Advanced Photocatalytic Decarboxylation for High-Purity Deuterated Pharmaceutical Intermediates
The landscape of isotopic labeling, particularly for deuterated pharmaceutical intermediates, is undergoing a significant transformation driven by the urgent need for greener and more cost-effective synthetic methodologies. A pivotal advancement in this domain is detailed in patent CN111718245A, which discloses a novel method for preparing deuterated chemicals through photocatalytic decarboxylation conversion. This technology represents a paradigm shift from traditional high-energy processes to a mild, visible-light-driven system utilizing a non-metal-based semiconductor catalyst. By leveraging hexagonal boron-nitrogen-carbon (h-BCN) as a photocatalyst, the process enables the direct replacement of carboxyl groups with deuterium atoms under ambient conditions. This breakthrough not only addresses the long-standing challenges of selective control and functional group compatibility but also offers a robust pathway for the reliable deuterated chemical supplier market to meet the escalating demands of the global pharmaceutical industry for labeled compounds used in ADME studies and metabolic tracing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of deuterated compounds has been plagued by severe operational constraints and economic inefficiencies that hinder widespread adoption in commercial scale-up of complex polymer additives and fine chemicals. Traditional deuteration strategies typically rely on heterogeneous catalysis using precious metals such as palladium or platinum, which necessitates the use of deuterium gas (D2) under high pressure and elevated temperatures. These harsh conditions often lead to poor chemoselectivity, resulting in the over-reduction of sensitive functional groups or the formation of complex impurity profiles that are difficult to separate. Furthermore, the requirement for specialized high-pressure equipment increases capital expenditure and introduces significant safety hazards associated with handling compressed deuterium gas. The reliance on expensive noble metal catalysts also creates a bottleneck in cost reduction in pharmaceutical intermediate manufacturing, as the removal of trace metal residues to meet stringent regulatory standards adds additional downstream processing steps and waste generation.
The Novel Approach
In stark contrast, the novel photocatalytic decarboxylation method described in the patent offers a streamlined and sustainable alternative that fundamentally alters the reaction landscape. By employing h-BCN, a graphite-like semiconductor with a specific surface area of 300-400 m²/g and an absorption band edge of 400-600 nm, the reaction is driven entirely by visible light energy, such as 420 nm LEDs, at room temperature. This approach eliminates the need for external heating and high-pressure infrastructure, thereby drastically simplifying the reactor design and operational protocol. The use of inexpensive and safe deuterium sources, specifically deuterium water or deuterated alcohols, replaces the hazardous deuterium gas, enhancing the overall safety profile of the manufacturing process. As illustrated in the general reaction scheme below, carboxylic acid substrates undergo decarboxylation to generate carbon radicals which subsequently capture deuterium, achieving high site-selectivity without compromising the integrity of other functional groups present in the molecule.
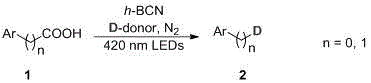
Mechanistic Insights into h-BCN Photocatalytic Decarboxylation
The core of this technological breakthrough lies in the unique electronic properties of the hexagonal boron-nitrogen-carbon (h-BCN) photocatalyst, which facilitates a radical-mediated mechanism under mild irradiation. Upon exposure to visible light, the h-BCN semiconductor absorbs photons to generate electron-hole pairs, initiating the oxidative decarboxylation of the carboxylic acid substrate. This process generates a transient alkyl or aryl radical species after the loss of carbon dioxide, a step that is thermodynamically favorable under the photocatalytic conditions. The generated radical then efficiently abstracts a deuterium atom from the deuterium source, such as deuterated methanol or heavy water, to form the final C-D bond. This radical pathway is distinct from ionic mechanisms often seen in acid-base catalysis, allowing for exceptional tolerance towards various functional groups including ethers, esters, and heterocycles, which are common motifs in drug candidates. The high surface area of the h-BCN material ensures ample active sites for substrate adsorption, contributing to the observed high conversion rates and yields.
From an impurity control perspective, this mechanism offers superior selectivity compared to thermal methods. Because the reaction proceeds at room temperature, thermal degradation pathways and side reactions such as polymerization or rearrangement are effectively suppressed. The specificity of the radical generation at the carboxyl position ensures that deuteration occurs exclusively at the desired site, minimizing the formation of regio-isomers that complicate purification. For instance, in the synthesis of 1,2,3-trimethoxy-5-(methyl-d)benzene, the method achieved a remarkable yield of 87%, demonstrating the efficiency of the radical capture step. The absence of transition metals further guarantees that the final product is free from heavy metal contamination, a critical quality attribute for high-purity OLED material and API intermediates intended for clinical use. This inherent purity reduces the burden on downstream purification units, aligning perfectly with the principles of green chemistry and process intensification.
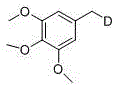
How to Synthesize Deuterated Chemicals Efficiently
The implementation of this photocatalytic protocol is straightforward and designed for ease of adoption in both laboratory and pilot plant settings. The process begins with the preparation of the h-BCN catalyst via a simple calcination of glucose, urea, and boric acid precursors, ensuring a consistent and reproducible supply of the photoactive material. Subsequently, the carboxylic acid substrate is mixed with the catalyst and a deuterium source in a solvent such as acetonitrile or dichloroethane, with the pH adjusted to 8-10 using a mild base like potassium carbonate. The reaction mixture is then stirred under a nitrogen atmosphere while being illuminated by visible light LEDs for approximately 48 hours. Detailed standardized synthesis steps see the guide below, which outlines the precise stoichiometric ratios and workup procedures required to maximize yield and purity.
- Prepare the h-BCN photocatalyst by calcining a mixture of glucose, urea, and boric acid at 1000-1200°C in an ammonia atmosphere.
- Combine the carboxylic acid substrate, h-BCN catalyst, deuterium source (D2O or deuterated alcohol), and base in a reactor under nitrogen protection.
- Stir the reaction mixture under visible light illumination at room temperature for 48 hours, followed by extraction and purification to isolate the deuterated product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photocatalytic technology presents a compelling value proposition centered on risk mitigation and operational efficiency. The elimination of high-pressure deuterium gas removes a major safety hazard from the facility, reducing insurance costs and regulatory compliance burdens associated with hazardous material storage. Furthermore, the substitution of expensive noble metal catalysts with a metal-free organic semiconductor leads to substantial cost savings in raw material procurement. The mild reaction conditions also imply lower energy consumption for heating and cooling, contributing to a reduced carbon footprint and alignment with corporate sustainability goals. These factors collectively enhance the economic viability of producing deuterated standards and labeled drugs, making the supply chain more resilient against fluctuations in the prices of precious metals and specialty gases.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the drastic simplification of the input materials and equipment requirements. By replacing costly deuterium gas and noble metal catalysts with inexpensive heavy water and recyclable h-BCN, the direct material costs are significantly lowered. Additionally, the ambient temperature operation eliminates the need for energy-intensive heating systems, while the absence of metal residues removes the necessity for expensive scavenging resins or complex purification steps. This holistic reduction in operational overhead translates into a more competitive pricing structure for the final deuterated products, allowing manufacturers to maintain healthy margins even in a price-sensitive market environment.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the geopolitical and logistical complexities surrounding the sourcing of rare earth metals and compressed gases. This photocatalytic method mitigates these risks by utilizing commodity chemicals such as urea, boric acid, and glucose for catalyst synthesis, alongside widely available deuterated solvents. The robustness of the h-BCN catalyst, which can be reused multiple times without significant loss of activity, further stabilizes the production schedule by reducing dependency on frequent catalyst replenishment. Consequently, manufacturers can guarantee shorter lead times for high-purity deuterated chemicals, ensuring that R&D pipelines and clinical trial material supplies remain uninterrupted by external supply shocks.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been challenging due to light penetration limits, but advancements in LED reactor technology have made this highly feasible for industrial applications. The modular nature of LED arrays allows for linear scale-up from gram to kilogram scales without the exponential increase in risk associated with high-pressure batch reactors. Moreover, the process generates minimal waste, as the primary byproduct is carbon dioxide, and the solvent can be easily recovered and recycled. This aligns with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions and heavy metal discharge, positioning the technology as a future-proof solution for sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic decarboxylation technology. These insights are derived directly from the patent specifications and experimental data, providing clarity on the practical aspects of adopting this method for industrial synthesis. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this green chemistry approach into their existing manufacturing workflows.
Q: What are the advantages of using h-BCN photocatalysis over traditional deuteration methods?
A: Traditional methods often require high temperatures, high pressures, and expensive noble metal catalysts. The h-BCN photocatalytic method operates at room temperature under visible light, uses inexpensive non-metal catalysts, and employs cheap deuterium sources like heavy water, significantly reducing operational costs and safety risks.
Q: What types of deuterium sources are compatible with this process?
A: The process is highly versatile and utilizes environmentally friendly and cost-effective deuterium sources such as deuterium oxide (heavy water) and deuterated alcohols (e.g., deuterated methanol), eliminating the need for hazardous deuterium gas.
Q: Is this photocatalytic method suitable for large-scale industrial production?
A: Yes, the method features mild reaction conditions and uses stable, reusable catalysts. The reliance on visible light LEDs and ambient pressure makes it highly scalable for commercial manufacturing without the complex engineering required for high-pressure hydrogenation reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Deuterated Chemicals Supplier
As the demand for isotopically labeled compounds continues to surge across the pharmaceutical and agrochemical sectors, partnering with a technically proficient manufacturer is essential for success. NINGBO INNO PHARMCHEM stands at the forefront of this innovation, leveraging advanced photocatalytic technologies to deliver high-quality deuterated intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We operate with stringent purity specifications and utilize rigorous QC labs to verify the isotopic enrichment and chemical purity of every batch, guaranteeing that our products meet the exacting standards required for global regulatory submissions.
We invite you to collaborate with us to optimize your supply chain for deuterated materials. Our technical experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific molecular targets, demonstrating how our photocatalytic capabilities can reduce your overall cost of goods. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your development timelines with our reliable and efficient manufacturing solutions.
