,5-dimethyl-4-chlorobenzene has relied heavily on electrophilic substitution reactions using sulfuryl chloride as the primary chlorinating agent. While this method was established as early as the 19th century, modern analysis reveals significant drawbacks that hinder its viability in contemporary manufacturing environments. The reaction typically generates equimolar amounts of hydrogen chloride and sulfur dioxide as by-products, leading to an extremely low atom economy of approximately 26.3%. These gaseous by-products are highly corrosive and pose severe challenges for waste management and equipment maintenance. Furthermore, the traditional catalyst systems, often involving complex organosulfur compounds and metal halides, struggle to maintain high regioselectivity. This results in the formation of unwanted ortho-isomers and polychlorinated impurities, which complicate downstream purification processes and increase overall production costs. The environmental burden of disposing of sulfur-containing waste, often through low-value recovery or direct emission, creates substantial liability for manufacturers. Consequently, the conventional route is increasingly viewed as unsustainable for large-scale commercial operations aiming for green chemistry certification and cost efficiency.
The Novel Approach
The innovative method disclosed in the patent fundamentally reengineers the chlorination process by replacing sulfuryl chloride with an oxychlorination system. This approach utilizes inexpensive and readily available chlorine sources such as hydrochloric acid or alkali metal chlorides, coupled with oxygen or air as the oxidant. The core breakthrough lies in the specific catalytic combination of divalent copper salts and lower fatty acids, which work synergistically to activate the chlorine source and direct the substitution to the para-position. This eliminates the generation of sulfur dioxide entirely, thereby resolving the major environmental and corrosion issues associated with the legacy process. The reaction conditions are remarkably mild, operating effectively between 40°C and 90°C, which reduces energy consumption compared to high-temperature alternatives. Moreover, the system demonstrates exceptional flexibility regarding the oxidant, allowing manufacturers to choose between oxygen, air, or hydrogen peroxide based on specific kinetic requirements. By avoiding the use of organic solvents or aliphatic chlorides in certain embodiments, the process further simplifies the workup procedure. This novel approach not only aligns with global sustainability goals but also offers a robust framework for scaling up production without the regulatory hurdles associated with sulfur emissions.
Mechanistic Insights into Cu(II)-Catalyzed Oxychlorination
The catalytic mechanism underpinning this synthesis involves a sophisticated interplay between the divalent copper species and the fatty acid co-catalyst. The divalent copper salt, such as copper chloride or copper acetate, acts as the primary redox mediator, facilitating the oxidation of the chloride ion to an active chlorinating species. The lower fatty acid, including acetic acid or propionic acid, serves a dual role as both a co-catalyst and a potential solvent. It stabilizes the copper complex and enhances the solubility of the reactants, thereby accelerating the reaction rate significantly. Experimental data suggests a positive correlation between the catalyst loading and the reaction speed, with optimal performance observed when the copper salt constitutes 10% to 60% of the substrate weight. The presence of the fatty acid is critical for achieving high para-selectivity; without it, the reaction yields drop precipitously, and selectivity for the desired isomer diminishes. This synergistic effect ensures that the electrophilic attack occurs predominantly at the 4-position of the phenolic ring, minimizing the formation of 2-chloro or 2,4-dichloro impurities. The mechanism allows for the continuous regeneration of the active catalytic species, enabling the system to maintain high turnover numbers over extended reaction periods.
Impurity control is another critical aspect where this mechanistic design excels. In traditional methods, the formation of polychlorinated by-products is a persistent issue that requires extensive purification steps, often involving recrystallization or chromatography. The copper-fatty acid system inherently suppresses over-chlorination due to the controlled generation of the active chlorine species. The selectivity for the mono-chlorinated product consistently exceeds 96%, with conversion rates reaching nearly 99.7% in optimized examples. This high level of specificity means that the crude product requires minimal downstream processing to meet stringent pharmaceutical or industrial grade specifications. Furthermore, the ability to recycle the catalytic system and the fatty acid co-catalyst reduces the accumulation of metal waste in the effluent. The process also accommodates various chlorine sources, including alkali metal chlorides, which can be regenerated in situ by reacting the resulting fatty acid salts with hydrochloric acid. This closed-loop potential for reagent recovery further enhances the purity profile of the final product by preventing the introduction of external contaminants.
How to Synthesize 1-Hydroxy-3,5-Dimethyl-4-Chlorobenzene Efficiently
Implementing this synthesis route requires careful attention to the preparation of the catalytic mixture and the control of reaction parameters. The process begins with the charging of the reactor with the substrate, 1-hydroxy-3,5-dimethylbenzene, along with the designated amounts of divalent copper salt and lower fatty acid. A chlorine source, such as concentrated hydrochloric acid or a chloride salt, is then introduced into the mixture. The reaction vessel is equipped with standard monitoring tools including thermometers and pressure gauges to ensure safety and precision. Once the mixture is homogenized, the oxidant, typically oxygen or air, is introduced at a controlled rate while maintaining the temperature within the 40°C to 90°C range. The detailed standardized synthesis steps, including specific molar ratios, stirring speeds, and workup procedures, are outlined in the guide below.
- Prepare the catalytic system by mixing divalent copper salts with lower fatty acids in the reactor.
- Introduce 1-hydroxy-3,5-dimethylbenzene and a chlorine source such as hydrochloric acid or alkali metal chlorides.
- Conduct the oxychlorination reaction at 40-90°C under 1-5 atm pressure using oxygen or air as the oxidant.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this oxychlorination technology offers substantial strategic benefits beyond mere technical performance. The elimination of sulfur dioxide production removes the need for expensive scrubbing systems and hazardous waste disposal contracts, leading to significant operational cost savings. The use of commodity chemicals like hydrochloric acid and air as reagents ensures a stable and resilient supply chain, reducing dependency on specialized chlorinating agents that may be subject to market volatility. The mild reaction conditions also translate to lower energy requirements for heating and cooling, further enhancing the economic viability of the process. Additionally, the high selectivity reduces the loss of raw materials to by-products, maximizing the yield per batch and improving overall material efficiency. These factors collectively contribute to a more predictable and cost-effective manufacturing model.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the drastic simplification of the waste treatment infrastructure. By avoiding the generation of corrosive sulfur dioxide gas, manufacturers can eliminate the capital and operational expenditures associated with acid gas scrubbing and neutralization systems. Furthermore, the ability to recycle the copper catalyst and fatty acid co-catalyst multiple times without loss of activity significantly lowers the consumption of expensive reagents. The high atom economy ensures that a greater proportion of the raw material input is converted into the valuable final product, reducing the effective cost per kilogram of PCMX produced. This efficiency gain is compounded by the reduced need for complex purification steps, as the high selectivity minimizes the presence of difficult-to-remove isomers.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical concern for long-term production planning, and this method excels by utilizing widely available feedstocks. Hydrochloric acid, oxygen, and basic copper salts are commodity chemicals with robust global supply networks, minimizing the risk of shortages that can plague specialized reagent markets. The process flexibility allows for the use of air as an oxidant, which is essentially free and infinitely available, removing a potential bottleneck associated with liquid oxidant delivery. Moreover, the tolerance for various chloride sources, including alkali metal salts, provides procurement teams with the flexibility to switch suppliers based on price and availability without requalifying the entire process. This adaptability ensures continuous production capability even during regional supply disruptions.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new engineering challenges, but this oxychlorination route is designed for industrial robustness. The reaction operates at near-atmospheric pressures and moderate temperatures, reducing the mechanical stress on reactor vessels and simplifying the engineering requirements for large-scale plants. The absence of toxic sulfur emissions aligns perfectly with modern environmental regulations, facilitating easier permitting and reducing the risk of regulatory fines or shutdowns. The liquid-phase nature of the reaction allows for efficient heat transfer and mixing, which are crucial for maintaining safety and consistency in large batches. This combination of operational safety and environmental friendliness makes the technology highly attractive for expansion into new markets with strict ecological standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation. They are intended to clarify the operational parameters and benefits for potential partners and technical stakeholders. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production lines or new facility designs.
Q: How does this new method improve environmental compliance compared to traditional sulfuryl chloride routes?
A: The traditional method generates equimolar amounts of corrosive sulfur dioxide and hydrogen chloride, resulting in low atom economy. This new oxychlorination process eliminates sulfur dioxide emissions entirely, significantly reducing waste treatment costs and environmental impact.
Q: What is the selectivity advantage of the copper-fatty acid catalytic system?
A: The synergistic effect of the divalent copper salt and lower fatty acid co-catalyst ensures high para-selectivity. Experimental data indicates selectivity levels exceeding 96%, minimizing the formation of unwanted ortho-isomers and polychlorinated by-products.
Q: Can the catalyst and solvents be recycled in this industrial process?
A: Yes, the patent specifies that the divalent copper salt catalyst and the fatty acid co-catalyst can be recovered and reused repeatedly without negatively affecting reaction rates or selectivity, enhancing overall process economics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable PCMX Supplier
The technological potential of this oxychlorination route represents a significant opportunity for companies seeking to optimize their supply of high-purity disinfectant intermediates. NINGBO INNO PHARMCHEM stands ready to leverage this advanced chemistry as a CDMO partner, bringing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with stringent purity specifications and rigorous QC labs to ensure that every batch of PCMX meets the exacting standards required by the pharmaceutical and personal care industries. We understand the critical nature of supply continuity and quality consistency, and our technical team is dedicated to maintaining the highest levels of operational excellence.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific application. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this greener manufacturing method. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your volume requirements. Our goal is to provide a transparent and data-driven partnership that supports your long-term strategic objectives.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
