Optimizing Rosuvastatin Intermediate Production via Mild Catalytic Routes for Commercial Scale
Optimizing Rosuvastatin Intermediate Production via Mild Catalytic Routes for Commercial Scale
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective synthetic routes for high-value Active Pharmaceutical Ingredients (APIs), particularly for blockbuster drugs like Rosuvastatin calcium. Patent CN1958593B introduces a transformative methodology for preparing the critical intermediate methyl (R)-7-[4-(4-fluorophenyl)-6-isopropyl-2-(N-methyl-N-mesylamino)pyrimidin-5-yl]-3-tert-butyldimethylsiloxyl-5-carbonyl-6(E)-hepetnate. This innovation addresses longstanding bottlenecks in the supply chain by replacing hazardous, expensive reagents with commodity chemicals while maintaining high stereochemical integrity. For R&D directors and procurement managers, understanding this shift from cryogenic, metal-heavy processes to mild, ambient-condition reactions is vital for strategic sourcing and process optimization. The following analysis dissects the technical merits and commercial implications of this patented technology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art, specifically exemplified by US Patent 5,260,440, relies on a synthetic pathway fraught with industrial inefficiencies and safety hazards. The initial reduction step utilizes diisobutylaluminum hydride (DIBAL-H), a reagent that is not only expensive and difficult to source in bulk but also requires stringent cryogenic conditions at -74°C to prevent side reactions. Such extreme低温 operations demand specialized equipment and high energy consumption, drastically inflating the cost of goods sold (COGS). Furthermore, the subsequent oxidation step employs tetrapropylammonium perruthenate (TPAP) and N-methyloxymorpholine (NMO), which are imported specialty reagents with limited global availability. The final condensation step in the conventional route is equally problematic, requiring prolonged heating at 80°C for 14 hours, often resulting in incomplete conversion that necessitates resource-intensive column chromatography for purification.
The Novel Approach
The methodology disclosed in CN1958593B fundamentally re-engineers this sequence to align with Green Chemistry principles and industrial pragmatism. In the reduction phase, the patent substitutes DIBAL-H with a borohydride/chloride system, such as potassium borohydride and zinc chloride. This switch allows the reaction to proceed at significantly higher temperatures, ranging from 60°C to 150°C, eliminating the need for cryogenic cooling while achieving comparable yields of 91.8%. 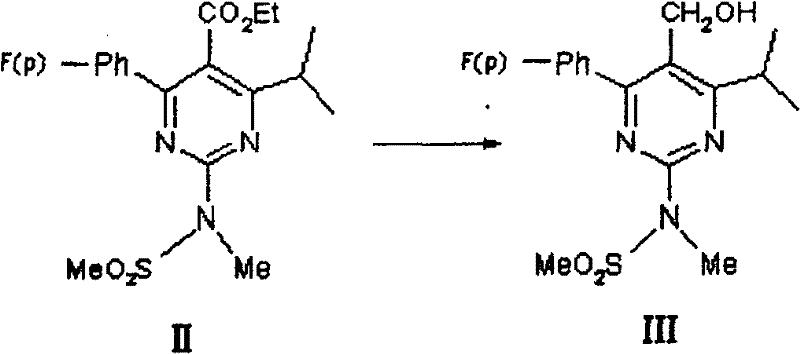 The oxidation step is similarly optimized by utilizing potassium dichromate, a ubiquitous and inexpensive oxidant, which boosts the yield to an impressive 93.2% compared to the 71.3% of the prior art. Finally, the condensation reaction is conducted under mild basic conditions at room temperature, reducing reaction time from 14 hours to merely 0.5-3 hours. This holistic approach not only simplifies the operational workflow but also ensures that byproducts are water-soluble, facilitating easy purification without chromatography.
The oxidation step is similarly optimized by utilizing potassium dichromate, a ubiquitous and inexpensive oxidant, which boosts the yield to an impressive 93.2% compared to the 71.3% of the prior art. Finally, the condensation reaction is conducted under mild basic conditions at room temperature, reducing reaction time from 14 hours to merely 0.5-3 hours. This holistic approach not only simplifies the operational workflow but also ensures that byproducts are water-soluble, facilitating easy purification without chromatography.
Mechanistic Insights into Borohydride-Mediated Reduction and HWE Condensation
The core innovation lies in the activation of the borohydride species by Lewis acidic chlorides. In the reduction of the pyrimidine carbonate ester (II) to the alcohol (III), the interaction between the borohydride anion and the metal chloride generates a more reactive hydride species in situ. This species mimics the selectivity of DIBAL-H but operates effectively at elevated temperatures, likely due to the stabilization of the transition state by the pyrimidine nitrogen atoms. The reaction mechanism avoids the over-reduction or decomposition often seen with stronger reducing agents at high temperatures, ensuring the integrity of the sensitive fluorophenyl and isopropyl substituents. This mechanistic robustness is crucial for maintaining high purity profiles required for downstream API synthesis.
Following oxidation to the aldehyde (IV), the process employs a Horner-Wadsworth-Emmons (HWE) type condensation with the chiral phosphonate (V). 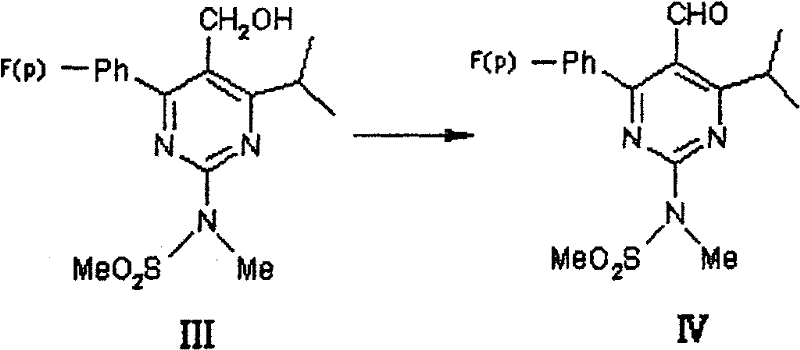 The use of bases like DBU or sodium hydride facilitates the deprotonation of the phosphonate to form a stabilized carbanion, which then attacks the aldehyde carbonyl. The patent specifies that this reaction proceeds efficiently at 0-100°C, with optimal results at room temperature. The stereoselectivity is inherent to the chiral phosphonate reagent, ensuring the formation of the desired (E)-alkene geometry and the (R)-configuration at the hydroxyl-bearing carbon.
The use of bases like DBU or sodium hydride facilitates the deprotonation of the phosphonate to form a stabilized carbanion, which then attacks the aldehyde carbonyl. The patent specifies that this reaction proceeds efficiently at 0-100°C, with optimal results at room temperature. The stereoselectivity is inherent to the chiral phosphonate reagent, ensuring the formation of the desired (E)-alkene geometry and the (R)-configuration at the hydroxyl-bearing carbon. 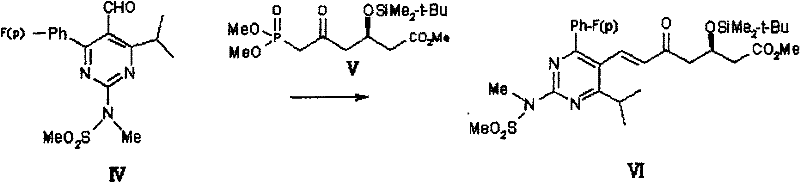 The elimination of the phosphonate byproduct into the aqueous phase during workup is a key mechanistic feature that simplifies isolation, as the organic product remains in the organic layer while polar impurities are washed away.
The elimination of the phosphonate byproduct into the aqueous phase during workup is a key mechanistic feature that simplifies isolation, as the organic product remains in the organic layer while polar impurities are washed away.
How to Synthesize Rosuvastatin Intermediate Efficiently
Implementing this synthesis route requires precise control over stoichiometry and temperature profiles to maximize the benefits outlined in the patent. The process begins with the preparation of the alcohol intermediate, followed by oxidation and final coupling. Each step has been optimized to minimize waste and maximize throughput, making it an ideal candidate for contract development and manufacturing organizations (CDMOs) looking to offer cost-effective solutions. The detailed standardized synthesis steps below outline the critical parameters for successful execution.
- Reduce pyrimidine carbonate (II) using borohydride/chloride system at 60-150°C to obtain pyrimidine methanol (III).
- Oxidize pyrimidine methanol (III) using potassium dichromate and sulfuric acid at 0-35°C to yield pyrimidine formaldehyde (IV).
- Condense aldehyde (IV) with chiral phosphonate (V) under basic conditions at room temperature to form the final intermediate (VI).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to the methodology described in CN1958593B represents a significant opportunity for cost reduction in pharmaceutical intermediate manufacturing. The primary driver of value is the substitution of exotic, import-dependent reagents with globally available commodity chemicals. By eliminating the reliance on DIBAL-H and TPAP, manufacturers can insulate their supply chains from geopolitical volatility and price fluctuations associated with specialty reagents. Additionally, the removal of cryogenic requirements reduces capital expenditure on specialized cooling infrastructure and lowers ongoing energy costs, contributing to a leaner operational model.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, driven largely by the drastic simplification of the purification workflow. In the conventional route, the need for column chromatography to purify the final condensation product creates a bottleneck, consuming vast amounts of silica gel and solvents while limiting batch size. The new method generates water-soluble byproducts, allowing for purification via simple aqueous extraction. This shift eliminates the cost of chromatography media and significantly reduces solvent consumption, leading to substantial cost savings per kilogram of produced intermediate without compromising quality.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical metric for long-term API production. The reagents specified in this patent, such as potassium borohydride, zinc chloride, and potassium dichromate, are produced at massive scales for various industries, ensuring consistent availability and competitive pricing. Unlike the prior art reagents which may have single-source suppliers or long lead times, these commodity chemicals can be sourced from multiple vendors globally. This diversification mitigates the risk of supply disruptions, ensuring continuous production schedules and reliable delivery timelines for downstream API manufacturers.
- Scalability and Environmental Compliance: From a scale-up perspective, the mild reaction conditions (0-150°C) and the absence of pyrophoric reagents make this process inherently safer and easier to scale from pilot plant to commercial tonnage. The reduction in hazardous waste generation, particularly the avoidance of aluminum waste from DIBAL-H quenching and heavy metal ruthenium residues, simplifies wastewater treatment protocols. This alignment with environmental regulations reduces the burden on EHS (Environment, Health, and Safety) departments and lowers the costs associated with waste disposal, further enhancing the overall sustainability profile of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on yield expectations, safety profiles, and purification strategies. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this technology for their specific production needs.
Q: What are the key advantages of the borohydride/chloride reduction method over DIBAL-H?
A: The borohydride/chloride system utilizes commercially available, low-cost reagents compared to the expensive and difficult-to-source diisobutylaluminum hydride (DIBAL-H). Furthermore, it eliminates the need for harsh cryogenic conditions (-74°C), operating instead at moderate temperatures (60-150°C), which significantly enhances operational safety and energy efficiency.
Q: How does the new oxidation method improve yield compared to prior art?
A: By replacing the complex tetrapropylammonium perruthenate/N-methyloxymorpholine system with potassium dichromate, the process achieves a substantial yield increase from 71.3% to 93.2%. This improvement is critical for maximizing throughput in large-scale API manufacturing.
Q: Is column chromatography required for purifying the final condensation product?
A: No. A major advantage of this patented method is that the byproducts generated during the condensation step are water-soluble. This allows for purification via simple aqueous washing, completely removing the need for costly and time-consuming column chromatography, thereby streamlining the production workflow.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rosuvastatin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, scalable synthetic routes in the competitive landscape of statin production. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of patents like CN1958593B are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required for GMP API synthesis. Our commitment to process excellence allows us to deliver high-purity pharmaceutical intermediates with consistent quality and reliability.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By leveraging our expertise, you can access a Customized Cost-Saving Analysis tailored to your volume needs. We encourage you to request specific COA data and route feasibility assessments to validate the performance of this method against your current benchmarks. Together, we can drive efficiency and value in the production of next-generation cardiovascular therapeutics.
