Scalable Synthetic Route for Leonurine: Enhancing Purity and Commercial Viability
Scalable Synthetic Route for Leonurine: Enhancing Purity and Commercial Viability
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for producing bioactive alkaloids that traditionally rely on inefficient botanical extraction. Patent CN102659638B introduces a transformative synthetic method for Leonurine, chemically known as syringic acid delta-guanidinobutyl ester, which serves as the primary active component in Motherwort Herb (Leonurus heterophyllus Sweet). This innovation addresses the critical bottleneck of low natural abundance, where extraction yields are typically below 0.1%, by establishing a fully synthetic pathway that utilizes cost-effective, semi-commercial starting materials. The disclosed technology not only stabilizes the supply chain for this gynecological therapeutic agent but also ensures a level of purity and consistency that natural extraction simply cannot guarantee, marking a significant advancement for reliable pharmaceutical intermediate supplier networks globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the procurement of syringic acid delta-guanidinobutyl ester has been plagued by severe inefficiencies inherent to botanical sourcing and early synthetic attempts. The natural content in Motherwort Herb is exceedingly low, necessitating the processing of massive quantities of plant material to isolate minute amounts of the active alkaloid, which drives costs prohibitively high and creates volatile supply dependencies on agricultural cycles. Furthermore, prior art synthetic methods, such as those utilizing methyl-isothiourea in DMF or complex multi-step protections involving ethoxycarbonyl intermediates, suffered from harsh reaction conditions, difficult-to-source reagents, and suboptimal overall yields. These legacy processes often resulted in products with inconsistent quality profiles, requiring extensive downstream purification that further eroded profit margins and delayed time-to-market for finished formulations.
The Novel Approach
The methodology outlined in the patent data revolutionizes this landscape by deploying a streamlined, six-step sequence that prioritizes atom economy and operational simplicity. By initiating the synthesis with readily available syringic acid and employing a strategic acetylation protection strategy, the process mitigates side reactions commonly associated with phenolic substrates. The subsequent esterification utilizes 4-chlorobutanol, a commodity chemical, catalyzed effectively by zinc chloride to form the carbon backbone with high fidelity. This approach eliminates the need for exotic reagents and allows for mild reaction temperatures, such as maintaining 60°C during the critical esterification phase, thereby reducing energy consumption and safety risks associated with high-pressure or high-temperature operations.
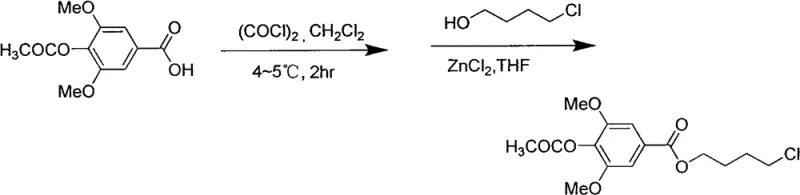
This initial phase sets the stage for a highly efficient introduction of the nitrogenous functionality. Unlike previous routes that struggled with regioselectivity or required cryogenic conditions, this novel pathway leverages standard organic solvents like dichloromethane and THF, which are easily recovered and recycled in a commercial setting. The transition from the acid chloride intermediate to the chloro-butyl ester is seamless, avoiding the isolation of unstable intermediates and thus minimizing material loss. This continuity is essential for cost reduction in pharmaceutical intermediates manufacturing, as it reduces unit operations and solvent handling requirements while maximizing the throughput of the reactor train.
Mechanistic Insights into FeCl3-Catalyzed Cyclization and Guanidinylation
While the initial esterification establishes the scaffold, the true chemical elegance of this process lies in the final functionalization steps involving the Gabriel synthesis modification and subsequent guanidinylation. The conversion of the chloro-intermediate to the phthalimide derivative proceeds via a classic nucleophilic substitution mechanism, where the phthalimide anion displaces the chloride leaving group. This step is crucial for introducing the nitrogen atom in a protected form, preventing premature polymerization or side reactions that free amines might undergo under acidic or basic conditions. The use of potassium phthalimide in a DMF-water mixture ensures optimal solubility and reactivity, driving the equilibrium towards the desired N-alkylated product with a reported yield of 67% in the exemplified embodiments.
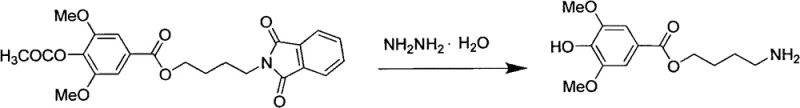
The deprotection and final coupling represent the pinnacle of this synthetic design, utilizing hydrazine hydrate to cleave the phthalimide group under reflux conditions to reveal the primary amine (Motherwort amine). This amine is then immediately subjected to guanidinylation using 1H-1,2,4-triazole-1-carboxamidine hydrochloride. The control of pH between 8 and 9 during this final step is mechanistically vital; it ensures the amine remains nucleophilic enough to attack the amidine reagent while preventing the hydrolysis of the sensitive ester linkage or the newly formed guanidine moiety. The result is a crystalline product with a sharp melting point of 215-216°C and HPLC purity exceeding 99.5%, demonstrating exceptional impurity control mechanisms that are critical for regulatory compliance in high-purity pharmaceutical intermediates.
How to Synthesize Leonurine Efficiently
The execution of this synthetic route requires precise adherence to the stoichiometric ratios and thermal profiles detailed in the patent embodiments to ensure reproducibility at scale. Operators must carefully manage the exothermic nature of the acetylation and acid chloride formation steps, utilizing ice baths to maintain temperatures near 0-5°C where specified to prevent degradation of the acid chloride intermediate. The transition to the esterification phase involves the addition of solid ZnCl2 in THF, which requires thorough stirring to ensure homogeneous catalysis throughout the reaction mass. For the final guanidinylation, the slow addition of the triazole reagent under strict pH monitoring is non-negotiable to achieve the high purity standards demanded by modern pharmacopeias.
- Acetylate syringic acid using acetic anhydride and acid catalyst to protect the phenolic hydroxyl group.
- Convert acetyl syringic acid to acid chloride using oxalyl chloride, then esterify with 4-chlorobutanol using ZnCl2 catalyst.
- Perform nucleophilic substitution with potassium phthalimide, followed by hydrazine hydrate deprotection to reveal the primary amine.
- React the amine intermediate with 1H-1,2,4-triazole-1-carboxamidine hydrochloride under controlled pH to form the final guanidine structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this synthetic methodology offers profound advantages that directly address the pain points of procurement managers and supply chain directors. By shifting the source of Leonurine from an agricultural extract to a chemical synthesis, organizations can decouple their supply from seasonal variations, crop failures, and geopolitical instability affecting herbal raw material exports. The reliance on syringic acid and 4-chlorobutanol, both of which are produced in large volumes for other industrial applications, ensures a stable and predictable pricing structure. This stability is paramount for long-term contract planning and budget forecasting, allowing companies to secure reliable supply chains without the risk of sudden price spikes associated with niche botanical extracts.
- Cost Reduction in Manufacturing: The economic rationale for adopting this process is compelling, primarily driven by the elimination of expensive and scarce reagents found in prior art. The substitution of complex protecting group strategies with a straightforward acetylation and phthalimide approach significantly lowers the bill of materials. Furthermore, the ability to perform reactions in common solvents like dichloromethane and ethanol, which are easily recoverable via distillation, drastically reduces waste disposal costs and solvent procurement expenses. The high overall yield reported in the embodiments implies less raw material waste per kilogram of final product, translating to substantial cost savings in commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of this chemical route enhances supply security by removing the dependency on the cultivation and harvesting of Motherwort Herb. Chemical synthesis can be performed year-round in controlled reactor environments, unaffected by weather patterns or agricultural pests. The use of standard unit operations—such as filtration, recrystallization, and liquid-liquid extraction—means that the process can be easily transferred between different manufacturing sites or CDMO partners without requiring specialized or custom-built equipment. This flexibility ensures reducing lead time for high-purity pharmaceutical intermediates and guarantees continuity of supply even in the face of regional disruptions.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scalability, utilizing reaction conditions that are safe and manageable in large-scale vessels. The avoidance of heavy metal catalysts or highly toxic reagents simplifies the environmental, health, and safety (EHS) profile of the manufacturing site. Waste streams are primarily aqueous salts and organic solvents that can be treated using standard effluent treatment protocols, ensuring compliance with increasingly stringent environmental regulations. This 'green' aspect of the synthesis not only reduces regulatory risk but also aligns with the sustainability goals of modern pharmaceutical companies, making it an attractive option for eco-conscious procurement strategies.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and quality assurance of this synthetic Leonurine process. These answers are derived directly from the experimental data and claims within the patent documentation, providing a transparent view of the technology's capabilities for potential partners and technical evaluators.
Q: What is the purity level achievable with this synthetic method?
A: The patented process demonstrates the capability to achieve HPLC purity levels of 99.5% through optimized recrystallization steps, specifically using ethanol for the final purification of the guanidine product.
Q: How does this method improve upon traditional extraction from Motherwort Herb?
A: Traditional extraction yields less than 0.1% content with difficult separation. This synthetic route utilizes inexpensive, semi-commercial raw materials like syringic acid and 4-chlorobutanol, significantly boosting overall yield and ensuring consistent supply independent of agricultural harvests.
Q: Is the process suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly states the method is suitable for industrialized mass production. It employs mild reaction conditions (e.g., 60°C for esterification) and avoids hazardous or prohibitively expensive reagents, facilitating safe scale-up from pilot to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Leonurine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable supply of high-quality active ingredients for the global healthcare market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering Leonurine and related pharmaceutical intermediates with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch against the highest international standards.
We invite you to collaborate with us to leverage this advanced synthetic technology for your product pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing expertise can optimize your supply chain and drive value for your organization.
