Scalable Manufacturing of Vilazodone Intermediates via Optimized Piperazine Cyclization and Cyanation
Introduction to Advanced Vilazodone Manufacturing
The global pharmaceutical landscape continues to demand robust, scalable, and cost-effective synthesis routes for next-generation antidepressants, particularly for complex molecules like Vilazodone (EMD 68843). As a dual-function agent combining serotonin reuptake inhibition (SSRI) and 5-HT1A receptor antagonism, Vilazodone represents a significant therapeutic advancement, yet its commercial viability hinges entirely on the efficiency of its chemical manufacturing process. The patent CN102180868B introduces a transformative methodology that addresses the critical bottlenecks of prior art, specifically targeting the low yields and purification challenges associated with earlier syntheses. This technical insight report analyzes the proprietary pathway detailed in the patent, highlighting its potential to redefine supply chain reliability for this high-value active pharmaceutical ingredient.
The core innovation lies in a streamlined seven-step sequence originating from ethyl 5-nitrobenzofuran-2-carboxylate, a readily accessible starting material. Unlike previous methods that relied heavily on labor-intensive purification techniques such as column chromatography, this novel approach leverages standard unit operations like crystallization and liquid-liquid extraction to achieve high purity. The structural complexity of Vilazodone, featuring a benzofuran moiety linked via a piperazine-butyl chain to a cyano-indole system, requires precise control over regioselectivity and functional group tolerance. 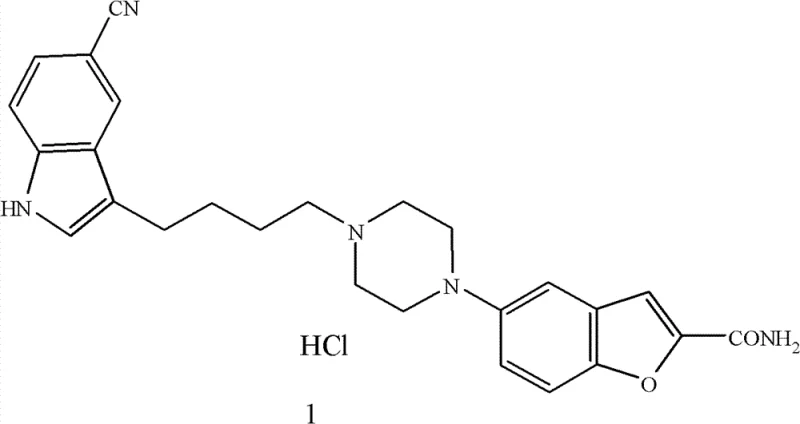
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations described in CN102180868B, the synthesis of Vilazodone, particularly the route reported by Merck & Co., Inc., faced severe scalability hurdles that rendered it economically unfeasible for mass production. The historical methodology suffered from an abysmal total recovery rate of merely 3.4%, a figure that is practically unacceptable for commercial API manufacturing. A primary driver of this inefficiency was the reliance on column chromatography for purification at two distinct stages of the synthesis. In an industrial setting, column chromatography is notoriously difficult to scale, requiring vast quantities of silica gel and solvents, which drastically inflates both operational expenditure and environmental waste generation. Furthermore, the synthesis of key intermediates, such as 5-cyano-3-(4-chlorobutyl)indole, exhibited very low yields, creating a compounding effect that diminished the overall process throughput. These technical deficiencies not only increased the cost of goods sold (COGS) but also introduced significant variability in batch-to-batch consistency, posing risks to supply chain continuity.
The Novel Approach
The patented process fundamentally re-engineers the synthetic strategy to overcome these impediments by prioritizing reactions that yield crystalline products amenable to simple filtration and recrystallization. By initiating the synthesis with the nitro reduction of ethyl 5-nitrobenzofuran-2-carboxylate using Raney Nickel, the process establishes a high-yielding foundation that avoids the use of expensive noble metal catalysts often found in alternative hydrogenation methods. The subsequent construction of the piperazine ring via cyclization with 4-aminobutyric acid (GABA) is executed in a manner that minimizes side reactions, ensuring a clean profile for downstream processing. Crucially, the introduction of microwave-assisted cyanation represents a significant technological leap, accelerating reaction kinetics and improving conversion rates without the need for prolonged heating that could degrade sensitive functional groups. This holistic optimization results in a process that is not only chemically superior but also operationally streamlined, eliminating the need for chromatographic purification entirely and thereby unlocking true industrial applicability.
Mechanistic Insights into Piperazine Cyclization and Microwave Cyanation
The chemical elegance of this synthesis is best observed in the construction of the central piperazine scaffold and the final installation of the nitrile group. The formation of the piperazine ring involves a nucleophilic substitution followed by an intramolecular cyclization. Initially, the amino-benzofuran intermediate reacts with chlorohydrin under alkaline conditions to form a bis-hydroxyethyl amine species, which is subsequently converted to a bis-chloroethyl derivative using phosphorus trichloride. This activated species then undergoes cyclization with 4-aminobutyric acid in the presence of a base such as diethyl isopropylamine. The mechanism proceeds through the nucleophilic attack of the amine nitrogen on the terminal chlorides, closing the six-membered piperazine ring. The use of thionyl chloride in the subsequent step converts the carboxylic acid tail of the GABA moiety into an acyl chloride, activating it for the critical Friedel-Crafts acylation with the indole nucleus. This sequence demonstrates remarkable chemoselectivity, preserving the integrity of the benzofuran ester while enabling robust bond formation.
Furthermore, the cyanation step utilizes microwave irradiation to drive the nucleophilic aromatic substitution or displacement reaction involving sodium cyanide. In conventional thermal conditions, cyanation reactions can be sluggish and prone to hydrolysis of the nitrile product or degradation of the substrate. However, the application of microwave energy in N-Methyl pyrrolidone (NMP) provides rapid, uniform heating that significantly lowers the activation energy barrier. This allows the reaction to reach completion within 30 to 60 minutes, a timeframe that is substantially shorter than traditional reflux methods. The mechanism likely involves the generation of free cyanide ions which attack the halogenated indole precursor (specifically the 5-bromo position), displacing the halide leaving group. The efficiency of this step is paramount, as it installs the crucial cyano pharmacophore required for the drug's biological activity. 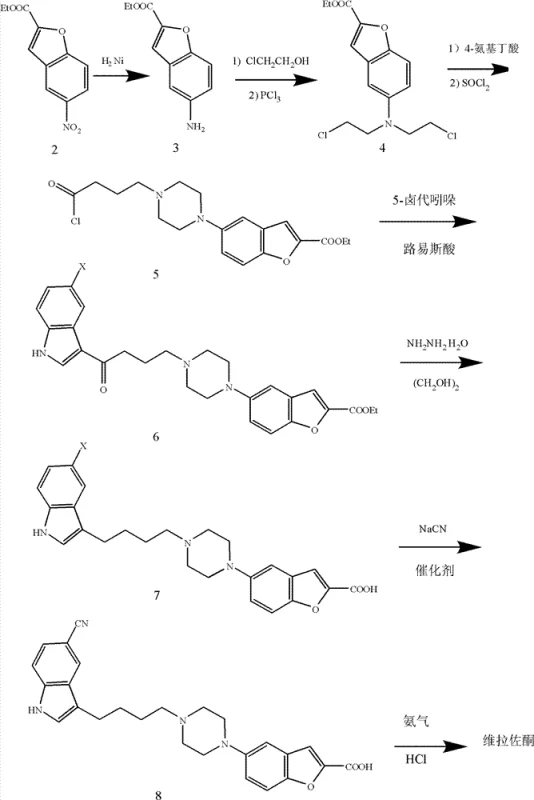
How to Synthesize Vilazodone Efficiently
The execution of this synthesis requires strict adherence to the optimized reaction parameters defined in the patent to ensure maximum yield and purity. The process begins with the catalytic reduction of the nitro group, followed by the sequential assembly of the piperazine linker and the indole coupling. Each step has been refined to utilize common industrial solvents like DMF, ethyl acetate, and dichloroethane, facilitating easy solvent recovery and recycling. The following guide outlines the critical operational phases necessary to replicate this high-efficiency pathway in a pilot or production environment.
- Perform catalytic hydrogenation of ethyl 5-nitrobenzofuran-2-carboxylate using Raney Nickel to generate the amino ester intermediate.
- Execute alkylation with chlorohydrin followed by phosphorus trichloride chlorination to form the bis-chloroethyl amine derivative.
- Cyclize with 4-aminobutyric acid (GABA) and thionyl chloride to construct the piperazine ring, followed by Friedel-Crafts acylation with 5-haloindole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from the legacy Merck process to this optimized route offers substantial strategic benefits that extend beyond simple yield improvements. The elimination of column chromatography is perhaps the most significant economic driver, as it removes a unit operation that is both capital intensive and slow. By replacing chromatography with crystallization and extraction, the manufacturing cycle time is drastically reduced, allowing for faster batch turnover and improved responsiveness to market demand fluctuations. Additionally, the use of readily available starting materials like ethyl 5-nitrobenzofuran-2-carboxylate mitigates the risk of raw material shortages, ensuring a more resilient supply chain that is less dependent on custom-synthesized precursors with long lead times.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the removal of expensive purification steps and the optimization of reagent usage. By avoiding column chromatography, the consumption of silica gel and large volumes of elution solvents is completely eradicated, leading to a direct reduction in variable manufacturing costs. Furthermore, the high yields achieved in key steps, such as the 93% yield in the initial nitro reduction and the 84.7% yield in the acylation step, mean that less raw material is wasted per kilogram of final API produced. This material efficiency translates directly into a lower cost of goods sold, providing a competitive pricing advantage in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route contributes significantly to supply security. The reliance on standard chemical transformations and commercially available reagents reduces the complexity of the supply network. Unlike processes that require exotic catalysts or unstable intermediates, this method utilizes stable compounds that can be sourced from multiple vendors, reducing single-source dependency risks. The simplified work-up procedures also mean that production schedules are more predictable, as there are fewer variables that could cause batch failures or delays, thereby ensuring consistent on-time delivery to downstream formulation partners.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process aligns well with green chemistry principles. The reduction in solvent usage, particularly the avoidance of large-scale chromatographic solvents, lowers the facility's overall volatile organic compound (VOC) emissions. The ability to scale the microwave cyanation step effectively means that production capacity can be expanded without a proportional increase in waste generation. This scalability is essential for meeting the growing global demand for antidepressants while maintaining compliance with increasingly stringent environmental regulations regarding chemical waste disposal and solvent recovery.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on the feasibility and advantages of the technology.
Q: How does this process improve upon the original Merck synthesis route for Vilazodone?
A: The patented process eliminates the need for column chromatography purification, which was a major bottleneck in the original Merck route that resulted in a total yield of only 3.4%. By utilizing crystallization and standard extraction techniques, this method achieves significantly higher yields suitable for industrial scale-up.
Q: What are the critical reaction conditions for the cyanation step?
A: The cyanation step utilizes sodium cyanide in N-Methyl pyrrolidone (NMP) under microwave irradiation. This specific condition accelerates the substitution reaction, reducing reaction time to 30-60 minutes while maintaining high conversion efficiency compared to conventional heating methods.
Q: Is the starting material ethyl 5-nitrobenzofuran-2-carboxylate commercially viable?
A: Yes, the patent explicitly states that all supplementary materials used, including the starting nitro-benzofuran derivative, are readily available commercially. This ensures a stable supply chain foundation without the need for complex custom synthesis of precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vilazodone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert process chemists has thoroughly evaluated the pathway described in CN102180868B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering high-purity Vilazodone intermediates and API that meet stringent purity specifications, utilizing our rigorous QC labs to ensure every batch conforms to the highest international standards. Our infrastructure is designed to handle complex heterocyclic chemistry safely and efficiently, making us an ideal partner for your long-term supply needs.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By leveraging our manufacturing capabilities, you can access a Customized Cost-Saving Analysis tailored to your volume needs. We encourage potential partners to contact us directly to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on transparent, data-driven insights into our production capabilities and quality assurance protocols.
