Advanced Synthesis of Favipiravir Intermediates: Technical Breakthroughs and Commercial Scalability
Advanced Synthesis of Favipiravir Intermediates: Technical Breakthroughs and Commercial Scalability
The global demand for effective antiviral therapeutics has placed intense scrutiny on the manufacturing efficiency of key active pharmaceutical ingredients like Favipiravir. Patent CN111471025A introduces a transformative synthesis methodology that addresses critical bottlenecks in the production of this vital RNA polymerase inhibitor. This technical disclosure outlines a robust pathway starting from readily available 2,5-dihalo pyrazines, utilizing a novel radical oxidation strategy to construct the pyrazine core functionality. Unlike legacy processes that rely on hazardous nitration or excessively long linear sequences, this approach leverages a catalytic system involving persulfates and iron salts to achieve high conversion rates under relatively mild thermal conditions. For R&D directors and process chemists, this represents a significant leap forward in designing safer, more sustainable manufacturing protocols that align with modern green chemistry principles while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Favipiravir has been plagued by severe safety hazards and operational inefficiencies that hinder scalable production. One prominent legacy route, often cited in earlier literature, relies heavily on nitration reactions to introduce necessary functional groups onto the pyrazine ring. As illustrated in the reaction scheme below, this pathway involves treating hydroxy-amide precursors with nitrating agents, a process inherently fraught with the risk of thermal runaway and potential explosion. Consequently, facilities adopting this method must invest heavily in specialized explosion-proof equipment and rigorous safety containment systems, drastically inflating capital expenditure. Furthermore, alternative routes reported in patents such as WO2010/087117 involve convoluted seven-step sequences starting from aminomalonate derivatives. These lengthy syntheses suffer from cumulative yield losses at every stage, generate excessive volumes of wastewater, and require complex purification protocols that erode profit margins and complicate supply chain logistics for bulk manufacturers.
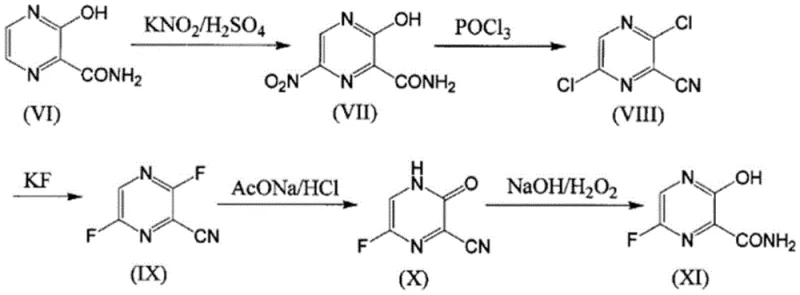
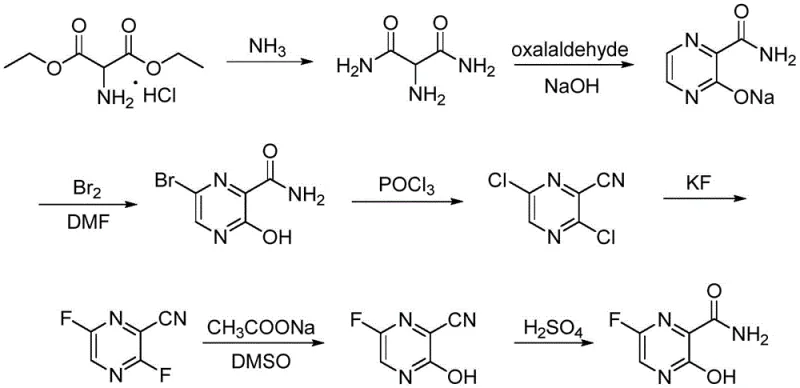
The Novel Approach
In stark contrast to these cumbersome legacy methods, the methodology disclosed in CN111471025A offers a streamlined, high-efficiency alternative that fundamentally reimagines the construction of the Favipiravir scaffold. The new process initiates with a direct radical amidation of 2,5-dihalo pyrazines, bypassing the need for dangerous nitration entirely. By employing a catalytic system of potassium persulfate and ferrous sulfide in an aqueous medium, the reaction achieves excellent selectivity for the desired amide intermediate. This is followed by a dehydration-chlorination step to install the cyano group, and finally, a nucleophilic aromatic substitution to introduce the critical fluorine atom. The entire sequence is remarkably concise, reducing the step count significantly compared to prior art. Moreover, the process exhibits exceptional tolerance for raw material variations, allowing the use of mixed chloro-bromo pyrazine feedstocks without sacrificing yield, which provides a strategic advantage in sourcing and cost management for large-scale production facilities.
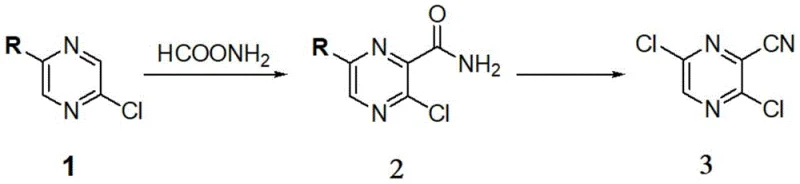
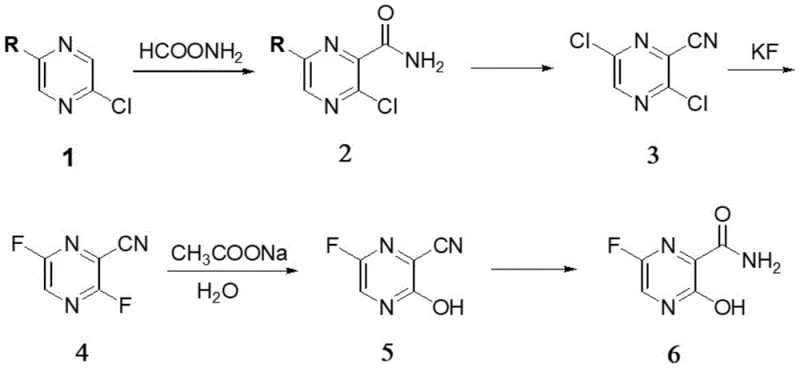
Mechanistic Insights into Radical Oxidation and Fluorination
The core innovation of this synthesis lies in the initial radical-mediated transformation of the dihalo pyrazine substrate. Under the influence of the persulfate oxidant and the iron catalyst, formamide undergoes a radical generation process that facilitates the direct attachment of the amide moiety to the electron-deficient pyrazine ring. This mechanism avoids the harsh acidic conditions typical of electrophilic aromatic substitution, thereby preserving the integrity of the halogen substituents required for subsequent fluorination. The choice of ferrous sulfide as a catalyst is particularly judicious, as it effectively mediates the single-electron transfer processes necessary for radical propagation while remaining cost-effective and easy to handle on an industrial scale. Following the formation of the amide, the subsequent conversion to the nitrile via phosphorus oxychloride proceeds through a standard dehydration mechanism, but with a critical modification in the base selection to manage impurity profiles effectively.
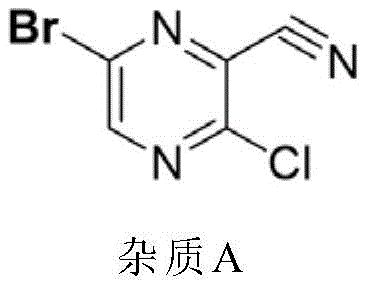
Impurity control is a paramount concern in the synthesis of pharmaceutical intermediates, and this patent provides deep insights into managing side reactions during the chlorination-dehydration phase. Specifically, the formation of a structurally related byproduct, designated as Impurity A, is shown to be highly dependent on the stoichiometry of the acid-binding agent, diisopropylethylamine. As detailed in the structural analysis of the byproduct, insufficient base leads to incomplete scavenging of HCl or promotes alternative elimination pathways that generate this persistent contaminant. By optimizing the molar ratio of the base to the substrate, manufacturers can suppress the formation of Impurity A to negligible levels, ensuring that the downstream fluorination step proceeds with high fidelity. The final fluorination step utilizes potassium fluoride in dimethyl sulfoxide, leveraging the high nucleophilicity of the fluoride ion in a polar aprotic solvent to displace the chlorine atom selectively, completing the assembly of the pharmacophore.
How to Synthesize Favipiravir Intermediates Efficiently
The practical implementation of this synthesis route requires careful attention to reaction parameters to maximize yield and purity. The process begins with the suspension of the dihalo pyrazine starting material in water, followed by the sequential addition of the oxidant and catalyst. Temperature control is critical during the exothermic addition of formamide, necessitating a gradual heating profile to maintain reaction stability. Once the amide intermediate is isolated, it is subjected to dehydration conditions using phosphorus oxychloride, where the precise dosing of the organic base determines the cleanliness of the crude product. The subsequent fluorination in DMSO requires elevated temperatures to overcome the activation energy for nucleophilic aromatic substitution, followed by a carefully controlled hydrolysis to reveal the final carboxamide functionality. For a comprehensive, step-by-step guide including specific reagent quantities and workup procedures, please refer to the standardized protocol outlined below.
- React 2,5-dihalo pyrazine with formamide using potassium persulfate and ferrous sulfide catalyst to generate the amide intermediate.
- Perform dehydration chlorination on the amide using phosphorus oxychloride and diisopropylethylamine to form the cyano-pyrazine derivative.
- Execute aromatic ring fluorination with potassium fluoride in DMSO, followed by hydrolysis to yield the final Favipiravir product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers compelling advantages for procurement managers and supply chain executives seeking to optimize their API sourcing strategies. The most significant benefit arises from the process's ability to utilize a mixture of 2,5-dichloropyrazine and 2-chloro-5-bromopyrazine as the starting material. In traditional synthesis, high-purity single-component halides are often required, commanding premium prices and subjecting buyers to volatile market fluctuations. By contrast, this technology tolerates mixed feedstocks, allowing manufacturers to source lower-cost, commodity-grade raw materials without compromising the quality of the final intermediate. This flexibility translates directly into substantial raw material cost savings, providing a buffer against supply chain disruptions and price spikes in the global halide market.
- Cost Reduction in Manufacturing: The elimination of hazardous nitration steps removes the necessity for expensive explosion-proof infrastructure, significantly lowering the capital barrier for production facilities. Furthermore, the shortened reaction sequence reduces the consumption of solvents, reagents, and energy per kilogram of product. The high overall yield reported in the patent minimizes waste generation and maximizes the throughput of existing reactor trains, driving down the unit cost of goods sold and enhancing the overall economic viability of the manufacturing process.
- Enhanced Supply Chain Reliability: By relying on widely available and stable starting materials like dihalo pyrazines and formamide, the supply chain becomes more resilient to shortages of exotic or highly regulated precursors. The robustness of the radical oxidation step ensures consistent batch-to-batch performance, reducing the risk of production delays caused by failed reactions or off-spec material. This reliability is crucial for maintaining continuous supply to downstream drug formulation units, especially during periods of heightened global demand for antiviral medications.
- Scalability and Environmental Compliance: The use of water as the primary solvent for the initial oxidation step aligns with green chemistry initiatives, reducing the environmental footprint associated with volatile organic compound emissions. The simplified workup procedures, involving straightforward filtration and recrystallization, facilitate easier scale-up from pilot plant to commercial tonnage production. Additionally, the reduced generation of hazardous waste streams simplifies effluent treatment requirements, ensuring compliance with increasingly stringent environmental regulations across different manufacturing jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Favipiravir synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, aiming to clarify the operational benefits and mechanistic nuances for potential licensees and manufacturing partners. Understanding these details is essential for evaluating the feasibility of integrating this route into existing production portfolios.
Q: How does the new synthesis route improve safety compared to traditional nitration methods?
A: The novel route eliminates the hazardous nitration step found in older methodologies, which carried significant explosion risks. By utilizing a radical oxidation mechanism with persulfates instead, the process operates under much milder and safer industrial conditions, removing the need for specialized explosion-proof infrastructure.
Q: What is the impact of using mixed halide raw materials on production costs?
A: The process demonstrates remarkable flexibility by accepting a mixture of 2,5-dichloropyrazine and 2-chloro-5-bromopyrazine as starting materials. This tolerance allows manufacturers to utilize lower-cost, less purified feedstocks without compromising the final yield or purity, leading to substantial raw material cost optimization.
Q: How is impurity control managed during the chlorination step?
A: Impurity formation, specifically Impurity A, is tightly controlled by optimizing the molar ratio of the acid-binding agent diisopropylethylamine. Maintaining a specific stoichiometric excess ensures that side reactions are suppressed, resulting in a cleaner crude product that simplifies downstream purification and recrystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Favipiravir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying efficient and safe synthesis routes for high-value antiviral intermediates. Our team of expert process chemists has extensively evaluated the methodology described in CN111471025A and possesses the technical capability to execute this radical oxidation pathway with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and robust. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Favipiravir intermediate we deliver meets the highest international standards for pharmaceutical grade materials.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced technology for your supply chain needs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and regional logistics. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can drive value and security into your antiviral drug production pipeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
