Scalable Metal-Free Photocatalytic Decarboxylation for High-Purity Deuterated Intermediates
Introduction to Next-Generation Deuteration Technology
The landscape of isotopic labeling, particularly for deuterated compounds essential in drug metabolism and pharmacokinetic (DMPK) studies, is undergoing a transformative shift driven by the innovations disclosed in patent CN111718245B. This pivotal intellectual property introduces a groundbreaking photocatalytic decarboxylation strategy that fundamentally alters how deuterated chemicals are synthesized, moving away from hazardous and capital-intensive legacy processes. By leveraging a non-metal-based semiconductor photocatalyst, specifically hexagonal boron carbon nitride (h-BCN), this technology enables the direct conversion of readily available carboxylic acids into valuable deuterated architectures under exceptionally mild conditions. For R&D directors and procurement strategists in the pharmaceutical sector, this represents a paradigm shift towards greener, safer, and more economically viable manufacturing pathways that align with modern sustainability goals while ensuring the high purity required for clinical trial materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of deuterated intermediates has been plagued by significant technical and economic hurdles that constrain supply chain flexibility and inflate production costs. Traditional methodologies predominantly rely on heterogeneous catalysis using precious metals such as palladium or platinum, necessitating the use of high-pressure deuterium gas (D2) which poses severe safety risks and requires specialized high-pressure reactor infrastructure. Furthermore, these harsh conditions often lead to poor functional group tolerance, resulting in complex impurity profiles that demand rigorous and costly downstream purification processes to meet regulatory standards. The reliance on noble metals also introduces supply chain vulnerabilities associated with the fluctuating market prices of rare earth elements and the environmental burden of heavy metal waste disposal, making these conventional routes increasingly unattractive for large-scale commercial manufacturing of fine chemical intermediates.
The Novel Approach
In stark contrast, the novel photocatalytic protocol described in the patent utilizes visible light as the sole energy driver to facilitate decarboxylative deuteration at ambient temperature and pressure, effectively bypassing the need for thermal activation or high-pressure equipment. This metal-free approach employs h-BCN, a graphitic-like semiconductor with a tunable bandgap that absorbs visible light efficiently, generating the necessary reactive species to cleave carbon-carbon bonds and install deuterium atoms with high regioselectivity. The use of inexpensive and safe deuterium donors, such as deuterated methanol or heavy water, further simplifies the reagent profile and eliminates the logistical challenges associated with handling compressed deuterium gas cylinders. As illustrated in the general reaction scheme below, this methodology offers a streamlined, one-pot solution that transforms simple carboxylic acid precursors into deuterated products with remarkable efficiency.
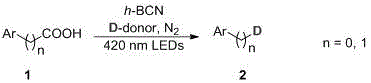
Mechanistic Insights into h-BCN Photocatalytic Decarboxylation
The core of this technological advancement lies in the unique electronic properties of the hexagonal boron carbon nitride (h-BCN) photocatalyst, which functions as a robust, metal-free semiconductor capable of driving redox reactions under visible light irradiation. Upon absorption of photons, typically from 420 nm LEDs, the h-BCN material generates electron-hole pairs that initiate a single-electron transfer (SET) process with the carboxylate anion formed in situ by the base. This oxidation event triggers the rapid decarboxylation of the substrate, generating a transient carbon-centered radical intermediate that is highly reactive yet sufficiently short-lived to prevent unwanted side reactions such as dimerization or over-oxidation. The subsequent hydrogen atom transfer (HAT) or deuterium atom transfer (DAT) from the deuterated solvent to this radical species occurs with high fidelity, ensuring that the deuterium label is installed precisely at the position formerly occupied by the carboxyl group, thereby achieving excellent positional selectivity.
From an impurity control perspective, the mildness of the reaction conditions plays a critical role in maintaining a clean reaction profile, which is paramount for pharmaceutical applications where genotoxic impurities must be strictly controlled. Unlike thermal radical initiators that can degrade sensitive functional groups or promote polymerization, the photo-induced radical generation is spatially and temporally controlled by the light source, allowing for precise modulation of the reaction rate. Furthermore, the absence of transition metals eliminates the risk of metal leaching into the final product, a common failure mode in traditional catalysis that often necessitates additional scavenging steps using specialized resins or activated carbon. This inherent cleanliness of the h-BCN system not only simplifies the purification workflow but also ensures that the final deuterated API intermediates meet the stringent residual metal specifications mandated by global health authorities without extensive post-processing.
How to Synthesize Deuterated Chemicals Efficiently
The practical implementation of this photocatalytic decarboxylation method is designed to be straightforward and adaptable to existing laboratory and pilot plant infrastructure, requiring minimal modification to standard glassware or lighting setups. The process begins with the preparation of the h-BCN catalyst via a simple calcination of glucose, urea, and boric acid, followed by the setup of the reaction vessel where the carboxylic acid substrate is mixed with the catalyst, a mild base, and the deuterium source under an inert atmosphere. Detailed standardized operating procedures regarding stoichiometry, light intensity optimization, and workup protocols are essential for reproducibility, and the comprehensive synthesis steps are outlined in the technical guide below for immediate reference by process chemists.
- Prepare the hexagonal boron carbon nitride (h-BCN) photocatalyst by calcining a mixture of glucose, urea, and boric acid at 1000-1200°C in an ammonia atmosphere.
- Combine the carboxylic acid substrate, h-BCN catalyst, deuterium source (such as deuterated methanol), and a base like potassium carbonate in a reactor under nitrogen protection.
- Irradiate the reaction mixture with visible light (e.g., 420 nm LEDs) at room temperature for approximately 48 hours, followed by extraction and purification to isolate the deuterated product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology translates into tangible strategic benefits that directly impact the bottom line and operational resilience of the manufacturing organization. By shifting away from precious metal catalysts and high-pressure gas inputs, companies can significantly reduce their exposure to volatile raw material markets and eliminate the capital expenditure associated with maintaining high-pressure hydrogenation suites. The ability to run reactions at room temperature using visible light LEDs also results in substantial energy savings compared to thermal processes that require continuous heating and cooling cycles, contributing to a lower overall carbon footprint and aligning with corporate sustainability mandates. Moreover, the simplified workup procedure, which avoids complex metal removal steps, accelerates the production cycle time and increases the throughput capacity of existing facilities without the need for major infrastructure upgrades.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts such as palladium or platinum removes a major cost driver from the bill of materials, while the use of commodity chemicals like deuterated methanol instead of specialized deuterium gas cylinders further drives down reagent costs. Additionally, the removal of heavy metal scavenging agents and the associated filtration steps reduces the consumption of auxiliary materials and lowers waste disposal fees, leading to a leaner and more cost-effective production model that enhances margin potential for high-value deuterated intermediates.
- Enhanced Supply Chain Reliability: Relying on a metal-free catalyst synthesized from abundant precursors like urea and boric acid mitigates the supply chain risks associated with the geopolitical instability often affecting the mining and refining of rare earth metals. The robustness of the h-BCN material allows for long-term storage and repeated use, ensuring a stable supply of catalytic activity that prevents production delays caused by catalyst degradation or supply shortages, thereby guaranteeing consistent delivery schedules for critical drug development timelines.
- Scalability and Environmental Compliance: The modular nature of photoreactors allows for easy scale-up from gram-scale discovery to kilogram-scale production by simply increasing the illuminated surface area or numbering up reactor units, avoiding the heat transfer limitations typical of batch thermal reactors. Furthermore, the green chemistry credentials of this process, characterized by the use of visible light, non-toxic catalysts, and benign solvents, simplify the regulatory approval process for new manufacturing sites and reduce the environmental compliance burden related to hazardous waste management and emissions monitoring.
Frequently Asked Questions (FAQ)
To address common technical inquiries regarding the implementation and scope of this photocatalytic deuteration technology, we have compiled a set of frequently asked questions based on the experimental data and mechanistic understanding derived from the patent literature. These insights are intended to clarify the operational parameters and potential applications for process development teams evaluating this route for their specific compound pipelines, ensuring a clear understanding of the technology's capabilities and limitations before committing to scale-up efforts.
Q: What are the primary advantages of this photocatalytic method over traditional deuteration?
A: Unlike traditional methods that require high temperatures, high pressures of deuterium gas, and expensive noble metal catalysts, this novel approach operates at room temperature under visible light using a metal-free semiconductor, significantly enhancing safety and reducing operational costs.
Q: Which deuterium sources are compatible with this synthesis protocol?
A: The method utilizes environmentally friendly and economically viable deuterium sources such as deuterium oxide (heavy water) or deuterated alcohols like deuterated methanol, eliminating the need for complex and costly deuterated organic reagents.
Q: Is the h-BCN photocatalyst reusable for industrial scale-up?
A: Yes, the hexagonal boron carbon nitride catalyst is robust and can be recovered and reused multiple times without significant loss of activity, which is a critical factor for maintaining cost-efficiency in large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Deuterated Chemicals Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to maintain a competitive edge in the fast-evolving landscape of pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory protocols like the h-BCN photocatalytic decarboxylation can be seamlessly translated into robust, GMP-compliant manufacturing processes. We are committed to delivering high-purity deuterated compounds that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation to verify isotopic enrichment levels and impurity profiles.
We invite you to collaborate with us to leverage this transformative technology for your next drug development project, where our experts can provide a Customized Cost-Saving Analysis tailored to your specific molecular targets. Please contact our technical procurement team today to request specific COA data for our catalog items or to discuss route feasibility assessments for your proprietary deuterated candidates, and let us help you optimize your supply chain for the future of isotopically labeled medicines.
