Revolutionizing Aromatic Azo Compound Production via Advanced Photochemical Catalysis
The landscape of fine chemical synthesis is undergoing a paradigm shift, driven by the urgent need for greener, safer, and more efficient manufacturing processes. A pivotal development in this domain is documented in patent CN111039822B, which introduces a novel photochemical catalytic synthesis method for aromatic azo compounds. This technology represents a significant departure from classical stoichiometric approaches, leveraging the power of light energy to drive selective deoxygenation coupling reactions. For R&D directors and procurement strategists alike, this innovation offers a compelling pathway to produce high-value intermediates used in dyes, pharmaceuticals, and advanced electronic materials. By utilizing nitro-substituted aromatic compounds as stable starting materials instead of hazardous diazonium salts, this method addresses critical safety and environmental bottlenecks that have long plagued the industry. As a leading reliable aromatic azo compound supplier, understanding the nuances of this patent allows us to offer superior supply chain solutions that align with modern sustainability goals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of aromatic azo compounds has relied heavily on the diazo coupling reaction, a process fraught with inherent dangers and inefficiencies. As illustrated in the reaction schemes below, this traditional pathway necessitates the generation of stoichiometric amounts of diazonium salts, which are notoriously unstable and pose severe explosion hazards if not handled with extreme care. Furthermore, the process requires strict control over reaction parameters such as temperature and pH; deviations often lead to the formation of substantial by-products or complete reaction failure, resulting in poor atom economy. The reliance on large quantities of inorganic nitrites not only generates significant saline waste but also contradicts the principles of green chemistry, creating a heavy burden on wastewater treatment facilities. These factors collectively inflate the cost of goods sold and introduce unacceptable risks into the supply chain, making the conventional route increasingly untenable for modern high-volume manufacturing.
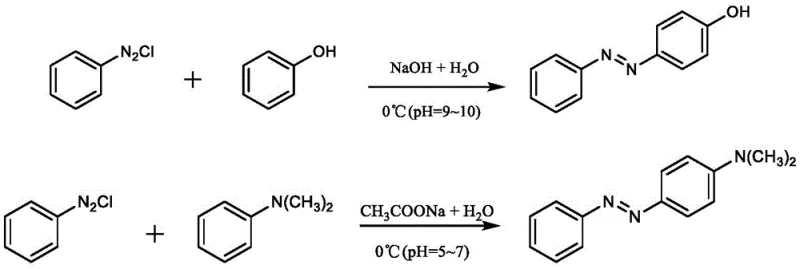
The Novel Approach
In stark contrast to these legacy methods, the photochemical catalytic synthesis described in the patent offers a transformative solution by utilizing light as a clean reagent to drive the transformation. This approach employs a sophisticated catalytic system comprising a semiconductor photocatalyst, such as titanium dioxide (TiO2), coupled with a nickel-based co-catalyst and specific ligands. Under irradiation, this system facilitates the selective deoxygenation of nitro groups, enabling their direct coupling to form the azo linkage without the intermediate formation of explosive diazonium species. The reaction proceeds under remarkably mild conditions, typically at ambient temperatures around 25°C, and tolerates a wide range of functional groups, thereby expanding the scope of accessible chemical space. This shift from thermal/chemical activation to photochemical activation not only enhances safety profiles drastically but also simplifies the downstream purification processes, offering a streamlined route to high-purity products.
Mechanistic Insights into TiO2-Ni Dual Catalytic Deoxygenation Coupling
The core of this technological breakthrough lies in the synergistic interaction between the heterogeneous photocatalyst and the homogeneous nickel co-catalyst. Upon irradiation with UV or visible light, the TiO2 semiconductor undergoes charge separation, generating electron-hole pairs that initiate the redox cycle. The photogenerated electrons are transferred to the nickel center, reducing the nitro-substituted aromatic substrate through a series of single-electron transfer steps. Crucially, the presence of a hydrogen donor, such as an alcohol or amine, serves as a sacrificial reagent to quench the holes and provide the necessary protons for the deoxygenation process. This mechanism avoids the non-selective oxidation often seen with bare TiO2, as the nickel complex directs the reactivity specifically towards the N-O bond cleavage and subsequent N-N bond formation. Understanding this mechanistic pathway is vital for R&D teams aiming to optimize reaction conditions for specific substrates, ensuring maximum efficiency and minimal degradation of sensitive functional groups.
Furthermore, the choice of ligands plays a pivotal role in stabilizing the active nickel species and modulating the electronic environment of the catalytic center. Ligands such as bipyridines or phenanthrolines coordinate with the nickel, preventing its aggregation and maintaining high catalytic turnover numbers throughout the reaction duration. This stabilization is essential for achieving the reported GC yields of over 85%, as it suppresses competing side reactions that could lead to impurity formation. From a quality control perspective, this high selectivity translates to a cleaner crude product, reducing the load on purification columns and crystallization steps. For manufacturers of electronic chemicals or pharmaceutical intermediates, where impurity profiles are strictly regulated, this level of mechanistic control provides a distinct competitive advantage, ensuring batch-to-batch consistency and compliance with stringent quality specifications.
How to Synthesize Aromatic Azo Compounds Efficiently
Implementing this photochemical protocol requires careful attention to the preparation of the reaction mixture and the management of light exposure. The process begins with the dissolution of the nitro-substituted aromatic compound, hydrogen donor, cocatalyst, ligand, and base in a suitable inert organic solvent such as methanol or acetonitrile. Once the homogeneous phase is established, the heterogeneous TiO2 photocatalyst is introduced, and the system is purged with inert gas to remove oxygen, which could otherwise quench the excited states or oxidize the intermediates. Detailed standard operating procedures regarding specific molar ratios, light source intensity, and irradiation times are critical for reproducibility. For a comprehensive guide on the exact experimental parameters and step-by-step execution, please refer to the standardized synthesis instructions provided below.
- Prepare the reaction mixture by dissolving nitro-substituted aromatic compounds, hydrogen donors, cocatalysts, ligands, and bases in an inert organic solvent.
- Add the heterogeneous photocatalyst (TiO2) to the transparent reactor and purge with inert gas to establish an oxygen-free environment.
- Irradiate the stirred mixture with UV or visible light sources (e.g., Xenon lamp or LED) at controlled temperatures (10-50°C) until conversion is complete.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photochemical synthesis route offers profound strategic benefits that extend beyond mere technical novelty. The elimination of unstable diazonium salts fundamentally alters the risk profile of the manufacturing facility, removing the need for specialized explosion-proof infrastructure and reducing insurance premiums associated with hazardous chemical storage. Moreover, the use of earth-abundant titanium dioxide as the primary photocatalyst ensures a stable and cost-effective supply of catalytic materials, insulating the production process from the volatility of precious metal markets. This robustness in raw material sourcing contributes significantly to supply chain resilience, ensuring continuous production even during periods of global logistical disruption. By transitioning to this greener methodology, companies can also align their procurement strategies with increasingly rigorous environmental, social, and governance (ESG) mandates, enhancing their corporate reputation among stakeholders.
- Cost Reduction in Manufacturing: The economic implications of this technology are substantial, primarily driven by the simplification of the synthetic route and the reduction of waste disposal costs. By avoiding the use of stoichiometric nitrites and the generation of large volumes of saline wastewater, the overall environmental compliance costs are drastically lowered. Additionally, the mild reaction conditions eliminate the need for energy-intensive heating or cryogenic cooling systems, leading to significant savings in utility consumption. The high selectivity of the reaction minimizes the loss of valuable starting materials to by-products, thereby improving the overall mass balance and reducing the cost per kilogram of the final active ingredient. These cumulative efficiencies result in a more competitive pricing structure without compromising on quality.
- Enhanced Supply Chain Reliability: The stability of the starting materials, specifically nitro-substituted aromatics, ensures a reliable feedstock supply compared to the transient nature of diazonium salts which must be generated in situ. This stability allows for better inventory management and reduces the risk of production stoppages due to reagent degradation. Furthermore, the scalability of the photochemical process is enhanced by the use of flow chemistry compatible setups, which can be easily expanded to meet increasing demand. The ability to utilize sunlight or standard LED arrays as energy sources also decentralizes the energy dependency, making the supply chain less vulnerable to local energy grid fluctuations. This reliability is crucial for maintaining just-in-time delivery schedules for downstream customers in the pharmaceutical and agrochemical sectors.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage levels is facilitated by the heterogeneous nature of the TiO2 catalyst, which can be readily separated via filtration and potentially regenerated for reuse. This ease of separation simplifies the work-up procedure, reducing the solvent usage and time required for isolation. From an environmental standpoint, the process adheres to the principles of green chemistry by minimizing hazardous substance use and maximizing atom economy. The reduction in toxic waste streams simplifies the permitting process for new manufacturing sites and ensures long-term operational continuity in regions with strict environmental regulations. This forward-looking approach future-proofs the manufacturing asset against tightening global emission standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photochemical synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and strategic value of the method for potential partners. Understanding these details is essential for making informed decisions about integrating this technology into existing production portfolios. We encourage technical teams to review these points thoroughly to assess the fit for their specific application requirements.
Q: What are the primary safety advantages of this photochemical method over traditional diazo coupling?
A: Unlike traditional methods that require unstable and potentially explosive diazonium salts, this photochemical route utilizes stable nitro-substituted aromatics, significantly reducing explosion risks and eliminating the need for hazardous nitrite reagents.
Q: Can this synthesis method be scaled for industrial production of electronic materials?
A: Yes, the process uses heterogeneous catalysts like TiO2 which are easily separable, and operates under mild conditions (25°C), making it highly suitable for commercial scale-up of complex functional materials like liquid crystals and optical storage media.
Q: What is the expected yield and purity profile for azobenzene derivatives using this route?
A: According to patent data, the GC yield can reach over 85% with high selectivity. The use of specific ligands and co-catalysts minimizes side reactions, ensuring a cleaner impurity profile suitable for high-tech applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Azo Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the photochemical catalytic synthesis method detailed in patent CN111039822B for the production of high-performance aromatic azo compounds. As a dedicated CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial reality is seamless. Our state-of-the-art facilities are equipped with advanced photochemical reactors capable of handling both batch and continuous flow processes, allowing us to optimize light penetration and reaction kinetics for maximum efficiency. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for pharmaceutical and electronic grade applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target cost structures. By partnering with us, you gain access to specific COA data and route feasibility assessments that demonstrate the tangible benefits of switching to this greener synthetic route. Let us help you engineer a supply chain that is not only cost-effective but also sustainable and resilient against future regulatory changes.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
