Scalable Alvimopan Production via Mild N-Alkylation and Salification Purification
Scalable Alvimopan Production via Mild N-Alkylation and Salification Purification
The pharmaceutical industry continuously seeks robust, scalable pathways for complex opioid antagonists like Alvimopan, particularly for managing postoperative ileus. Patent CN101967118A introduces a transformative preparation method that fundamentally restructures the synthetic approach to this critical API intermediate. Unlike traditional routes that rely on hazardous cryogenic conditions or dangerous hydrogenation steps, this invention utilizes a sophisticated two-chain convergent strategy. By designing a process where initial raw materials are synthesized separately and then condensed, the method achieves directional synthesis of a single optical isomer through a mild N-alkylation reaction under alkaline conditions. This technical breakthrough ensures that the configuration of chiral centers remains intact throughout both the reaction and post-processing phases, addressing a long-standing challenge in stereochemical control. Furthermore, the final product can be hydrolyzed under gentle alkaline conditions, bypassing the need for harsh acidic environments that often compromise product integrity. For global procurement teams and R&D directors, this patent represents a pivotal shift towards safer, more cost-effective manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Alvimopan has been plagued by significant operational hurdles that hinder industrial scalability. Early methods, such as those disclosed in Chinese Patent ZL 92102213.1, relied on a five-step sequence involving Michael addition and chiral column chromatography. While the reaction steps were relatively short, the process generated "soup" compounds that were exceptionally difficult to separate and purify. The reliance on chiral separation for every intermediate resulted in low yields and exorbitant costs, rendering the method unsuitable for large-scale production. Subsequent improvements, like the 1996 JOC method, attempted to streamline the process but introduced severe safety and infrastructure liabilities. This route necessitated the use of diisopropyl lithium as a basifying agent, requiring strict anhydrous conditions and cryogenic temperatures ranging from -50°C to -40°C. Such extreme conditions demand specialized, energy-intensive equipment and nitrogen protection systems, drastically increasing capital expenditure. Additionally, other approaches involving benzyl esters required catalytic hydrogenation with 10% Pd/C, introducing potential explosion hazards and the risk of chiral center racemization under acidic acetate conditions.
The Novel Approach
The methodology outlined in CN101967118A offers a decisive break from these inefficiencies by employing a mesylate displacement strategy that operates under thermally mild conditions. Instead of cryogenic lithiation, the novel approach utilizes a nucleophilic substitution between a chiral piperidine derivative and a mesylated glycine ester intermediate. This reaction proceeds efficiently in organic solvents like toluene with triethylamine as a base, typically under reflux conditions, eliminating the need for sub-zero temperature control. A key innovation lies in the purification strategy: rather than relying on chromatographic separation, the process leverages the formation of specific salts (such as sodium, potassium, or triethylamine salts) to precipitate the product while leaving impurities in the mother liquor. This salification step, followed by acidification to liberate the free base, effectively upgrades the crude content from approximately 95% to over 99.6% purity. By avoiding hydrogenation and strong cryogenic bases, this route not only enhances safety but also simplifies the downstream processing workflow, making it ideally suited for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into N-Alkylation and Chiral Stability
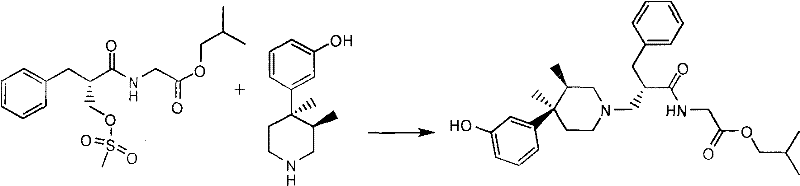
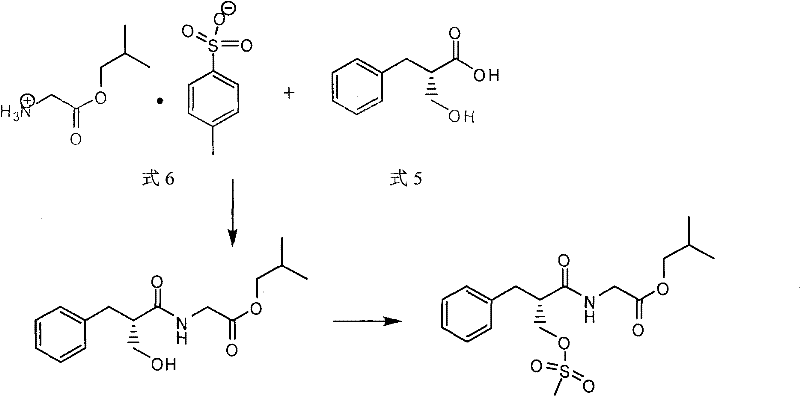
The core chemical transformation in this patent is the N-alkylation reaction between (3R, 4R)-3-(3,4-dimethyl-4-piperidine base) phenol (Formula 3) and (S)-2-methylsulfonyl methylbenzene propionyl-glycine isobutyl ester (Formula 2). Mechanistically, this is a classic SN2 displacement where the secondary amine of the piperidine ring acts as the nucleophile, attacking the methylene carbon attached to the mesylate leaving group. Crucially, the chiral center of the phenylpropionic acid moiety is located at the alpha-position relative to the carbonyl group, whereas the reaction site is at the beta-position (the hydroxymethyl group converted to mesylate). Because the bond breaking and forming occur at a carbon atom distinct from the stereocenter, the electronic environment of the chiral carbon is not disturbed during the transition state. This spatial separation ensures that the optical purity established in the starting material is faithfully transmitted to the product without epimerization or racemization. The use of mild organic bases like triethylamine further supports this stability, as they are sufficiently basic to deprotonate the ammonium species formed during alkylation but not strong enough to induce enolization at the alpha-carbon, which is a common pathway for racemization in peptide-like structures.
Impurity control is another critical aspect of this mechanism, achieved through the strategic selection of the isobutyl ester protecting group. Unlike benzyl esters that require reductive cleavage, the isobutyl ester is susceptible to hydrolysis under mild alkaline conditions (e.g., aqueous NaOH). This selectivity allows for the cleavage of the ester bond without affecting the amide linkage or the sensitive chiral centers. The subsequent purification via salt formation exploits the differential solubility of the Alvimopan salt versus neutral organic impurities. By dissolving the crude product in a solvent system and adding a stoichiometric amount of base, the Alvimopan converts to a crystalline salt that precipitates upon cooling, while non-ionic impurities remain dissolved in the supernatant. This physical separation mechanism is far more robust and scalable than chromatographic methods, ensuring that the final API meets stringent pharmacopeial standards for single maximum impurities, which are reported to be as low as 0.04% in optimized embodiments.
How to Synthesize Alvimopan Efficiently
The synthesis of Alvimopan described in this patent is a multi-step process that prioritizes yield and purity through careful control of reaction parameters. The procedure begins with the preparation of the electrophilic partner, (S)-2-methylsulfonyl methylbenzene propionyl-glycine isobutyl ester, which involves condensing (S)-2-hydroxymethyl-3-phenylpropionic acid with glycine isobutyl ester tosylate using DCC and HOBT, followed by mesylation with methanesulfonyl chloride. This intermediate is then coupled with the chiral piperidine fragment in a refluxing toluene solution. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures required to replicate this high-purity outcome, are provided in the guide below.
- Synthesize (S)-2-methylsulfonyl methylbenzene propionyl-glycine isobutyl ester (Formula 2) by condensing (S)-2-hydroxymethyl-3-phenylpropionic acid with glycine isobutyl ester, followed by mesylation.
- Perform N-alkylation by reacting Formula 2 with (3R, 4R)-3-(3,4-dimethyl-4-piperidine base) phenol (Formula 3) in toluene with triethylamine under reflux.
- Hydrolyze the resulting ester intermediate using aqueous sodium hydroxide, followed by purification via salt formation (e.g., sodium or triethylamine salt) and acidification to obtain high-purity Alvimopan.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers profound logistical and financial benefits derived directly from its chemical design. The elimination of cryogenic reagents like diisopropyl lithium removes the dependency on specialized low-temperature reactors and the associated high energy costs for cooling, leading to substantial cost savings in utility consumption. Furthermore, the avoidance of catalytic hydrogenation mitigates the safety risks associated with high-pressure hydrogen gas and pyrophoric catalysts, thereby reducing insurance premiums and safety compliance overheads. The process relies on commodity chemicals such as toluene, triethylamine, and sodium hydroxide, which are readily available in the global market, ensuring supply chain continuity and reducing lead time for high-purity pharmaceutical intermediates. The robustness of the salification purification step means that production batches are less likely to fail quality control due to trace impurities, enhancing overall manufacturing reliability.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the complete removal of chiral column chromatography from the production workflow. Traditional methods required repeated chiral separations, which consume vast amounts of expensive stationary phases and solvents while yielding low recovery rates. By achieving high optical purity through stereospecific synthesis and simple crystallization, this method drastically reduces solvent usage and waste disposal costs. Additionally, the higher yields reported in the embodiments (often exceeding 60% for the coupling step and 80% for the final purification) mean that less raw material is required per kilogram of finished API, directly lowering the cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: The synthetic route is designed for resilience, utilizing stable intermediates that do not require inert atmosphere storage or ultra-low temperature transport. The starting materials, such as the piperidine derivative and the phenylpropionic acid precursor, are synthesized via reliable, established chemistries that are less prone to batch-to-batch variability compared to organometallic reactions. This stability allows manufacturers to maintain strategic stockpiles of intermediates without degradation, ensuring that production schedules can be met even during fluctuations in raw material availability. The simplified process flow also reduces the number of unit operations, decreasing the likelihood of mechanical failures or bottlenecks in the production line.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns with green chemistry principles by minimizing the use of hazardous reagents and heavy metals. The absence of palladium catalysts eliminates the need for rigorous metal scavenging steps and the associated heavy metal waste streams, simplifying regulatory compliance for residual metals in the final drug product. The aqueous workup and crystallization steps generate waste streams that are easier to treat compared to the complex organic mixtures from chromatographic purifications. This environmental profile facilitates easier permitting for new manufacturing facilities and supports the sustainability goals of modern pharmaceutical companies seeking to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and advantages of this Alvimopan synthesis technology. These answers are derived directly from the experimental data and comparative analysis presented in patent CN101967118A, providing clarity on how this method outperforms legacy processes in terms of safety, purity, and operational simplicity.
Q: Why does this new Alvimopan synthesis method avoid catalytic hydrogenation?
A: Previous methods required hydrogenolysis of benzyl esters using Pd/C, which poses explosion risks and requires high-pressure equipment. This patent utilizes an isobutyl ester that can be cleaved via mild alkaline hydrolysis, significantly enhancing operational safety and reducing equipment costs.
Q: How is the optical purity of Alvimopan maintained during the N-alkylation step?
A: The reaction occurs at the mesylate group on the side chain of the phenylpropionic acid derivative, which is distant from the chiral alpha-carbon. Since the chiral center is not involved in the bond-breaking or bond-forming events of the SN2 displacement, the configuration remains stable without racemization.
Q: What represents the primary cost advantage of this purification strategy?
A: The method employs a salification and recrystallization technique (forming sodium, potassium, or amine salts) to remove impurities. This eliminates the need for expensive and low-yield chiral column chromatography, which was a bottleneck in earlier synthetic routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alvimopan Supplier
The technical advancements detailed in CN101967118A underscore the potential for producing Alvimopan with exceptional purity and efficiency, a standard that NINGBO INNO PHARMCHEM is fully equipped to deliver. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facility is outfitted with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including the low impurity profiles (<0.1%) achievable through this salification-based purification method. We understand that transitioning to a new synthetic route requires confidence in both the chemistry and the manufacturer, and our team is dedicated to validating every step of the process to guarantee product integrity.
We invite you to leverage our expertise to optimize your supply chain for this critical gastrointestinal therapeutic. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data from our pilot runs and comprehensive route feasibility assessments to demonstrate how our implementation of this patent can drive value for your organization. Let us collaborate to bring safer, more affordable Alvimopan to the market.
