Advanced Refining Technology for High-Purity Methylnaltrexone Bromide Manufacturing
Advanced Refining Technology for High-Purity Methylnaltrexone Bromide Manufacturing
The pharmaceutical industry continuously demands higher purity standards for active pharmaceutical ingredients (APIs) and their critical intermediates, particularly for potent opioid antagonists like Methylnaltrexone Bromide. Patent CN102558189A introduces a groundbreaking refining methodology that addresses the persistent challenges of impurity removal in MNTX production. As a selective peripheral μ-opioid receptor antagonist, Methylnaltrexone Bromide requires exceptional purity to ensure patient safety and efficacy, especially given its use in treating opioid-induced constipation without compromising central analgesia. This technical insight explores how the integration of reverse-phase silica gel column chromatography with strategic recrystallization steps creates a robust pathway for producing medicinal-grade material. By moving beyond simple precipitation techniques, this approach offers a reliable pharmaceutical intermediates supplier with the capability to meet stringent pharmacopeial requirements.
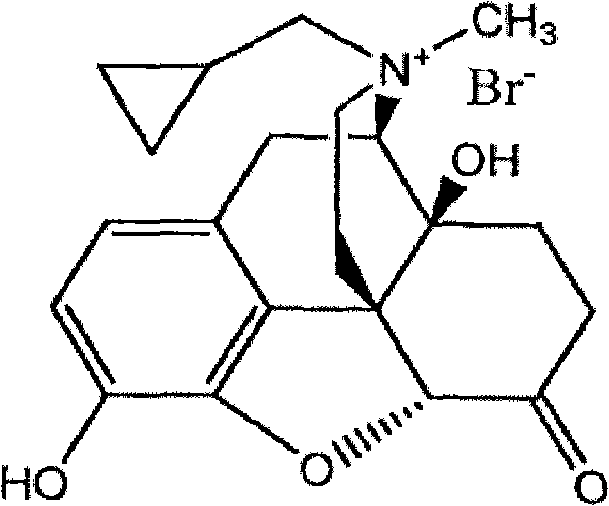
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of Methylnaltrexone Bromide has relied heavily on traditional recrystallization techniques, such as those described in US Patent 4176186. These conventional methods typically involve dissolving the crude product in organic solvents like acetone or methanol and inducing crystallization through cooling or anti-solvent addition. However, these approaches suffer from significant limitations regarding impurity clearance. Data indicates that products refined solely through traditional methanol/ether recrystallization often exhibit total related substance levels as high as 1.7%, with individual impurities reaching 0.5%. Such impurity profiles are unacceptable for modern parenteral formulations, where strict limits on genotoxic or structurally similar by-products are enforced. Furthermore, the reliance on volatile ethers and multiple unoptimized crystallization cycles can lead to inconsistent yields and batch-to-batch variability, complicating the supply chain for high-purity opioid antagonists.
The Novel Approach
The innovative strategy outlined in CN102558189A fundamentally shifts the purification paradigm by introducing reverse-phase silica gel column chromatography as a core separation step. Unlike normal phase chromatography, reverse-phase systems utilize a non-polar stationary phase and a polar mobile phase, which is exceptionally effective for separating polar quaternary ammonium salts from their synthetic by-products. This method is often applied to crude materials generated from direct methylation routes, such as the reaction of naltrexone free base with methyl bromide shown in prior art. 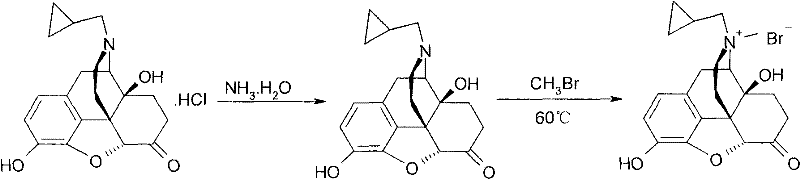 By combining this chromatographic precision with optimized methanol-water recrystallization (specifically at a 4:1 volume ratio), the process achieves a synergistic effect. The result is a dramatic enhancement in product quality, with assay values consistently exceeding 99.0% and total impurities driven down to negligible levels below 0.1%, effectively solving the cost reduction in API manufacturing associated with failed batches and re-processing.
By combining this chromatographic precision with optimized methanol-water recrystallization (specifically at a 4:1 volume ratio), the process achieves a synergistic effect. The result is a dramatic enhancement in product quality, with assay values consistently exceeding 99.0% and total impurities driven down to negligible levels below 0.1%, effectively solving the cost reduction in API manufacturing associated with failed batches and re-processing.
Mechanistic Insights into Reverse-Phase Chromatographic Separation
The core mechanism driving the success of this refining process lies in the differential partitioning of the target molecule and its impurities between the hydrophobic stationary phase and the hydrophilic mobile phase. Methylnaltrexone Bromide, being a quaternary ammonium salt, possesses distinct polarity characteristics compared to unreacted naltrexone or over-alkylated by-products. In the reverse-phase silica gel column, the stationary phase retains non-polar contaminants more strongly, while the highly polar MNTX elutes earlier under low organic solvent conditions. The patent specifies a gradient elution strategy, initiating with 1% aqueous methanol to wash away highly polar inorganic salts and very hydrophilic impurities, followed by a 5% aqueous methanol wash to elute the target compound. This precise control over solvent strength ensures that the main component is collected in a narrow, high-concentration fraction, minimizing the co-elution of structurally related substances that share similar solubility profiles in standard recrystallization solvents.
Furthermore, the integration of activated carbon treatment prior to chromatography plays a critical role in removing colored impurities and high-molecular-weight tarry by-products that could otherwise foul the chromatography column or co-crystallize with the product. The subsequent recrystallization steps serve as a polishing mechanism, leveraging the specific solubility curve of MNTX in methanol-water mixtures. By heating the solution to reflux and slowly cooling to room temperature, the crystal lattice forms selectively around the pure MNTX molecules, excluding remaining trace impurities that remain in the mother liquor. This multi-barrier approach to purification—adsorption, chromatographic separation, and crystal lattice exclusion—ensures that the final commercial scale-up of complex opioid antagonists meets the rigorous demands of global regulatory bodies without the need for hazardous solvent exchanges.
How to Synthesize Methylnaltrexone Bromide Efficiently
The synthesis and subsequent refinement of Methylnaltrexone Bromide require a disciplined approach to process parameters to ensure reproducibility and high yield. The patented method outlines a specific sequence that begins with the preparation of the crude material, followed by a rigorous three-stage purification protocol. Operators must pay close attention to solvent ratios, particularly the 4:1 methanol-to-water ratio which has been empirically determined to optimize both solubility during dissolution and recovery during crystallization. The following guide summarizes the critical operational phases derived from the patent examples, providing a roadmap for technical teams aiming to implement this high-efficiency workflow in a GMP environment. Detailed standardized synthesis steps are provided in the guide below.
- Dissolve crude Methylnaltrexone Bromide in a 4: 1 Methanol-Water mixture, treat with activated carbon, reflux, and crystallize.
- Purify the intermediate via reverse-phase silica gel column chromatography using gradient elution with 1% and 5% aqueous methanol.
- Perform a final recrystallization of the chromatographed product in Methanol-Water (4: 1) followed by vacuum drying at 80°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this advanced refining technology translates into tangible operational improvements and risk mitigation. Traditional purification methods often struggle with consistency, leading to variable yields and the potential for batch rejection due to impurity spikes. By implementing a chromatography-based refining strategy, manufacturers can stabilize their output quality, ensuring a consistent supply of medicinal-grade intermediates. This stability is crucial for maintaining uninterrupted production schedules for downstream formulation partners who rely on just-in-time delivery of high-quality raw materials. The ability to consistently meet purity specifications reduces the administrative and financial burden associated with quality investigations and out-of-specification (OOS) results.
- Cost Reduction in Manufacturing: While chromatography is often perceived as expensive, the overall cost structure is optimized by significantly improving the yield of usable product per batch. Traditional methods may require multiple re-crystallizations to approach acceptable purity, each step incurring solvent, energy, and time costs while losing product to mother liquors. The high selectivity of the reverse-phase column minimizes product loss, and the use of common solvents like methanol and water eliminates the need for specialized, high-cost reagents. Additionally, the removal of toxic ether solvents simplifies waste disposal protocols and reduces environmental compliance costs, contributing to substantial cost savings in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: The robustness of this refining method enhances supply chain continuity by reducing the likelihood of production delays caused by purification failures. The process utilizes mature, industrially available reverse-phase silica gel columns, meaning that equipment sourcing is straightforward and maintenance is well-understood by plant engineers. This accessibility ensures that production capacity can be ramped up quickly to meet market demand without the long lead times associated with custom reactor fabrication. Furthermore, the mild reaction conditions (ambient pressure and moderate temperatures) reduce equipment stress and maintenance downtime, ensuring a steady flow of high-purity opioid antagonists to the market.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this process represents a significant advancement. The elimination of diethyl ether, a highly flammable and volatile solvent, drastically improves plant safety profiles and lowers insurance premiums. The primary solvent system consists of methanol and water, which are easier to recover and recycle using standard distillation infrastructure. This aligns with green chemistry principles by reducing the generation of hazardous waste streams. The scalability is further supported by the linear nature of column chromatography; increasing batch size simply requires proportionally larger columns, a standard engineering practice that facilitates the transition from pilot scale to multi-ton commercial production without fundamental process redesign.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this refining technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on the feasibility and benefits of the method. Understanding these details is essential for technical procurement teams evaluating potential suppliers or considering technology transfer for internal production capabilities.
Q: How does this refining method improve purity compared to traditional recrystallization?
A: Traditional methods like US4176186 rely solely on recrystallization, often yielding products with total impurities around 1.7%. This patented method integrates reverse-phase silica gel column chromatography, reducing total impurities to below 0.1% and achieving content purity exceeding 99%.
Q: Is this refining process suitable for large-scale industrial production?
A: Yes, the process utilizes mature industrial equipment for reverse-phase chromatography and standard recrystallization vessels. The mild conditions (ambient pressure, moderate temperatures) and scalable solvent systems make it highly adaptable for commercial manufacturing from 100 kgs to multi-ton scales.
Q: What specific solvents are used in the purification workflow?
A: The process primarily uses a Methanol-Water system. A 4:1 volume ratio is preferred for recrystallization steps, while the chromatography step employs gradient elution starting with 1% methanol in water, followed by 5% methanol in water, ensuring effective separation of polar impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methylnaltrexone Bromide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the final drug product is inextricably linked to the purity of its starting materials. Our technical team has extensively analyzed advanced refining pathways, including the chromatography-based methods described in CN102558189A, to ensure our manufacturing processes deliver exceptional value. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require clinical trial materials or full-scale commercial supply, our capacity is ready to meet your needs. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Methylnaltrexone Bromide we supply meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to optimize your supply chain for this critical opioid antagonist. Our experts are prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply chain that prioritizes purity, reliability, and technical excellence, ensuring your downstream formulations succeed in the competitive global marketplace.
