Advanced Microchannel Fluorination Process for Commercial Scale-up of Favipiravir Intermediates
Introduction: Revolutionizing Antiviral Intermediate Manufacturing
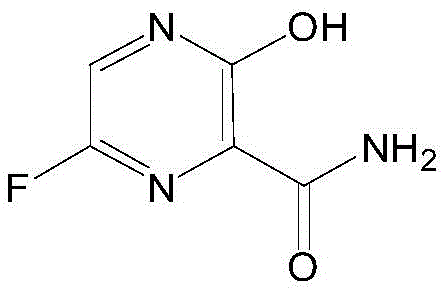
The global pharmaceutical landscape has witnessed an unprecedented surge in demand for broad-spectrum antiviral agents, with Favipiravir standing out as a critical RNA polymerase inhibitor. However, the commercial viability of such life-saving medications often hinges on the efficiency and safety of their synthetic routes. Patent CN111349049A introduces a transformative methodology for synthesizing Favipiravir and its key intermediates, specifically addressing the bottlenecks of traditional batch processing. This innovative approach leverages continuous flow chemistry and microchannel technology to achieve high-selectivity fluorination, a step that has historically plagued manufacturers with safety hazards and low yields. By shifting from hazardous nitration and expensive palladium-catalyzed couplings to a streamlined cyclization and fluorination sequence, this technology offers a robust pathway for industrial production.
For R&D directors and supply chain strategists, the implications of this patent extend far beyond mere chemical curiosity. It represents a paradigm shift towards safer, more cost-effective manufacturing of high-purity pharmaceutical intermediates. The process utilizes readily available bulk chemicals like 2-aminomalonic diamide and glyoxal, bypassing the need for complex, multi-step precursor synthesis. Furthermore, the integration of silicon carbide microchannel reactors allows for the safe handling of elemental fluorine, a reagent known for its extreme reactivity, thereby unlocking a direct and efficient route to the fluorinated pyrazine core. This report analyzes the technical depth and commercial potential of this novel synthesis route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Favipiravir has been fraught with significant technical and economic challenges that hinder large-scale commercialization. Early routes, such as those described in patent WO0010569, relied heavily on palladium-catalyzed amino substitution reactions. These methods necessitate the use of expensive catalysts like tris(dibenzylideneacetone)dipalladium and chiral ligands such as BINAP, which not only inflate raw material costs but also introduce complex purification steps to remove trace heavy metals to meet pharmaceutical standards. Additionally, alternative pathways like those in WO0160834 involve nitration reactions that pose severe explosion risks, requiring specialized explosion-proof equipment and rigorous safety protocols that drive up capital expenditure.
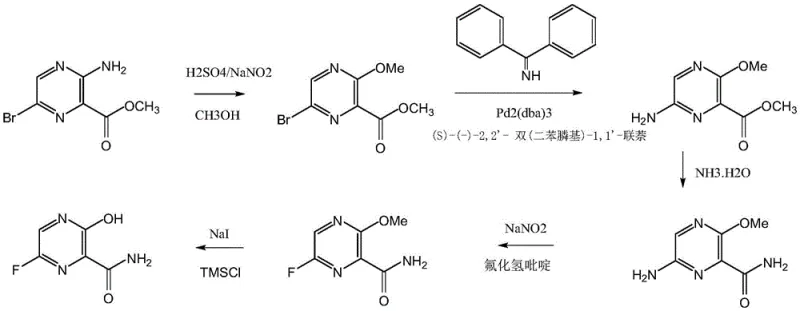
Furthermore, other existing methodologies suffer from long reaction sequences and harsh conditions that compromise overall yield and product quality. For instance, routes involving bromination followed by fluorination often struggle with regioselectivity issues, leading to difficult-to-separate impurities that reduce the purity of the final API. The reliance on hazardous reagents and the generation of substantial toxic waste streams further complicate environmental compliance. These cumulative factors result in a fragile supply chain where production delays are common, and the cost of goods sold remains prohibitively high for widespread access, making these conventional methods unsuitable for the demands of modern, high-volume antiviral drug manufacturing.
The Novel Approach
In stark contrast, the process outlined in CN111349049A offers a streamlined and economically superior alternative that fundamentally redesigns the synthetic logic. The new route initiates with a simple cyclization between 2-aminomalonic diamide and glyoxal to form the pyrazine ring, followed by a strategic benzyl protection step. The crown jewel of this innovation is the subsequent fluorination step, which utilizes elemental fluorine gas within a continuous flow microchannel reactor. This setup allows for precise control over reaction parameters such as temperature and residence time, ensuring high selectivity for the desired 6-fluoro substitution while minimizing over-fluorination or ring degradation.
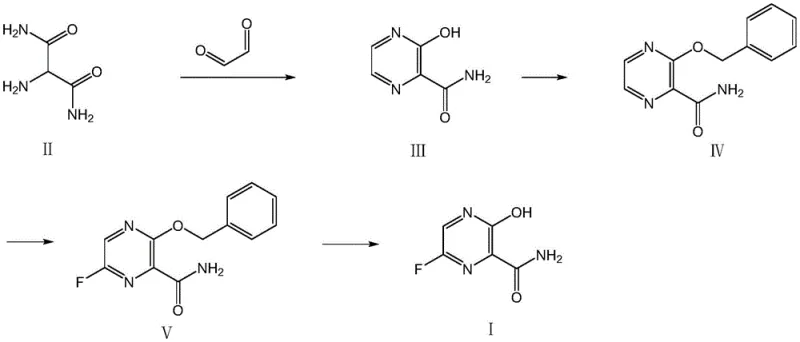
This novel approach effectively shortens the entire process route by eliminating the need for multiple protection-deprotection cycles and expensive transition metal catalysts found in prior art. By adopting a continuous flow system, the reaction safety is dramatically enhanced, as the small reactor volume minimizes the inventory of hazardous fluorine gas at any given moment. The result is a synthesis pathway that is not only shorter and higher yielding but also inherently safer and more environmentally friendly. This technological leap facilitates the production of high-purity pharmaceutical intermediates at a fraction of the cost and time required by legacy methods, positioning it as the preferred choice for reliable agrochemical intermediate supplier networks and pharma manufacturers alike.
Mechanistic Insights into Microchannel Fluorination and Protection Strategies
The core technical breakthrough of this patent lies in the sophisticated application of microchannel technology to manage the kinetics of electrophilic fluorination. In traditional batch reactors, the introduction of fluorine gas often leads to localized hot spots and uncontrolled exotherms, resulting in poor selectivity and potential safety incidents. However, the silicon carbide microchannel reactor utilized in this process provides exceptional heat transfer coefficients and rapid mixing capabilities. When the solution of the protected intermediate (Compound IV) is pumped into the reactor alongside a diluted stream of fluorine and nitrogen, the reactants mix almost instantaneously on a molecular level. This ensures that the concentration of fluorine remains low and uniform throughout the reaction zone, promoting selective mono-fluorination at the 6-position of the pyrazine ring.
Moreover, the choice of formic acid as both solvent and catalyst plays a pivotal role in the reaction mechanism. Formic acid likely acts as a proton source that stabilizes the intermediate species formed during the electrophilic attack, while also solubilizing the organic substrate effectively. The benzyl protection group installed in the preceding step is crucial; it shields the hydroxyl group at the 3-position from unwanted side reactions with fluorine, ensuring that the fluorination occurs exclusively on the carbon skeleton. This strategic protection-deprotection sequence, combined with the precision of flow chemistry, results in a clean reaction profile with minimal byproduct formation. Such mechanistic control is essential for achieving the stringent purity specifications required for clinical-grade antiviral drugs.
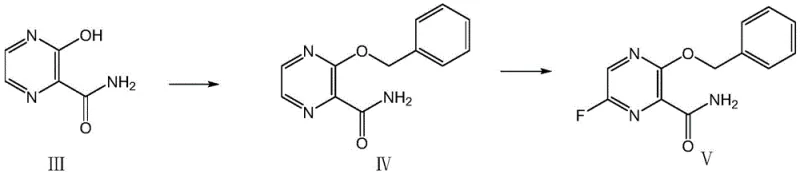
How to Synthesize 6-Fluoro-3-benzyloxy-2-pyrazinecarboxamide Efficiently
The synthesis of this critical intermediate involves a logical progression of unit operations designed for maximum efficiency and safety. The process begins with the cyclization of inexpensive starting materials to build the heterocyclic core, followed by protection to enable selective functionalization. The defining step is the continuous flow fluorination, which requires specialized equipment but offers unparalleled control. Finally, the removal of the protecting group yields the target molecule ready for downstream conversion to the API. This sequence exemplifies modern process chemistry principles, prioritizing atom economy and operational safety. For detailed standard operating procedures and specific parameter optimization, please refer to the technical guide below.
- Cyclization of 2-aminomalonic diamide and glyoxal under alkaline conditions to form the pyrazine ring structure.
- Benzyl protection of the hydroxyl group using benzyl bromide and potassium carbonate to prevent side reactions.
- High-selectivity fluorination using fluorine gas in a silicon carbide microchannel reactor with formic acid as the solvent.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthesis route offers transformative benefits that directly impact the bottom line and operational resilience. The most immediate advantage is the drastic reduction in raw material costs achieved by substituting expensive noble metal catalysts and complex halogenated precursors with bulk commodity chemicals. 2-aminomalonic diamide and glyoxal are widely available in the global chemical market, ensuring a stable and diversified supply base that is less susceptible to geopolitical disruptions or vendor monopolies. This shift not only lowers the direct cost of goods but also simplifies the logistics of raw material sourcing, allowing procurement managers to negotiate better terms due to the commoditized nature of the inputs.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts and chiral ligands removes a significant cost center from the manufacturing budget. Furthermore, the removal of hazardous nitration steps negates the need for costly explosion-proof infrastructure and specialized safety containment systems. The continuous flow nature of the fluorination step also implies higher throughput per unit of reactor volume compared to batch processing, leading to better asset utilization and lower fixed costs per kilogram of product. These factors combine to deliver substantial cost savings in pharmaceutical intermediate manufacturing without compromising on quality.
- Enhanced Supply Chain Reliability: By simplifying the synthetic route and reducing the number of unit operations, the overall lead time for production is significantly compressed. The robustness of the microchannel process against thermal runaways ensures consistent batch-to-batch quality, reducing the risk of production failures or off-spec material that could disrupt supply schedules. Additionally, the use of stable, non-hazardous starting materials for the initial steps allows for flexible production planning, enabling manufacturers to respond rapidly to surges in demand for antiviral medications during public health emergencies.
- Scalability and Environmental Compliance: The modular nature of microchannel reactors facilitates linear scale-up; increasing capacity often involves numbering up reactor modules rather than building massive new vessels, which accelerates time-to-market for commercial volumes. Environmentally, the process generates less hazardous waste by avoiding nitration byproducts and heavy metal residues, simplifying wastewater treatment and disposal. This alignment with green chemistry principles reduces regulatory burdens and enhances the sustainability profile of the supply chain, a key metric for modern corporate responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and feasibility of the process for potential partners and stakeholders. Understanding these nuances is critical for evaluating the technology's fit within existing manufacturing frameworks.
Q: How does the microchannel reactor improve safety in fluorination compared to batch processes?
A: The microchannel reactor provides superior heat transfer and mixing efficiency, allowing for precise control of the highly exothermic fluorine gas reaction. This eliminates the thermal runaway risks associated with traditional batch nitration or fluorination methods, significantly enhancing operational safety.
Q: What are the cost advantages of using 2-aminomalonic diamide as a starting material?
A: Unlike previous routes that rely on expensive palladium catalysts or complex halogenated precursors, 2-aminomalonic diamide and glyoxal are bulk commodity chemicals. This shift drastically reduces raw material costs and simplifies the supply chain by avoiding specialized reagents.
Q: Does this process eliminate the need for explosion-proof equipment?
A: Yes, by replacing the hazardous nitration steps found in older patents with a controlled microchannel fluorination process, the requirement for specialized explosion-proof infrastructure is removed, lowering capital expenditure for manufacturing facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Favipiravir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped to handle continuous flow chemistry and hazardous reagents safely, adhering to stringent purity specifications and rigorous QC labs to guarantee the highest quality standards for every batch of Favipiravir intermediate we produce.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your antiviral drug programs. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this technology can optimize your budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive edge in the global pharmaceutical market.
