Advanced Manufacturing of 2-Amino-6-Chloropurine: A High-Yield Route for Global API Production
The global demand for antiviral therapeutics continues to drive the need for robust, scalable synthesis routes for key nucleoside intermediates. Specifically, the production of Famciclovir, a potent second-generation acyclic nucleotide analogue used to treat herpes virus infections, relies heavily on the availability of high-purity 2-amino-6-chloropurine. Recent intellectual property developments, specifically patent CN108892669B, disclose a transformative manufacturing methodology that addresses long-standing inefficiencies in this sector. This technical insight report analyzes the strategic value of this new oxidation-chlorination-reduction cascade, offering a compelling alternative to legacy processes that have historically plagued R&D and supply chain teams with low yields and complex waste management issues. By shifting the synthetic logic to activate the purine ring prior to halogenation, this process unlocks substantial operational improvements for any reliable pharmaceutical intermediate supplier seeking to optimize their portfolio.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2-amino-6-chloropurine has been hindered by significant physicochemical barriers inherent to the guanine substrate. Traditional routes, such as those disclosed in WO93/15075, attempt direct chlorination of guanine using phosphorus oxychloride. However, guanine exhibits notoriously poor solubility in standard organic chlorinating media, necessitating the use of large quantities of expensive phase transfer catalysts to facilitate the reaction. Even with these additives, the reaction kinetics remain sluggish, often capping yields at a commercially unviable 30% to 42%. Furthermore, alternative pathways like the one described in EP0543095A2 involve reacting guanine with DMF and chlorinating agents to form unstable imine intermediates.
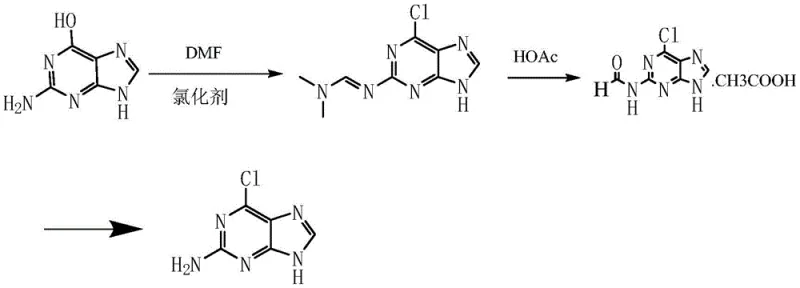
As illustrated in the conventional pathway above, these legacy methods generate viscous reaction masses that are exceptionally difficult to filter or centrifuge, creating severe bottlenecks in downstream processing. The post-treatment often requires multiple recrystallization steps and alkalization procedures, which not only extend the production cycle but also generate substantial volumes of saline wastewater. For procurement managers, these inefficiencies translate directly into higher cost of goods sold (COGS) and unpredictable lead times, making the secure sourcing of this critical intermediate a persistent challenge in the antiviral supply chain.
The Novel Approach
In stark contrast to the direct substitution strategies of the past, the methodology outlined in CN108892669B introduces a logical, stepwise activation of the purine scaffold. This novel approach fundamentally reorders the synthetic sequence by performing an oxidation reaction on the amino group of guanine prior to chlorination. By converting the 2-amino group into a 2-nitro group (Compound A), the electron density of the ring is altered, which remarkably enhances the reactivity of the 6-hydroxyl group towards nucleophilic substitution. This strategic modification allows the subsequent chlorination step to proceed rapidly and completely without the need for harsh conditions or exotic catalysts.
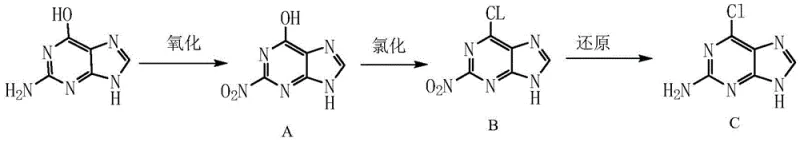
The visual representation of this new route highlights the elegance of the three-step cascade: Oxidation, Chlorination, and Reduction. Following the formation of the chloro-nitro intermediate (Compound B), a final reduction step restores the amino functionality, delivering the target 2-amino-6-chloropurine (Compound C) with exceptional purity. This sequence not only bypasses the solubility issues of raw guanine by utilizing soluble intermediates but also simplifies the workup procedure to basic distillation or filtration. For a reliable pharmaceutical intermediate supplier, adopting this route represents a paradigm shift from struggling with heterogeneous mixtures to managing a streamlined, homogeneous process flow that is inherently safer and more scalable.
Mechanistic Insights into Oxidation-Chlorination-Reduction Cascade
The core innovation of this process lies in the precise control of electronic effects within the purine ring system during the initial oxidation phase. In Step S1, the reaction utilizes mild oxidants such as hydrogen peroxide or sodium hypochlorite in a solvent system that can range from polar aprotic organics like DMF to environmentally benign 2% to 15% alkaline water. The use of alkaline water is particularly noteworthy from a mechanistic standpoint; it ensures the complete dissolution of guanine through salt formation, creating a true homogeneous phase. This homogeneity is critical for heat transfer and reaction uniformity, allowing the temperature to be tightly controlled between 15°C and 30°C. The conversion of the electron-donating amino group to an electron-withdrawing nitro group deactivates the ring towards unwanted side reactions while activating the C6-OH bond for the subsequent nucleophilic attack by chlorine species.
Following oxidation, the chlorination mechanism in Step S2 leverages the activated state of Compound A. Reagents such as thionyl chloride (SOCl2) or phosphorus oxychloride (POCl3) are introduced at low temperatures (0-5°C) to manage exotherms before heating to reflux. The nitro group's presence facilitates the displacement of the hydroxyl group, forming the stable 6-chloro-2-nitro-9H-purine (Compound B). Finally, the reduction in Step S3 employs reagents like hydrazine hydrate or sodium sulfite to selectively reduce the nitro group back to an amine without affecting the newly installed chloro substituent. This chemoselectivity is vital for impurity control, ensuring that the final product meets stringent purity specifications of over 99.7% without requiring complex chromatographic purification, thereby preserving yield and reducing solvent consumption.
How to Synthesize 2-Amino-6-Chloropurine Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and thermal profiles to maximize the reported 81.6% yield. The process begins with the suspension of guanine in a solvent, followed by the controlled addition of oxidant to generate the nitro-intermediate. Once isolated or carried forward, this intermediate undergoes chlorination under reflux conditions, followed by a final reduction step. The detailed standardized operating procedures, including specific molar ratios of oxidant to guanine (1:1 to 1:3) and precise temperature ramps for each stage, are critical for reproducibility. For process chemists looking to replicate this high-efficiency pathway, the following guide outlines the essential operational parameters derived from the patent examples.
- Oxidize Guanine using hydrogen peroxide or sodium hypochlorite in alkaline water or organic solvents at 15-30°C to obtain 2-nitro-9H-purine-6-ol (Compound A).
- Chlorinate Compound A using thionyl chloride or phosphorus oxychloride in methanol or dichloroethane, refluxing to form 6-chloro-2-nitro-9H-purine (Compound B).
- Reduce Compound B using hydrazine hydrate or sodium sulfite in methanol at 50°C to reflux, yielding high-purity 2-amino-6-chloropurine (Compound C).
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the transition to this novel synthesis route offers profound economic and logistical benefits. The elimination of expensive phase transfer catalysts and the ability to use cheaper, safer oxidants like hydrogen peroxide directly impact the raw material cost structure. Furthermore, the shift towards homogeneous reaction conditions in the first step drastically reduces processing time and equipment wear, leading to higher throughput per batch. These factors combine to create a manufacturing profile that is not only more cost-effective but also more resilient to market fluctuations in reagent pricing.
- Cost Reduction in Manufacturing: The replacement of complex, multi-step purification protocols with simple distillation and filtration significantly lowers utility and labor costs. By avoiding the use of large amounts of phosphorus-containing reagents in favor of thionyl chloride where appropriate, the process minimizes the generation of hazardous phosphorus wastewater, which is costly to treat. Additionally, the high yield of over 80% means less raw material is wasted per kilogram of finished product, directly enhancing the margin potential for cost reduction in API manufacturing.
- Enhanced Supply Chain Reliability: The raw materials required for this process, such as guanine, hydrogen peroxide, and hydrazine hydrate, are commodity chemicals with robust global supply chains. Unlike specialized catalysts that may have single-source suppliers, these inputs are readily available from multiple vendors, reducing the risk of supply disruption. The simplified operation steps also mean that production can be scaled up or down more flexibly to meet demand spikes, ensuring a steady flow of high-purity pharmaceutical intermediates to downstream drug manufacturers.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this route is superior due to the avoidance of chlorine gas and the reduction of viscous waste streams. The use of alkaline water as a solvent in the oxidation step is a green chemistry win, reducing the volume of organic solvents needed. This aligns with increasingly strict environmental regulations, making the commercial scale-up of complex pharmaceutical intermediates smoother and less prone to regulatory shutdowns, thereby securing long-term supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is crucial for R&D directors evaluating process feasibility and procurement officers assessing vendor capabilities. The answers are derived directly from the experimental data and technical disclosures within the patent literature, providing a factual basis for decision-making.
Q: Why is the oxidation step performed before chlorination in this new process?
A: Performing oxidation first converts the amino group to a nitro group, which significantly increases the activity of the hydroxyl group at the 6-position. This activation allows the subsequent chlorination reaction to proceed under milder conditions with higher efficiency compared to direct chlorination of guanine.
Q: What are the primary advantages of using alkaline water as a solvent in Step 1?
A: Using 2% to 15% alkaline water ensures that guanine is fully dissolved, creating a homogeneous reaction system. This eliminates mass transfer limitations found in heterogeneous systems, improves reaction safety by avoiding volatile organic solvents in the initial step, and significantly reduces the generation of hazardous waste.
Q: How does this method improve yield compared to traditional WO93/15075 processes?
A: Traditional direct chlorination methods often suffer from low solubility of guanine and require expensive phase transfer catalysts, resulting in yields of only 30-42%. This novel three-step route achieves yields up to 81.6% by optimizing reaction kinetics through intermediate activation and using safer, more effective reagents like thionyl chloride and hydrazine hydrate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-6-Chloropurine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a new patent must be translated into tangible commercial reality. As a dedicated CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this optimized route to life. Our facilities are equipped with rigorous QC labs capable of verifying the stringent purity specifications demanded by the antiviral market, ensuring that every batch of 2-amino-6-chloropurine meets the highest international standards. We are committed to leveraging this advanced chemistry to deliver a product that balances exceptional quality with competitive economics.
We invite global partners to collaborate with us to unlock the full potential of this efficient synthesis method. Whether you require a Customized Cost-Saving Analysis for your specific volume requirements or need to review specific COA data and route feasibility assessments, our technical procurement team is ready to assist. Contact us today to discuss how we can secure your supply chain with high-purity intermediates produced via this state-of-the-art, environmentally responsible process.
