Advanced Manufacturing Strategy for High-Purity Leonurine via Optimized Esterification and Guanidinylation
The pharmaceutical industry continuously seeks robust manufacturing pathways for bioactive natural products that can be produced at scale with consistent quality. Patent CN102659639B presents a significant advancement in the preparation technology of Leonurine, also known as syringic acid delta-guanidinobutyl ester, a key alkaloid found in Motherwort Herb (Leonurus heterophyllus Sweet). Historically, obtaining this compound relied heavily on extraction from plant sources, a method plagued by extremely low natural content (below 0.1%) and complex separation challenges. This patent introduces a rational, economical, and stable synthetic route that addresses these critical bottlenecks. By utilizing readily available starting materials such as syringic acid and employing mild reaction conditions, the technology ensures a substantial improvement in both overall yield and product quality. For R&D directors and procurement specialists, this represents a viable transition from unreliable botanical extraction to a controlled chemical manufacturing process, ensuring a reliable pharmaceutical intermediates supplier can meet the growing demand for gynecological therapeutic agents without the volatility of agricultural supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for acquiring Leonurine have been fundamentally constrained by the biological limitations of the source material. As documented in the background art, the content of syringic acid delta-guanidinobutyl ester in Motherwort Herb is negligible, typically falling below 0.1%, which renders large-scale extraction economically prohibitive due to the massive volume of biomass required and the intricate purification steps needed to isolate the active alkaloid from complex plant matrices. Furthermore, existing synthetic methodologies disclosed in prior literature, such as those utilizing methyl-isothiourea in DMF or multi-step sequences involving phthalimide protection, suffer from significant drawbacks including the use of expensive or difficult-to-obtain starting materials, harsh reaction conditions, and low overall productivity. These legacy processes often result in poor cost reduction in pharmaceutical intermediates manufacturing because they require specialized reagents and generate substantial waste, making them unsuitable for the rigorous demands of modern commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in CN102659639B offers a streamlined and economically rational pathway that leverages cheap, semi-commercial raw materials to drive efficiency. The novel approach initiates with the protection of syringic acid followed by a strategic acylation and amidation sequence, culminating in a highly selective guanidinylation step. This route is characterized by mild reaction temperatures and the use of common solvents like ethanol and dichloromethane, which simplifies process engineering and safety protocols. By optimizing the synthetic sequence to minimize side reactions and facilitate easy purification through recrystallization, this technology effectively solves the problems of low yield and high cost associated with prior art. It provides a clear pathway for reducing lead time for high-purity pharmaceutical intermediates by establishing a reproducible chemical synthesis that is not dependent on seasonal crop yields or variable botanical potency.
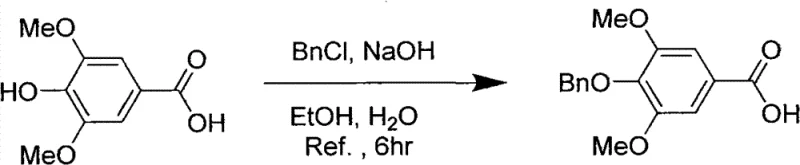
Mechanistic Insights into the Multi-Step Synthesis of Leonurine
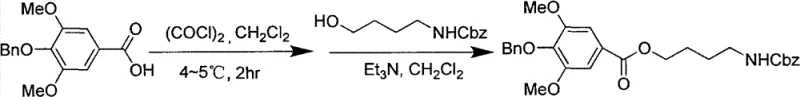
The core of this manufacturing strategy lies in a carefully orchestrated five-step sequence that balances reactivity with selectivity. The process begins with the O-alkylation of syringic acid using benzyl chloride in an alkaline ethanol solution under reflux. This step serves a dual purpose: it protects the phenolic hydroxyl group to prevent unwanted side reactions during subsequent acylation, and it converts the acid into a more manageable intermediate. Following this, the protected acid is activated using oxalyl chloride in dichloromethane at low temperatures (4-5°C) to form the corresponding acyl chloride. This activation is crucial for the next coupling reaction, where the acyl chloride reacts with N-Cbz-4-aminobutanol in the presence of triethylamine. This amidation step constructs the critical ester-amide backbone of the molecule while maintaining the integrity of the amine protecting group, ensuring that the final guanidinylation occurs selectively at the desired position.
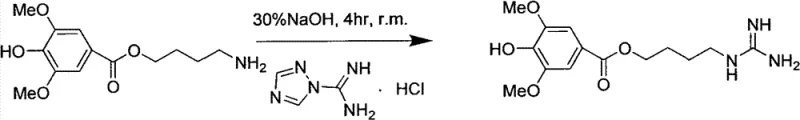
The final stages of the synthesis involve the removal of protecting groups and the installation of the guanidine moiety. Catalytic hydrogenation using 10% Pd/C under normal pressure and room temperature efficiently cleaves both the benzyl ether and the Cbz carbamate protecting groups, revealing the free phenolic hydroxyl and the primary amine simultaneously. This deprotection strategy is elegant in its simplicity, avoiding the need for harsh acidic or basic hydrolysis conditions that could degrade the sensitive ester linkage. The final transformation involves reacting the liberated amine with 1,2,4-triazole-1-amidine hydrochloride in an alkaline solution. The mechanism here relies on the nucleophilic attack of the amine on the amidine carbon, facilitated by the basic environment which maintains the amine in its unprotonated, nucleophilic form. Strict pH control (pH 8-9) during this step is essential to maximize conversion while minimizing hydrolysis of the ester bond, ultimately yielding the target Leonurine with high purity after recrystallization.
How to Synthesize Leonurine Efficiently
The synthesis of Leonurine described in this patent offers a practical guide for laboratory and pilot-scale production, emphasizing operational simplicity and safety. The procedure utilizes standard organic synthesis techniques such as reflux, low-temperature addition, and catalytic hydrogenation, making it accessible for most chemical manufacturing facilities equipped with basic reactor systems. The detailed standardized synthesis steps below outline the precise stoichiometry, solvent choices, and workup procedures required to achieve the reported yields and purity levels, serving as a foundational protocol for process chemists aiming to implement this technology.
- Protect syringic acid by reacting with benzyl chloride in alkaline ethanol solution under reflux to form benzyl syringate.
- Convert benzyl syringate to its acyl chloride derivative using oxalyl chloride in dichloromethane at low temperature.
- React the acyl chloride with N-Cbz-4-aminobutanol in the presence of triethylamine to form the protected intermediate.
- Perform catalytic hydrogenation using Pd/C to remove the benzyl and Cbz protecting groups, yielding the free amine.
- React the free amine with 1,2,4-triazole-1-amidine hydrochloride in alkaline solution to finalize the guanidine structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers profound strategic advantages over traditional extraction or older synthetic methods. The primary benefit stems from the utilization of inexpensive and widely available starting materials, such as syringic acid and benzyl chloride, which decouples production costs from the volatility of agricultural markets. This shift ensures a stable supply chain reliability, as the manufacturing process is no longer subject to the risks of crop failure, seasonal variations, or geopolitical issues affecting herbal sourcing. Furthermore, the simplified purification process, which relies on standard recrystallization rather than complex chromatography, drastically reduces processing time and solvent consumption, leading to substantial cost savings in manufacturing operations.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of expensive reagents and the optimization of reaction yields. By avoiding the use of precious metal catalysts (other than recyclable Pd/C) and utilizing common solvents like ethanol and dichloromethane, the direct material costs are significantly lowered. The high overall yield reported in the embodiments implies that less raw material is wasted per kilogram of final product, which directly translates to a lower cost of goods sold (COGS). Additionally, the ability to perform reactions at ambient pressure and moderate temperatures reduces energy consumption and equipment stress, further enhancing the economic efficiency of the production line.
- Enhanced Supply Chain Reliability: Transitioning to a fully synthetic route mitigates the supply risks inherent in botanical extraction. Since the raw materials are commodity chemicals with established global supply networks, procurement teams can secure long-term contracts with multiple vendors, ensuring continuity of supply even during market disruptions. The robustness of the synthetic process also means that production schedules can be forecasted with greater accuracy, allowing for better inventory management and the ability to respond rapidly to spikes in demand for Leonurine-based therapeutics without the long lead times associated with farming and harvesting cycles.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing unit operations that are easily transferred from the laboratory to multi-ton reactors. The use of mild reaction conditions minimizes the formation of hazardous byproducts, simplifying waste treatment and ensuring compliance with increasingly stringent environmental regulations. The efficient atom economy of the coupling steps and the recyclability of the hydrogenation catalyst contribute to a greener manufacturing profile, aligning with the sustainability goals of modern pharmaceutical companies and reducing the environmental footprint of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Leonurine using this patented technology. These answers are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on yield expectations, purity standards, and operational parameters for potential manufacturing partners.
Q: Why is synthetic Leonurine preferred over natural extraction?
A: Natural extraction from Motherwort Herb yields less than 0.1% Leonurine, making it economically unviable and difficult to purify. Synthetic routes offer significantly higher yields and consistent quality.
Q: What are the key advantages of the patented CN102659639B process?
A: The process utilizes cheap, commercially available raw materials like syringic acid and employs mild reaction conditions. It avoids complex purification steps, resulting in improved overall yield and product purity suitable for industrial production.
Q: How is high purity achieved in the final step?
A: High purity (HPLC >99.5%) is achieved through controlled pH during the guanidinylation reaction followed by recrystallization from ethanol, which effectively removes impurities and byproducts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Leonurine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for the production of high-value pharmaceutical intermediates like Leonurine. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale optimization to full-scale manufacturing is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch meets the highest international standards, providing our clients with the confidence needed to advance their drug development programs.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your project's success and reduce your time to market.
