Advanced Synthesis of Alvimopan Intermediates for Commercial API Production
Advanced Synthesis of Alvimopan Intermediates for Commercial API Production
The pharmaceutical landscape for gastrointestinal therapeutics has been significantly shaped by the development of selective peripheral mu-opioid receptor antagonists, most notably Alvimopan (marketed as Entereg). As demand for this critical API grows, the efficiency of its supply chain becomes paramount for global health stakeholders. Patent CN102127005A discloses a groundbreaking synthetic methodology that addresses long-standing challenges in the production of Alvimopan intermediates. This technical insight report analyzes the novel route, which leverages chiral oxazolidinone auxiliaries to achieve superior stereocontrol and process efficiency. By fundamentally restructuring the synthetic sequence, this approach offers a robust pathway for reliable alvimopan intermediate supplier partnerships, ensuring consistent quality and supply continuity for downstream API manufacturers.
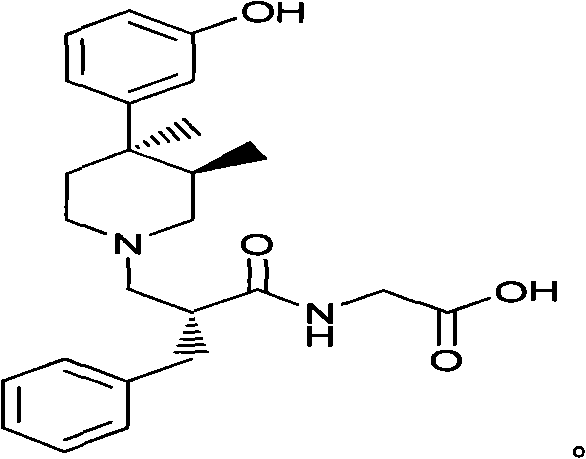
Alvimopan is a highly selective antagonist approved for accelerating upper and lower gastrointestinal recovery following bowel resection surgery. The molecule's complexity, featuring multiple chiral centers and sensitive functional groups, necessitates a synthesis strategy that balances precision with economic viability. The disclosed invention provides not only a new synthesis method but also identifies novel intermediates that serve as critical checkpoints for quality assurance. For procurement leaders, understanding the mechanistic advantages of this route is essential for evaluating cost reduction in API manufacturing and securing a stable supply of high-purity precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthesis routes, such as those disclosed in earlier patents like CN1065455, typically introduce the complex chiral piperidine moiety, specifically (+)-(3R,4R)-4-(3-hydroxyphenyl)-3,4-dimethylpiperidine, at a very early stage of the synthesis. This premature introduction creates significant process vulnerabilities. When the chiral center is established or reacted upon in the presence of the sensitive piperidine ring, there is a high risk of generating diastereoisomers due to a lack of rigorous chiral control during subsequent transformations. These unwanted isomers often possess similar physical properties to the desired product, making their removal extremely difficult and requiring resource-intensive purification techniques like column chromatography or resolution.
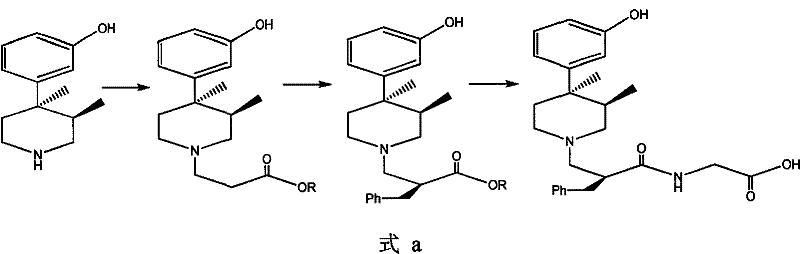
Furthermore, the wastage of the expensive chiral piperidine starting material is a major economic drawback. If a reaction step fails to maintain stereochemical integrity or yields a mixture of isomers, the high-value piperidine component attached to the failed batch is essentially lost or requires costly recovery processes. This inefficiency inflates the cost of goods sold (COGS) and introduces variability into the supply chain. For supply chain heads, relying on such inefficient routes poses a risk to commercial scale-up of complex pharmaceutical intermediates, as yield losses and purification bottlenecks can severely impact delivery timelines and overall production capacity.
The Novel Approach
The innovative strategy presented in CN102127005A fundamentally reverses this logic by employing a "late-stage coupling" approach. Instead of starting with the expensive piperidine, the synthesis begins with readily available and inexpensive achiral or singly-chiral building blocks, specifically utilizing phenylpropionic acid and a chiral oxazolidinone auxiliary. This allows for the precise construction of the chiral side chain with high fidelity before the valuable piperidine is ever introduced. By deferring the coupling of the piperidine to the final stages (specifically the reaction of intermediate VIII with the piperidine), the process ensures that the expensive chiral amine is only exposed to mild, high-yielding conditions.
This strategic shift drastically simplifies the purification profile. Since the chiral integrity is locked in by the robust oxazolidinone chemistry early on, the formation of diastereomers is minimized. Consequently, the need for complex chromatographic separations is eliminated, replaced by standard crystallization and extraction techniques that are far more amenable to large-scale manufacturing. This results in a process that is not only chemically superior but also economically optimized, directly supporting reducing lead time for high-purity pharmaceutical intermediates by streamlining the production workflow and minimizing batch failure rates associated with difficult purifications.
Mechanistic Insights into Chiral Auxiliary-Mediated Alkylation
The core of this synthetic breakthrough lies in the use of Evans chiral auxiliaries to dictate stereochemistry. The process initiates with the condensation of phenylpropionic acid with an R-configuration 3-substituted oxazolidinone to form a chiral imide (Compound II). This imide serves as a rigid template that controls the facial selectivity of subsequent reactions. In the critical alkylation step, the enolate of the imide is generated and reacted with chloromethyl benzyl ether under the catalytic action of titanium tetrachloride (TiCl4). The Lewis acid catalyst coordinates with the carbonyl oxygens, locking the conformation and ensuring that the alkyl group attacks from the less hindered face, thereby establishing the (S)-configuration at the alpha-carbon with high enantiomeric excess.
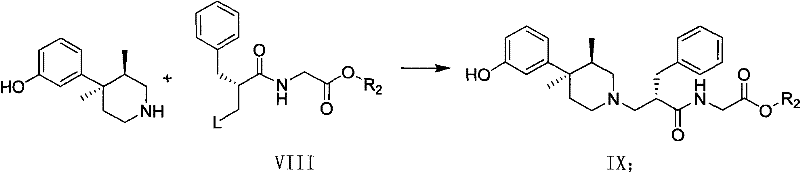
Following the establishment of the chiral center, the auxiliary is hydrolyzed and recycled, yielding the free carboxylic acid (Compound IV) which retains the induced chirality. This acid is then condensed with glycine ester to form the peptide bond, followed by deprotection of the benzyl group to reveal the primary alcohol. The alcohol is subsequently converted into a good leaving group, such as a mesylate (Compound VIII), activating it for nucleophilic substitution. The final coupling involves the reaction of this activated intermediate with the chiral piperidine. Because the electrophile (VIII) is already chirally pure, the nucleophilic attack by the piperidine proceeds cleanly to form the Alvimopan ester (IX) without generating significant diastereomeric impurities, ensuring the final hydrolysis yields Alvimopan of exceptional purity.
Impurity control is inherently built into this mechanism. The use of the oxazolidinone auxiliary acts as a "chiral filter," preventing the formation of the wrong enantiomer at the source rather than trying to remove it later. Additionally, the choice of reagents, such as using mesylates instead of more reactive halides, minimizes side reactions like elimination or over-alkylation. The patent data indicates that the intermediate IX hydrochloride formed is a new crystal form that can be directly filtered, further enhancing purity by excluding soluble impurities before the final hydrolysis step. This rigorous control over the reaction pathway is critical for R&D directors focused on maintaining a clean impurity profile for regulatory filings.
How to Synthesize Alvimopan Intermediate Efficiently
The synthesis of the key intermediate VIII1 involves a sequence of highly controlled transformations designed to maximize yield and stereochemical purity. The process begins with the activation of phenylpropionic acid and its coupling with a chiral oxazolidinone, followed by a titanium-mediated alkylation to set the stereocenter. Subsequent steps involve hydrolysis to remove the auxiliary, amidation with glycine ester, hydrogenolytic deprotection, and finally mesylation to activate the chain for coupling. Detailed operational parameters, including specific solvent choices like THF and dichloromethane, temperature controls ranging from ice bath conditions to reflux, and precise stoichiometric ratios, are critical for success. The standardized synthesis steps outlined below provide a roadmap for replicating this high-efficiency route in a GMP environment.
- Condense phenylpropionic acid with R-configuration phenyl substituted oxazolidinone to form the chiral imide intermediate.
- Perform titanium tetrachloride catalyzed alkylation with chloromethyl benzyl ether to establish the second chiral center.
- Hydrolyze the auxiliary, couple with glycine ester, deprotect, and mesylate to obtain the electrophilic intermediate ready for piperidine coupling.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this novel synthetic route translates into tangible operational benefits that extend beyond simple chemistry. The primary advantage lies in the decoupling of expensive raw material consumption from process risk. By utilizing cheap, commodity-grade starting materials like phenylpropionic acid for the bulk of the synthesis, the financial exposure per batch is significantly lowered. The expensive chiral piperidine is reserved for the final step, where the intermediate is already highly purified, ensuring that this costly component is not wasted on off-spec intermediates. This structural change in the bill of materials (BOM) drives substantial cost savings and improves margin stability for the final API.
- Cost Reduction in Manufacturing: The elimination of column chromatography and resolution steps represents a massive reduction in processing costs. Traditional methods often require silica gel columns and large volumes of solvents to separate diastereomers, which is both expensive and environmentally burdensome. The new route relies on crystallization and extraction, which are scalable and cost-effective unit operations. Furthermore, the recycling of the oxazolidinone auxiliary adds another layer of economic efficiency, reducing the net consumption of chiral reagents. These factors combined result in a significantly lower cost of production, allowing for more competitive pricing in the global market without compromising quality standards.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials mitigates the risk of supply disruptions. Phenylpropionic acid and glycine esters are commodity chemicals with robust global supply chains, unlike specialized chiral building blocks that may have limited suppliers. By shifting the synthesis to rely on these stable inputs, manufacturers can secure long-term supply agreements with greater confidence. Additionally, the simplified process flow reduces the number of potential failure points in the manufacturing line, leading to higher batch success rates and more predictable delivery schedules for downstream partners relying on a reliable alvimopan intermediate supplier.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial production, utilizing solvents and conditions that are manageable at multi-ton scales. The avoidance of heavy metal catalysts (using Titanium which is easier to manage than some alternatives, or avoiding them where possible) and the reduction of solvent waste through streamlined purification align with modern green chemistry principles. This facilitates easier regulatory approval and reduces the environmental footprint of the manufacturing site. The ability to scale from kilogram to hundred-ton quantities while maintaining high purity (demonstrated at 99.6% in examples) ensures that the supply chain can flexibly respond to market demand surges for Alvimopan.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the technical specifications and experimental data provided in patent CN102127005A, offering clarity on the feasibility and advantages of the method for potential manufacturing partners and technical evaluators.
Q: How does this new route improve chiral purity compared to prior art?
A: The novel route utilizes an Evans auxiliary (oxazolidinone) to strictly control stereochemistry early in the synthesis, avoiding the formation of difficult-to-separate diastereoisomers that plague earlier methods where the chiral piperidine was introduced too soon.
Q: What are the cost advantages of this synthesis method?
A: By delaying the introduction of the expensive chiral piperidine raw material until the final coupling steps, the process prevents the waste of this high-value starting material on failed stereocenters, significantly reducing overall raw material costs.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly states the method is suitable for industrial production due to the use of readily available starting materials, simple reaction conditions, and the ability to achieve high purity (over 99%) without complex chromatographic separations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alvimopan Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team specializes in the commercial scale-up of complex pharmaceutical intermediates, leveraging advanced process chemistry to optimize routes like the one described in CN102127005A. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel synthesis are fully realized in a GMP-compliant setting. Our stringent purity specifications and rigorous QC labs guarantee that every batch of Alvimopan intermediate meets the highest global standards for safety and efficacy.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to unlock the full potential of this efficient synthesis route. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance your supply chain resilience and drive down the overall cost of Alvimopan production.
