Advancing Pharmaceutical Intermediates Production via Efficient Nickel-Catalyzed Cyclization Technology
Advancing Pharmaceutical Intermediates Production via Efficient Nickel-Catalyzed Cyclization Technology
The landscape of organic synthesis for complex nitrogen-containing heterocycles is undergoing a significant transformation, driven by the urgent need for more sustainable and cost-effective catalytic systems. A pivotal development in this arena is detailed in patent CN110467613B, which discloses a novel reaction method for the nickel-catalyzed intramolecular alkylation of imine cations. This technology specifically targets the synthesis of 3-substituted isoindolinones, a privileged scaffold found in numerous bioactive molecules such as pagoclone and pazinaclone. By shifting away from traditional precious metal catalysts, this innovation offers a robust pathway for generating high-purity pharmaceutical intermediates. The methodology leverages inexpensive divalent nickel compounds to activate hydroxyl groups, facilitating the formation of reactive iminium ions that undergo efficient cyclization. For R&D directors and procurement specialists alike, this represents a critical opportunity to optimize synthetic routes, reducing reliance on volatile precious metal markets while maintaining rigorous quality standards essential for drug substance manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 3-substituted isoindolinone frameworks via imine cation intermediates has relied heavily on sophisticated and often prohibitive catalytic systems. Literature precedents frequently cite the use of gold (Au) or silver (Ag) complexes, which, while effective, impose a substantial financial burden on large-scale operations due to the high cost of the metals and the ligands required to stabilize them. Additionally, alternative approaches developed by research groups such as Stephenson utilize ruthenium (Ru) catalyzed photoredox chemistry, which introduces significant complexity regarding reactor design, light source maintenance, and energy consumption. Beyond the economic constraints, many conventional protocols necessitate harsh reaction environments, including strong acidic or basic conditions, which can compromise the integrity of sensitive functional groups present in advanced intermediates. These aggressive conditions often lead to broader impurity profiles, necessitating extensive and costly purification steps that erode overall process efficiency and yield. Consequently, the industry has long sought a method that balances reactivity with operational simplicity and economic viability.
The Novel Approach
The methodology outlined in patent CN110467613B presents a paradigm shift by employing earth-abundant nickel catalysts to drive the intramolecular alkylation of lactams. This approach effectively generates the crucial imine cation intermediate in situ under remarkably mild thermal conditions, typically ranging from 70°C to 130°C. Unlike photoredox methods, this thermal process does not require specialized irradiation equipment, allowing it to be conducted in standard glass-lined or stainless steel reactors commonly found in multipurpose chemical plants. The tolerance for various functional groups is exceptional, accommodating electron-donating and electron-withdrawing substituents on the aromatic rings without significant loss in efficiency. By utilizing simple divalent nickel salts such as nickel perchlorate, nickel bromide, or nickel chloride, the process drastically lowers the raw material costs associated with catalysis. This accessibility, combined with the high yields reported (85% to 98%), positions this nickel-catalyzed route as a superior alternative for the commercial scale-up of complex pharmaceutical intermediates, directly addressing the pain points of both cost and operational complexity.
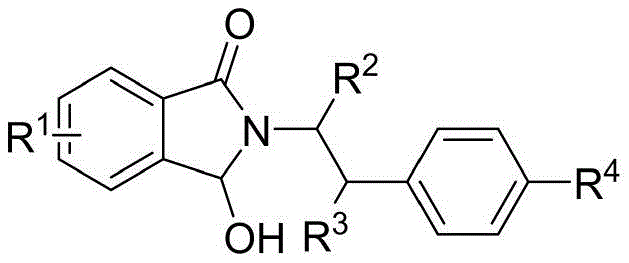
Mechanistic Insights into Nickel-Catalyzed Imine Cation Formation
At the heart of this transformative synthesis lies a fascinating mechanistic pathway involving the activation of the hydroxyl group by the nickel center. The reaction initiates with the coordination of the divalent nickel catalyst to the hydroxyl moiety of the 3-hydroxy-2-phenethyl-isoindolin-1-one substrate. This interaction weakens the carbon-oxygen bond, facilitating the departure of the hydroxyl group and the subsequent generation of a highly reactive iminium cation species. This electrophilic intermediate is then poised for an intramolecular nucleophilic attack by the pendant aromatic ring or alkene chain, depending on the specific substrate structure. The cyclization step forms the new carbon-carbon bond that defines the fused heterocyclic core of the product. Understanding this mechanism is vital for process chemists, as it highlights the dual role of the nickel catalyst not just as a Lewis acid, but as a promoter of leaving group ability under neutral to mild conditions. This mechanistic clarity allows for precise tuning of reaction parameters to maximize conversion while minimizing side reactions such as polymerization or decomposition of the sensitive cationic intermediate.
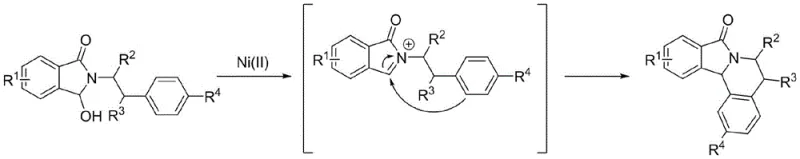
Furthermore, the control of impurities in this nickel-catalyzed system is inherently superior to acid-catalyzed alternatives. In traditional strong acid media, the high concentration of protons can lead to non-selective protonation of other basic sites within the molecule, potentially triggering rearrangement or degradation pathways. In contrast, the nickel-mediated generation of the iminium ion is more localized and controlled. The specific coordination geometry imposed by the nickel center helps direct the trajectory of the nucleophilic attack, favoring the desired cyclization over intermolecular oligomerization. This selectivity is crucial for maintaining a clean impurity profile, which is a non-negotiable requirement for regulatory compliance in pharmaceutical manufacturing. The ability to achieve high purity directly from the reaction crude, or with minimal chromatographic effort, underscores the robustness of this catalytic cycle. For quality assurance teams, this translates to a more predictable and stable manufacturing process, reducing the risk of batch failures due to trace impurities that are difficult to purge in later stages.
How to Synthesize 3-Substituted Isoindolinones Efficiently
The practical implementation of this nickel-catalyzed protocol is designed for straightforward execution in a standard laboratory or pilot plant setting. The general procedure involves dissolving the 3-hydroxy-2-phenethyl-isoindolin-1-one starting material in a polar aprotic or chlorinated solvent, with 1,2-dichloroethane (DCE) being the preferred medium for optimal solubility and reaction kinetics. Following dissolution, a stoichiometric amount of the divalent nickel catalyst is introduced, typically ranging from 5 mol% to 20 mol% depending on the electronic nature of the substrate. The mixture is then heated to the specified temperature window of 70-130°C and maintained under agitation for a duration of 7 to 12 hours. Upon completion, the workup is conventional, involving aqueous quenching, organic extraction, and standard drying procedures. The detailed standardized synthesis steps for specific derivatives are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Dissolve the 3-hydroxy-2-phenethyl-isoindolin-1-one substrate in a suitable organic solvent such as 1,2-dichloroethane.
- Add a divalent nickel catalyst, such as Ni(ClO4)2 or NiBr2, typically at a loading of 5-20 mol%.
- Heat the mixture to 70-130°C and stir for 7-12 hours to facilitate the cyclization reaction.
- Perform standard workup including extraction, washing, drying, and silica gel column chromatography to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nickel-catalyzed technology offers tangible strategic benefits that extend beyond mere chemical curiosity. The primary advantage lies in the drastic reduction of raw material costs associated with the catalyst itself. Replacing gold or ruthenium with nickel eliminates the exposure to the extreme price volatility characteristic of precious metal markets, allowing for more accurate long-term budget forecasting and cost stabilization. Moreover, the mild reaction conditions reduce the energy load on the facility, as there is no need for cryogenic cooling or high-energy UV irradiation sources. This energy efficiency contributes to a lower carbon footprint, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The simplicity of the reagent profile also simplifies inventory management, as nickel salts are stable, easy to handle, and widely available from multiple global suppliers, mitigating the risk of single-source supply disruptions.
- Cost Reduction in Manufacturing: The substitution of expensive precious metal catalysts with inexpensive nickel salts results in a significant decrease in the bill of materials for every kilogram of product produced. Since the catalyst loading is relatively low and the metal is abundant, the overall cost per mole of reaction is minimized. Additionally, the high yields reported (85-98%) mean that less starting material is wasted, further driving down the effective cost of goods sold (COGS). The elimination of complex purification steps required to remove trace precious metals from the final API also reduces downstream processing costs, making the entire value chain more economically efficient.
- Enhanced Supply Chain Reliability: Relying on nickel, a base metal with a robust global supply chain, insulates the manufacturing process from the geopolitical and logistical fragilities often associated with rare earth or precious metal sourcing. The reagents used, such as DCE or DMF, are commodity chemicals available in bulk quantities worldwide. This ubiquity ensures that production schedules can be maintained without interruption due to material shortages. Furthermore, the stability of the nickel catalysts allows for easier storage and handling compared to air- or moisture-sensitive organometallic complexes, reducing waste due to reagent degradation and ensuring consistent batch-to-batch performance.
- Scalability and Environmental Compliance: The thermal nature of this reaction makes it inherently scalable from gram to tonne quantities without the engineering challenges posed by photochemical flow reactors. Standard batch reactors can be utilized, facilitating a smoother technology transfer from R&D to commercial production. From an environmental perspective, the avoidance of strong acids and bases reduces the generation of hazardous saline waste streams, simplifying wastewater treatment protocols. The use of recyclable solvents and the potential for catalyst recovery further enhance the green chemistry profile of the process, supporting compliance with modern environmental, social, and governance (ESG) mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed synthesis. These insights are derived directly from the experimental data and claims within patent CN110467613B, providing a factual basis for evaluating the technology's fit within your existing portfolio. Understanding these nuances is essential for making informed decisions about process adoption and vendor selection. We encourage technical teams to review these details closely to appreciate the full scope of the method's capabilities and limitations.
Q: What are the primary advantages of using nickel catalysts over gold or ruthenium for this transformation?
A: Nickel catalysts are significantly more cost-effective and abundant compared to precious metals like gold (Au) or ruthenium (Ru). Furthermore, the nickel-catalyzed process described in patent CN110467613B operates under milder conditions without requiring strong acidic or basic environments, simplifying downstream processing and reducing equipment corrosion risks.
Q: What is the typical yield range for this nickel-catalyzed cyclization?
A: According to the experimental data provided in the patent, the reaction demonstrates high efficiency with isolated yields ranging from 85% to 98%, depending on the specific substituents on the aromatic rings and the choice of solvent.
Q: Is this method scalable for commercial production of pharmaceutical intermediates?
A: Yes, the method utilizes simple reagents and standard heating conditions (70-130°C) in common solvents like DCE or DMF. The absence of sensitive photoredox conditions or extremely low temperatures makes it highly amenable to scale-up for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Substituted Isoindolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the nickel-catalyzed imine cation alkylation method for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess the technical expertise to rapidly adapt this patented methodology to your specific molecular targets, ensuring a seamless transition from benchtop discovery to commercial supply. Our facilities are equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your supply needs are met with precision and consistency. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch meets the exacting standards required for drug substance manufacturing. Our commitment to quality ensures that the complex heterocyclic structures generated via this route are delivered with the highest level of chemical integrity.
We invite you to collaborate with us to leverage this cost-effective and robust synthetic route for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and target molecules. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this nickel-catalyzed technology can optimize your supply chain. Contact us today to discuss how we can support your R&D and commercial goals with reliable, high-quality chemical solutions.
