Revolutionizing Abiraterone Acetate Production: A Technical Analysis of Low-Cost Metal-Catalyzed Synthesis
Introduction to Next-Generation Abiraterone Acetate Manufacturing
The global demand for effective prostate cancer treatments continues to drive innovation in the synthesis of critical oncology intermediates. Abiraterone acetate, a potent CYP17 inhibitor, remains a cornerstone therapy for metastatic castration-resistant prostate cancer (mCRPC). However, traditional manufacturing routes have long been plagued by environmental hazards, complex purification steps, and suboptimal yields. The recent disclosure in patent CN114853838B introduces a transformative preparation method that addresses these historical bottlenecks through a robust metal-catalyzed cross-coupling strategy. This technical insight report analyzes the mechanistic breakthroughs and commercial implications of this novel pathway, providing R&D and procurement leaders with a clear roadmap for adopting more efficient supply chains.
This patented methodology shifts the paradigm from multi-step, hazardous sequences to a streamlined process centered on the coupling of a 3-position protected 17-hydroxy ester androstane derivative with 3-halogenated pyridine. By leveraging versatile catalytic systems involving palladium, nickel, copper, or iron, the invention achieves remarkable purity levels exceeding 99% while drastically simplifying the operational workflow. For stakeholders seeking a reliable API intermediate supplier, understanding the nuances of this technology is essential for securing long-term cost competitiveness and supply continuity in the competitive oncology market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of abiraterone acetate has relied on routes that are increasingly untenable in modern green chemistry frameworks. One prominent prior art method, described in WO 9509178, utilizes dehydroepiandrosterone (DHEA) as a starting material but requires a cumbersome sequence involving hydrazono formation with hydrazine hydrate, followed by iodination with elemental iodine under tetramethylguanidine catalysis. This approach not only suffers from an excessively long production cycle—requiring up to 5 days for the first step and 4 days for the third—but also generates a total yield of merely 36.9%. Furthermore, the reliance on malodorous and toxic reagents like hydrazine hydrate and iodine creates significant environmental pollution burdens and safety risks for plant personnel.
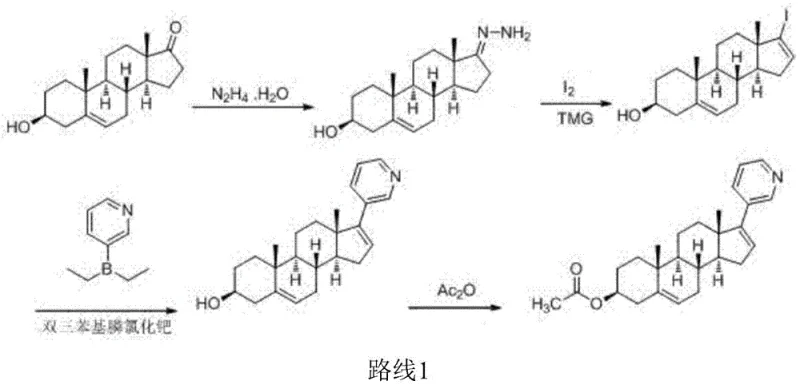
Another common pathway, reported in WO 2006021777, attempts to improve selectivity by converting DHEA acetate into a trifluoromethane sulfonyl derivative using trifluoromethanesulfonic anhydride. While this enables a Suzuki coupling with diethyl (3-pyridyl) borane, the process is economically inefficient due to the high cost of the triflating agent and the complex preparation of the borane nucleophile. The total yield for this route stagnates around 32.8%, and the subsequent salification step required for purification adds further complexity. These legacy methods illustrate a critical need for cost reduction in pharmaceutical intermediate manufacturing, as the cumulative effect of low yields and expensive reagents severely impacts the final cost of goods sold (COGS).
The Novel Approach
In stark contrast to these legacy processes, the method disclosed in CN114853838B offers a direct and highly efficient alternative. The core innovation lies in the direct coupling of a doubly protected androstane derivative (Formula II) with a simple 3-halogenated pyridine (Formula III). This reaction proceeds smoothly in the presence of a metal catalyst, a ligand, and a reducing agent, bypassing the need for unstable Grignard reagents or expensive triflates. The versatility of this approach allows for the use of inexpensive halides such as 3-bromopyridine or 3-chloropyridine, which are commodity chemicals with stable supply chains. By eliminating the need for pre-formed organoboron or organozinc species that require immediate use, the new method significantly enhances operational safety and scalability.
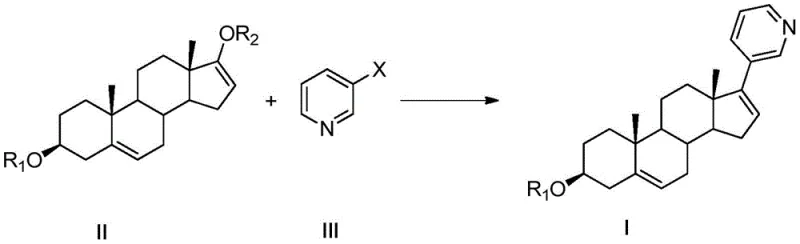
The structural flexibility of the new route is another major advantage. The protecting groups R1 and R2 can be independently selected from a broad range of acyl, sulfonyl, or silyl groups, allowing process chemists to tune the solubility and reactivity of the intermediate for optimal crystallization and purification. For instance, the use of acetyl groups for both positions leads directly to abiraterone acetate precursors, while other combinations allow for the isolation of stable derivatives that can be converted later. This modularity ensures that the process can be adapted to existing facility capabilities without requiring massive capital expenditure on new reactor types, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Metal-Catalyzed Reductive Coupling
The heart of this technological advancement is the metal-catalyzed reductive coupling mechanism, which facilitates the formation of the carbon-carbon bond between the steroid backbone and the pyridine ring. Unlike traditional Suzuki-Miyaura couplings that require pre-functionalized boronic acids, this method likely operates via an in-situ generation of the organometallic species or a direct oxidative addition/reductive elimination cycle facilitated by the reducing agent. The patent specifies a wide array of compatible catalysts, including palladium bistriphenylphosphine chloride, nickel acetate, cuprous iodide, and even ferric chloride. This breadth of compatibility suggests a radical-based or low-valent metal mechanism that is tolerant to various functional groups, thereby minimizing side reactions that typically lead to impurities.
Impurity control is rigorously addressed through the strategic use of protecting groups at the 3 and 17 positions. In the starting material (Formula IV), the 3-beta-hydroxyl and 17-hydroxyl groups are susceptible to oxidation or unwanted side reactions under harsh coupling conditions. By converting these to esters or silyl ethers (Formula II), the molecule is stabilized against degradation. The reaction conditions are equally optimized, with temperatures ranging from 20°C to 120°C and solvents like DMF, toluene, or acetonitrile ensuring homogeneous reaction mixtures. The inclusion of ligands such as L-proline, 2,2'-bipyridine, or triphenylphosphine further stabilizes the active catalytic species, preventing metal aggregation and ensuring consistent turnover numbers throughout the batch. This level of mechanistic control is what enables the reported HPLC purities of over 99% in the final recrystallized product.
How to Synthesize Abiraterone Acetate Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step guide for replicating these high-yield results in a pilot or production setting. The process begins with the dual protection of dehydroepiandrosterone, typically using isopropenyl acetate or acid chlorides to install the R1 and R2 groups. This is followed by the critical coupling step where the protected steroid reacts with the halopyridine in the presence of the chosen metal catalyst system. Detailed standard operating procedures regarding stoichiometry, addition rates, and workup protocols are essential for maintaining the high purity profiles described in the examples. For a comprehensive breakdown of the specific reaction parameters and workup techniques, please refer to the standardized synthesis guide below.
- Protect the 3-position and 17-position hydroxyl groups of dehydroepiandrosterone (DHEA) using acylating agents to form the di-ester intermediate (Formula II).
- React the protected intermediate with 3-halogenated pyridine in the presence of a metal catalyst (Pd, Ni, Cu, or Fe), a ligand, and a reducing agent to form the coupled product (Formula I).
- Perform hydrolysis to remove protecting groups if necessary, followed by acetylation to yield high-purity abiraterone acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route represents a strategic opportunity to optimize the cost structure and reliability of the abiraterone acetate supply chain. The shift away from hazardous and volatile reagents like hydrazine and iodine not only reduces the regulatory burden associated with waste disposal but also mitigates the risk of production stoppages due to safety incidents. Furthermore, the ability to use commodity halopyridines instead of custom-synthesized organoboron reagents drastically simplifies the raw material sourcing landscape, reducing dependency on niche suppliers and enhancing overall supply chain resilience.
- Cost Reduction in Manufacturing: The elimination of expensive reagents such as trifluoromethanesulfonic anhydride and diethyl (3-pyridyl) borane directly translates to substantial cost savings in raw material procurement. Additionally, the significant improvement in total yield—from roughly 36% in older methods to over 90% in key steps of this new process—means that less starting material is required to produce the same amount of API. This efficiency gain reduces the cost per kilogram of the final product, allowing for more competitive pricing strategies in the generic pharmaceutical market without compromising margin.
- Enhanced Supply Chain Reliability: By utilizing widely available metal catalysts and simple halogenated pyridines, the manufacturing process becomes less vulnerable to supply disruptions. The robustness of the reaction conditions, which tolerate a variety of solvents and catalysts, ensures that production can continue even if a specific grade of reagent is temporarily unavailable. This flexibility is crucial for maintaining continuous supply to downstream formulation partners, reducing lead time for high-purity pharmaceutical intermediates and ensuring that patient demand is met without interruption.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial mass production, featuring simple operation steps that do not require cryogenic conditions or ultra-high vacuum equipment. The reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, lowering the cost of compliance and waste treatment. The ability to scale from laboratory benchtop to multi-ton production without fundamental changes to the chemistry ensures a smooth technology transfer, enabling rapid capacity expansion to meet market growth.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and claims within CN114853838B, providing a factual basis for decision-making. Understanding these details helps bridge the gap between laboratory innovation and commercial reality, ensuring that all stakeholders have a clear view of the technology's capabilities and limitations.
Q: What are the primary advantages of this new synthesis method over conventional routes?
A: The new method eliminates the use of hazardous reagents like hydrazine hydrate and iodine, avoids expensive trifluoromethanesulfonic anhydride, and achieves significantly higher yields (over 90% in key steps) compared to the 36.9% total yield of older methods.
Q: Which metal catalysts are suitable for this coupling reaction?
A: The process is highly versatile, supporting a wide range of metal catalysts including palladium salts (e.g., PdCl2(PPh3)2), nickel salts, copper salts, and even iron or cobalt salts, allowing for cost optimization based on availability.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly highlights suitability for industrial mass production due to simple operation, mild reaction conditions (20-120°C), and the use of readily available organic solvents like DMF, toluene, and acetonitrile.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Abiraterone Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant manufacturing processes in the oncology sector. Our team of expert process chemists has extensively evaluated the methodology described in CN114853838B and possesses the technical capability to implement this advanced metal-catalyzed route at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of abiraterone acetate intermediate meets the highest global pharmacopeial standards.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to discuss specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability in the evolving landscape of prostate cancer therapeutics.
