Advanced Manufacturing of High-Purity Methylnaltrexone Bromide for Global Pharmaceutical Supply Chains
Advanced Manufacturing of High-Purity Methylnaltrexone Bromide for Global Pharmaceutical Supply Chains
The global demand for effective treatments for opioid-induced constipation (OIC) has placed significant pressure on the supply chains of critical active pharmaceutical ingredients (APIs) such as Methylnaltrexone Bromide. As a peripheral mu-opioid receptor antagonist, this compound offers a vital therapeutic window by alleviating gastrointestinal side effects without compromising central analgesic efficacy. The technical landscape for producing this complex morphinan derivative has historically been fraught with challenges regarding yield, purity, and scalability. However, the technological breakthroughs detailed in patent CN102351869B present a transformative approach to synthesis that addresses these longstanding industrial bottlenecks. This report provides a deep technical analysis of this novel preparation method, highlighting its potential to redefine cost structures and supply reliability for multinational pharmaceutical enterprises seeking a reliable API intermediate supplier.
At the core of this innovation is a strategic shift from protective group chemistry to a direct alkylation and recovery strategy. Traditional synthetic routes often struggle with the reactivity of the phenolic hydroxyl group at the 3-position of the morphinan skeleton, leading to unwanted O-alkylation byproducts that reduce overall yield and complicate purification. The method disclosed in CN102351869B elegantly circumvents this issue by accepting the formation of the O-methylated byproduct during the initial quaternary amination step and subsequently converting it back to the desired phenolic structure through a controlled demethylation process. This not only simplifies the reaction sequence but also drastically reduces solvent consumption and waste generation, aligning perfectly with modern green chemistry principles and cost reduction in pharmaceutical manufacturing goals.
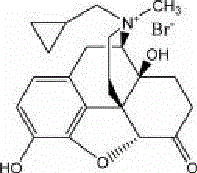
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical precedents for synthesizing Methylnaltrexone Bromide, such as those described in US4176186 and WO2006/127899, rely heavily on multi-step protection and deprotection strategies that are inherently inefficient for large-scale production. For instance, early methods involved direct N-alkylation without base catalysis, which frequently resulted in incomplete conversion of the starting Naltrexone Hydrochloride and significant formation of phenolic hydroxyl alkylates that were difficult to separate. Other approaches necessitated the use of protecting groups like isobutyryl or benzyl to mask the phenolic functionality prior to quaternization. While these methods technically prevent O-alkylation, they introduce additional synthetic steps, including hydrolysis or hydrogenation, which increase the cumulative loss of material and extend the production cycle time considerably.
Furthermore, a critical bottleneck in many prior art methods is the reliance on column chromatography for the purification of intermediates and the final product. In an industrial setting, column chromatography is notoriously difficult to scale, consumes vast quantities of expensive solvents, and creates significant hazardous waste disposal issues. The low refining yields associated with these traditional purification techniques often result in a final product that struggles to consistently meet the rigorous purity specifications required for injectable formulations. Consequently, manufacturers utilizing these legacy processes face higher operational expenditures and greater supply chain volatility, making them less competitive in a market that demands both high quality and cost efficiency.
The Novel Approach
The innovative methodology presented in patent CN102351869B fundamentally reimagines the synthetic pathway by turning a traditional liability—the O-alkylated byproduct—into a recoverable asset. Instead of expending resources to prevent the formation of the 3-O-methyl derivative during the quaternary amination of Naltrexone Hydrochloride, this process allows the reaction to proceed to completion, generating a mixture of the target quaternary ammonium salt and the O-methylated byproduct. This mixture is then subjected to a directed demethylation treatment using mineral acids, which selectively cleaves the methyl ether bond at the 3-position without degrading the sensitive quaternary ammonium structure or the morphinan backbone. This clever chemical maneuver effectively recycles the byproduct into the target molecule, thereby boosting the overall yield and simplifying the downstream processing requirements.
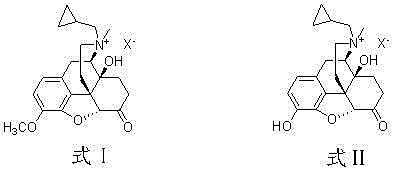
By eliminating the need for protective group manipulation and column chromatography, this novel approach offers a streamlined workflow that is exceptionally well-suited for commercial scale-up of complex pharmaceutical intermediates. The process utilizes readily available reagents such as methyl iodide or dimethyl sulfate and common industrial solvents like acetonitrile or acetone, ensuring robust supply chain continuity. The final purification is achieved through ion exchange and recrystallization from mixed solvents, techniques that are easily adaptable to multi-tonne production scales. This results in a high-purity finished product with a purity exceeding 99.8% and single impurity levels below 0.1%, satisfying the most stringent regulatory standards for parenteral drugs while simultaneously driving down the cost of goods sold through process intensification.
Mechanistic Insights into Quaternary Amination and Acid-Catalyzed Demethylation
The success of this synthetic route hinges on the precise control of reaction conditions during the quaternary amination and subsequent demethylation phases. In the initial step, Naltrexone Hydrochloride reacts with an alkylating agent in the presence of a base within an anhydrous non-proton dipole solvent. The choice of solvent, such as DMF, NMP, or acetonitrile, is critical as it facilitates the solubility of the ionic species and promotes the nucleophilic attack of the tertiary amine on the alkylating agent. The reaction conditions are optimized to ensure complete consumption of the starting material, inevitably leading to the formation of the quaternary ammonium salt alongside the O-alkylated side product. Understanding the kinetics of this competition is vital for R&D teams aiming to replicate this process, as the ratio of products can be influenced by temperature, stoichiometry, and the specific nature of the alkylating reagent employed.
Following the formation of the crude mixture, the pivotal mechanistic step involves the acid-catalyzed hydrolysis of the aryl methyl ether. Under elevated temperatures ranging from 10°C to 120°C, strong mineral acids such as hydrobromic acid or sulfuric acid protonate the ether oxygen, rendering the methyl group susceptible to nucleophilic attack or elimination, ultimately regenerating the free phenolic hydroxyl group. This demethylation must be carefully monitored to prevent Hofmann degradation of the quaternary ammonium moiety, a common side reaction under alkaline or harsh thermal conditions. The patent specifies the use of specific acid concentrations and reaction times to maximize the conversion of the byproduct while maintaining the integrity of the morphinan scaffold. This level of mechanistic control ensures that the impurity profile remains exceptionally clean, with single impurities maintained below 0.1%, which is a critical parameter for ensuring patient safety and regulatory approval.
How to Synthesize Methylnaltrexone Bromide Efficiently
The implementation of this synthesis route requires strict adherence to the optimized parameters regarding solvent selection, acid concentration, and thermal profiles to ensure reproducibility and high yield. The process begins with the preparation of the quaternary ammonium mixture, followed by the critical demethylation step that converts impurities into product, and concludes with a robust purification protocol involving ion exchange and crystallization. For R&D directors and process engineers looking to adopt this technology, understanding the nuances of each unit operation is essential for successful technology transfer. The following guide outlines the standardized synthesis steps derived from the patent data, providing a clear roadmap for laboratory and pilot-scale execution.
- Perform direct N-alkylation on Naltrexone Hydrochloride using an alkylating reagent in an anhydrous non-proton dipole solvent to obtain a quaternary ammonium mixture.
- Treat the resulting mixture with mineral acid to induce directed demethylation, converting the 3-O-methyl byproduct back into the target Methylnaltrexone structure.
- Execute ion exchange followed by recrystallization using mixed solvents to isolate the final product with purity exceeding 99.8%.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis method offers profound advantages for procurement managers and supply chain heads tasked with optimizing the cost and reliability of API sourcing. The primary value driver is the significant reduction in manufacturing complexity, which directly translates to lower operational costs and improved margin structures. By removing the need for column chromatography, the process eliminates a major bottleneck that typically limits throughput and increases solvent procurement costs. Furthermore, the ability to convert byproducts into the final active ingredient maximizes the utility of raw materials, reducing the effective cost per kilogram of the produced API. This efficiency gain is particularly valuable in a volatile raw material market, where maximizing yield from expensive starting materials like Naltrexone is paramount for maintaining profitability.
- Cost Reduction in Manufacturing: The elimination of protective group chemistry and column chromatography results in a drastic simplification of the production workflow, leading to substantial cost savings. Without the need for multiple protection and deprotection steps, the consumption of reagents and solvents is significantly reduced, lowering the variable costs associated with each production batch. Additionally, the recycling of the O-methylated byproduct into the target molecule enhances the overall mass balance of the process, ensuring that a higher percentage of input materials are converted into saleable product. This efficient utilization of resources allows for a more competitive pricing strategy, enabling suppliers to offer high-purity intermediates at a price point that supports the economic viability of the final drug product.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and standard unit operations greatly enhances the robustness of the supply chain, reducing the risk of disruptions caused by specialized reagent shortages. Since the process avoids complex catalytic systems or exotic protecting groups that may have limited availability, manufacturers can secure raw materials more easily and maintain consistent production schedules. The simplified workflow also shortens the overall lead time for production, allowing for faster response to market demand fluctuations. For supply chain heads, this means a more predictable and resilient sourcing channel for high-purity pharmaceutical intermediates, minimizing the risk of stockouts that could impact downstream drug formulation and patient access.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scalability, utilizing reaction conditions and equipment that are standard in modern chemical manufacturing facilities. The avoidance of column chromatography not only improves throughput but also significantly reduces the volume of hazardous organic waste generated, facilitating easier compliance with environmental regulations. The use of ion exchange and crystallization for purification is a proven, scalable technology that can be seamlessly expanded from pilot plants to multi-tonne commercial reactors. This scalability ensures that the supply of Methylnaltrexone Bromide can grow in tandem with market demand, supporting the long-term commercial success of opioid antagonist therapies without encountering the technical barriers often associated with scaling complex organic syntheses.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and quality of Methylnaltrexone Bromide synthesized via this advanced method. These insights are derived directly from the patent specifications and are intended to clarify the capabilities and limitations of the technology for potential partners and stakeholders. Understanding these details is crucial for making informed decisions about process adoption and supply agreements.
Q: How does this novel method handle the phenolic hydroxyl alkylation byproduct?
A: Unlike conventional methods that view phenolic alkylation as waste, this process utilizes acid-catalyzed demethylation to convert the 3-O-methyl byproduct back into the target Methylnaltrexone, significantly improving overall yield.
Q: Does this synthesis route require column chromatography for purification?
A: No, the patented method explicitly overcomes the defect of using column chromatography by employing ion exchange and specific mixed-solvent recrystallization, making it highly suitable for industrial scale-up.
Q: What purity levels can be achieved with this manufacturing process?
A: The process is designed to achieve a final product purity greater than 99.8%, with single impurity content controlled below 0.1%, meeting stringent requirements for injectable pharmaceutical ingredients.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methylnaltrexone Bromide Supplier
The technical superiority of the synthesis route described in CN102351869B underscores the immense potential for producing high-quality Methylnaltrexone Bromide at a commercial scale. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that this innovative chemistry can be translated into a reliable supply stream for your organization. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of API intermediate meets the exacting standards required for global pharmaceutical markets. We understand the critical nature of supply continuity in the healthcare sector and are committed to delivering consistent quality through our advanced manufacturing capabilities.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this more efficient production method. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project needs. Let us partner with you to enhance the efficiency and reliability of your opioid antagonist supply chain, ensuring that life-saving medications reach patients without delay.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
