Advanced Synthetic Strategy for Leonurine: Enhancing Purity and Scalability for Global Pharmaceutical Supply Chains
Advanced Synthetic Strategy for Leonurine: Enhancing Purity and Scalability for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust manufacturing pathways for bioactive natural products, and the synthesis of Leonurine (syringic acid delta-guanidinobutyl ester) represents a critical area of development for gynecological therapeutics. Patent CN102659638B discloses a novel synthetic method that addresses the longstanding economic and technical bottlenecks associated with producing this key alkaloid found in Motherwort Herb. Unlike traditional extraction methods which suffer from extremely low content levels below 0.1% in plant sources, this chemical synthesis route offers a viable alternative for securing high-purity supply. The disclosed process leverages a rational design starting from syringic acid, utilizing acetylation, acyl chloride activation, and a specialized zinc-catalyzed esterification to construct the carbon backbone efficiently. For R&D directors and procurement specialists, this patent signifies a shift towards more predictable and cost-stable manufacturing of complex pharmaceutical intermediates, ensuring that the supply chain for Motherwort-derived medications remains resilient against agricultural variability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of syringic acid delta-guanidinobutyl ester has been plagued by significant technical hurdles that hindered its widespread clinical application in refined formulations. Prior art methods, such as those described in early literature from 1969 or Chinese patent CN1415602A, often relied on starting materials that were either prohibitively expensive or difficult to source in bulk quantities, creating fragile supply chains. For instance, some routes utilized 4-ethoxycarbonyl-oxygen base-3,5-dimethoxy-benzoic acid derivatives which required complex multi-step preparations before the core structure could even be assembled. Furthermore, older synthetic strategies frequently encountered issues with low overall yields and harsh reaction conditions that necessitated specialized equipment and rigorous safety protocols, driving up the operational expenditure for manufacturers. The reliance on extraction from natural sources was equally problematic, as the low concentration of the active alkaloid in the plant material resulted in massive solvent consumption and extensive purification burdens, making it economically unfeasible to produce the high volumes required for modern pharmaceutical dosages.
The Novel Approach
The methodology outlined in CN102659638B introduces a streamlined and economically rational pathway that fundamentally restructures the synthesis logic to maximize efficiency and minimize waste. By selecting syringic acid as the primary building block, the process capitalizes on the availability and low cost of this semi-commercial raw material, immediately lowering the entry barrier for production. The innovation lies in the strategic use of acetic anhydride for protection and oxalyl chloride for activation, followed by a highly efficient esterification step catalyzed by zinc chloride in tetrahydrofuran. This approach not only simplifies the operational workflow by reducing the number of isolation steps but also significantly improves the reaction kinetics, allowing for milder temperatures that preserve the integrity of the sensitive functional groups. The result is a robust process capable of delivering high-quality intermediates consistently, solving the prior art problems of instability and low productivity while establishing a foundation for reliable large-scale manufacturing of this valuable therapeutic agent.
Mechanistic Insights into ZnCl2-Catalyzed Esterification and Guanidinylation
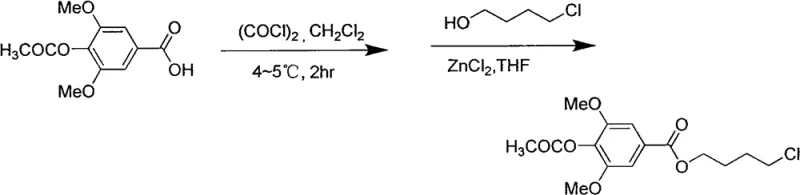
The core chemical transformation in this synthetic route involves the precise construction of the ester linkage between the aromatic acid moiety and the aliphatic amine precursor, a step that is critically enabled by Lewis acid catalysis. In the second and third steps of the process, acetyl syringic acid is first converted into its corresponding acyl chloride using oxalyl chloride in dichloromethane, generating a highly reactive electrophile. This intermediate is then subjected to nucleophilic attack by 4-chloro-1-butanol in the presence of anhydrous zinc chloride (ZnCl2) within a THF solvent system. The zinc chloride acts as a potent Lewis acid, coordinating with the carbonyl oxygen to enhance the electrophilicity of the acyl carbon, thereby facilitating the esterification under relatively mild heating conditions around 60°C. This catalytic cycle ensures high conversion rates and minimizes the formation of hydrolysis byproducts, which is essential for maintaining the purity profile required for downstream pharmaceutical processing.
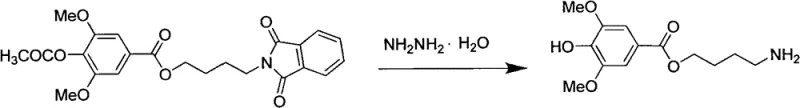
Following the successful assembly of the ester backbone, the synthetic strategy employs a classic Gabriel synthesis modification to introduce the nitrogen functionality with high regioselectivity. The chloro-group on the butyl chain is displaced by potassium phthalimide in a polar aprotic solvent like DMF, effectively protecting the amine during subsequent manipulations. The final stages involve the removal of the phthalimide protecting group using hydrazine hydrate under reflux conditions to liberate the primary amine, followed by the crucial guanidinylation reaction. This final coupling with 1H-1,2,4-triazole-1-carboxamidine in an alkaline medium allows for the formation of the guanidine moiety under controlled pH conditions, typically between 8 and 9. The mechanistic precision of these steps ensures that the final product, Leonurine, is obtained with exceptional purity, as evidenced by HPLC analysis showing levels up to 99.5%, demonstrating the efficacy of this protective group strategy in suppressing side reactions.
How to Synthesize Leonurine Efficiently
The implementation of this synthetic protocol requires careful attention to reaction parameters and purification techniques to replicate the high yields reported in the patent documentation. The process begins with the acetylation of syringic acid, followed by activation and esterification, which sets the stage for the introduction of the nitrogenous side chain. Operators must maintain strict control over temperature and stoichiometry, particularly during the exothermic addition of oxalyl chloride and the subsequent zinc-catalyzed coupling. The detailed standardized synthesis steps, including specific solvent volumes, reaction times, and workup procedures such as recrystallization from normal hexane and ethanol, are critical for achieving the reported 81% yield in the final step. For a comprehensive guide on executing this chemistry in a GMP environment, please refer to the structured protocol below.
- Acetylate syringic acid using acetic anhydride and acid catalyst to form acetyl syringic acid.
- Convert acetyl syringic acid to acyl chloride using oxalyl chloride, followed by ZnCl2-catalyzed esterification with 4-chloro-1-butanol.
- Substitute the chloride with potassium phthalimide, deprotect with hydrazine hydrate, and react with 1H-1,2,4-triazole-1-carboxamidine to yield Leonurine.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic methodology offers profound advantages for procurement managers and supply chain directors seeking to optimize the cost structure of pharmaceutical intermediate sourcing. The primary driver of value creation is the substitution of scarce or expensive starting materials with commoditized chemicals like syringic acid, acetic anhydride, and 4-chloro-1-butanol, which are readily available in the global market. This shift drastically reduces the raw material cost basis and mitigates the risk of supply disruptions associated with niche reagents. Furthermore, the high overall yield of the process means that less raw material is wasted per kilogram of finished product, directly translating to improved material efficiency and lower waste disposal costs. The ability to produce high-purity material without resorting to complex chromatographic purification also reduces the consumption of expensive silica gel and solvents, contributing to a leaner and more sustainable manufacturing operation.
- Cost Reduction in Manufacturing: The elimination of costly transition metal catalysts and the use of inexpensive reagents like oxalyl chloride and hydrazine hydrate significantly lowers the variable cost of production. By avoiding the need for precious metal scavengers or complex downstream purification trains, the process simplifies the manufacturing workflow, leading to substantial operational savings. The high yield at each step, particularly the 89% yield in the initial acetylation and 81% in the final guanidinylation, ensures that the throughput of the facility is maximized, spreading fixed costs over a larger volume of output and enhancing the overall economic viability of the project.
- Enhanced Supply Chain Reliability: Sourcing reliability is greatly improved because the key inputs are bulk chemicals with established global supply networks, reducing dependency on single-source suppliers of exotic intermediates. The robustness of the reaction conditions, which tolerate standard industrial equipment and do not require cryogenic temperatures or ultra-high vacuum, means that production can be easily transferred between different manufacturing sites without significant requalification delays. This flexibility ensures business continuity and allows for rapid scaling in response to market demand fluctuations, securing the supply chain against external shocks.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing common organic solvents like dichloromethane, THF, and ethanol that can be efficiently recovered and recycled in a closed-loop system. The reduction in reaction steps and the avoidance of heavy metal contaminants simplify the environmental compliance burden, making it easier to meet stringent regulatory standards for wastewater and emissions. The solid-state nature of several intermediates allows for efficient isolation via filtration rather than energy-intensive distillation, further reducing the carbon footprint of the manufacturing process and aligning with modern green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Leonurine synthesis technology, derived directly from the patent specifications and process data. These insights are intended to clarify the feasibility of the route for potential licensees or contract manufacturing partners, highlighting the specific operational parameters that define its success. Understanding these details is crucial for technical teams evaluating the fit of this process within their existing infrastructure.
Q: What are the key advantages of the synthetic method in CN102659638B compared to prior art?
A: The method utilizes cheap, commercially available raw materials like syringic acid and 4-chloro-1-butanol, avoiding expensive precursors. It features mild reaction conditions and achieves high overall yields with product purity reaching 99.5% via HPLC.
Q: How does the process ensure high purity for pharmaceutical applications?
A: The route employs a phthalimide protection strategy which minimizes side reactions during alkylation. Subsequent recrystallization steps, particularly the final ethyl alcohol recrystallization, effectively remove impurities to meet stringent pharmacopoeia standards.
Q: Is this synthetic route suitable for industrial scale-up?
A: Yes, the patent explicitly states suitability for industrialized mass production. The use of common solvents like dichloromethane, THF, and ethanol, along with standard unit operations like filtration and recrystallization, facilitates easy technology transfer to large-scale reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Leonurine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of securing a stable and high-quality supply of critical pharmaceutical intermediates like Leonurine for the global healthcare market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to verify stringent purity specifications, guaranteeing that every batch meets the highest pharmacopoeial standards required for drug substance manufacturing. We are committed to leveraging our technical expertise to optimize this patented route, delivering cost-effective solutions that empower your drug development programs.
We invite you to collaborate with us to explore the full potential of this synthetic technology for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized process can reduce your total cost of ownership. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how NINGBO INNO PHARMCHEM can become your trusted partner in bringing high-value therapeutic agents to market efficiently.
