Advanced Manufacturing of Ramosetron Intermediates via Streamlined Direct Condensation Technology
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical antiemetic agents, and the synthesis of Ramosetron, a potent 5-HT3 receptor antagonist, remains a focal point for process optimization. Patent CN101585831B introduces a groundbreaking methodology that fundamentally alters the production landscape by employing a direct condensation strategy mediated by trifluoroacetic anhydride (TFAA) and polyphosphoric acid (PPA). This innovation addresses the longstanding inefficiencies of prior art, which often relied on cumbersome resolution steps or hazardous reagents, by enabling a convergent synthesis directly from chiral precursors. The technical significance of this approach lies in its ability to synchronize high yield with operational safety, creating a pathway that is inherently more amenable to the rigorous demands of Good Manufacturing Practice (GMP) environments. By eliminating the need for intermediate isolation of unstable acid chlorides or the use of expensive organometallic reagents, this patent establishes a new benchmark for the reliable API intermediate supplier seeking to optimize their portfolio. The strategic implementation of PPA as a dual-purpose catalyst and dehydrating agent ensures that the reaction proceeds with exceptional selectivity, minimizing the formation of regioisomers that typically complicate downstream purification efforts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Ramosetron has been plagued by significant thermodynamic and economic hurdles, primarily stemming from the reliance on racemic synthesis followed by optical resolution. As illustrated in early methodologies, the condensation of 1-Methyl-1H-indole with benzimidazole derivatives in the presence of phosphoryl chloride (POCl3) yields a racemic mixture that necessitates a subsequent, yield-halving resolution step to isolate the active (R)-enantiomer. 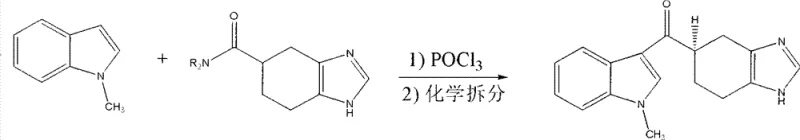 This inherent 50% theoretical loss of material represents a massive inefficiency in atom economy and drastically inflates the cost of goods sold (COGS). Furthermore, alternative routes described in Chinese patent CN1696128 involve the pre-formation of acid chlorides, which introduces stability issues and requires stringent moisture control, thereby complicating the engineering controls required for large-scale reactors. Another competing method utilizes aluminum alkyls under cryogenic conditions ranging from -25°C to -40°C, imposing severe energy penalties and limiting the throughput capacity of standard stainless steel equipment. These conventional approaches collectively suffer from extended processing times, high consumption of auxiliary chemicals, and significant safety risks associated with handling corrosive or pyrophoric reagents, making them suboptimal for modern cost reduction in pharmaceutical manufacturing.
This inherent 50% theoretical loss of material represents a massive inefficiency in atom economy and drastically inflates the cost of goods sold (COGS). Furthermore, alternative routes described in Chinese patent CN1696128 involve the pre-formation of acid chlorides, which introduces stability issues and requires stringent moisture control, thereby complicating the engineering controls required for large-scale reactors. Another competing method utilizes aluminum alkyls under cryogenic conditions ranging from -25°C to -40°C, imposing severe energy penalties and limiting the throughput capacity of standard stainless steel equipment. These conventional approaches collectively suffer from extended processing times, high consumption of auxiliary chemicals, and significant safety risks associated with handling corrosive or pyrophoric reagents, making them suboptimal for modern cost reduction in pharmaceutical manufacturing.
The Novel Approach
In stark contrast to these legacy processes, the novel approach detailed in CN101585831B leverages a synergistic catalytic system comprising trifluoroacetic anhydride and polyphosphoric acid to drive the condensation directly. This method bypasses the isolation of reactive intermediates entirely, allowing for a telescoped process where the carboxylic acid is activated in situ and immediately coupled with the indole nucleus. 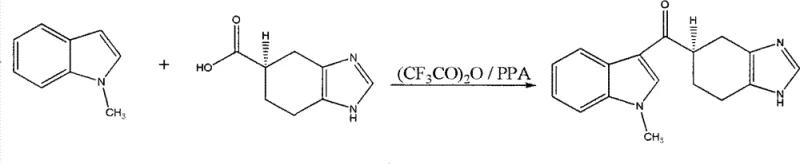 The reaction operates effectively within a moderate temperature window of 20°C to 80°C, eliminating the need for energy-intensive cryogenic cooling and allowing for the use of standard heating utilities found in any multipurpose chemical plant. By utilizing optically pure (R)-4,5,6,7-tetrahydrochysene-1H-benzoglyoxaline-5-formic acid as the starting material, the process preserves the stereochemical integrity throughout the synthesis, rendering the wasteful resolution step completely obsolete. The versatility of the solvent system, which accommodates common organic media such as acetonitrile, dichloromethane, or toluene, provides procurement teams with the flexibility to source materials based on market availability and price volatility. This streamlined workflow not only accelerates the overall cycle time but also significantly reduces the environmental footprint by minimizing solvent exchanges and waste generation, aligning perfectly with the principles of green chemistry and sustainable commercial scale-up of complex pharmaceutical intermediates.
The reaction operates effectively within a moderate temperature window of 20°C to 80°C, eliminating the need for energy-intensive cryogenic cooling and allowing for the use of standard heating utilities found in any multipurpose chemical plant. By utilizing optically pure (R)-4,5,6,7-tetrahydrochysene-1H-benzoglyoxaline-5-formic acid as the starting material, the process preserves the stereochemical integrity throughout the synthesis, rendering the wasteful resolution step completely obsolete. The versatility of the solvent system, which accommodates common organic media such as acetonitrile, dichloromethane, or toluene, provides procurement teams with the flexibility to source materials based on market availability and price volatility. This streamlined workflow not only accelerates the overall cycle time but also significantly reduces the environmental footprint by minimizing solvent exchanges and waste generation, aligning perfectly with the principles of green chemistry and sustainable commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into TFAA/PPA Catalyzed Condensation
The core mechanistic advantage of this synthesis lies in the unique activation profile generated by the combination of trifluoroacetic anhydride and polyphosphoric acid. In this system, the TFAA acts as a potent dehydrating agent that reacts with the carboxylic acid group of the chiral benzimidazole precursor to form a mixed anhydride or a highly reactive acylium ion species. The polyphosphoric acid serves a dual function: it acts as a strong Brønsted acid to protonate the carbonyl oxygen, thereby increasing the electrophilicity of the carbonyl carbon, and simultaneously functions as a Lewis acid to stabilize the transition state during the nucleophilic attack. When the N-skatalole (1-methylindole) is introduced, the electron-rich C3 position of the indole ring attacks the activated carbonyl center in a classic Friedel-Crafts acylation manifold. The presence of PPA ensures that the water produced during the condensation is sequestered effectively, driving the equilibrium towards product formation according to Le Chatelier's principle. This mechanism avoids the formation of free acid chlorides, which are prone to hydrolysis and side reactions, thus ensuring a cleaner reaction profile. The mild acidity of the PPA/TFAA system compared to harsh Lewis acids like aluminum chloride prevents the degradation of the sensitive benzimidazole ring system, which is crucial for maintaining high assay purity.
From an impurity control perspective, this mechanistic pathway offers superior selectivity that simplifies the purification train. Traditional methods often generate regioisomers where acylation occurs at the C2 position of the indole or undergoes poly-acylation, requiring complex chromatographic separations that are difficult to scale. The specific steric and electronic environment created by the PPA matrix favors attack exclusively at the C3 position of the indole, which is the most nucleophilic site, thereby suppressing the formation of these structural analogs. Furthermore, because the reaction starts with an enantiomerically pure acid, there is no risk of racemization at the chiral center adjacent to the carbonyl group, a common pitfall in basic or high-temperature acidic conditions. The workup procedure, involving quenching with aqueous alkali and pH adjustment, effectively neutralizes the acidic catalysts and precipitates the product as a hydrochloride salt, which can be easily purified by recrystallization from ethanol. This robustness in impurity rejection means that the final API intermediate meets stringent purity specifications with minimal processing, reducing the load on quality control laboratories and ensuring batch-to-batch consistency essential for regulatory filings.
How to Synthesize Ramosetron Efficiently
The operational protocol for this synthesis is designed for maximum reproducibility and safety, utilizing standard unit operations found in modern chemical facilities. The process begins with the activation of the chiral acid in an inert solvent, followed by the controlled addition of the indole component and a defined heating period to ensure complete conversion. Detailed standardized operating procedures regarding stoichiometry, addition rates, and crystallization parameters are critical for maintaining the high yields reported in the patent examples.
- Dissolve (R)-4,5,6,7-tetrahydrochysene-1H-benzoglyoxaline-5-formic acid in an inert solvent containing trifluoroacetic anhydride and polyphosphoric acid, stirring at room temperature for activation.
- Add the N-skatalole solution to the activated mixture and heat to a moderate temperature range of 20-80°C to facilitate the condensation reaction.
- Quench the reaction with ice-water containing alkali, adjust pH to neutral, and purify the resulting solid through recrystallization to obtain high-purity Ramosetron.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this TFAA/PPA mediated synthesis represents a strategic opportunity to de-risk the supply of critical antiemetic intermediates while driving down overall manufacturing costs. The elimination of the optical resolution step is perhaps the most significant economic driver, as it theoretically doubles the yield relative to racemic routes by utilizing 100% of the chiral starting material rather than discarding the unwanted enantiomer. This improvement in atom economy translates directly into substantial cost savings on raw materials, particularly given the high value of chiral benzimidazole precursors. Additionally, the removal of expensive and hazardous reagents like aluminum alkyls removes a major cost center and supply bottleneck, as these materials often require specialized handling and storage protocols that increase overhead. The ability to run the reaction at near-ambient or moderate temperatures significantly reduces utility costs associated with cryogenic cooling, allowing for higher batch turnover rates in existing reactor trains without the need for capital-intensive retrofitting. These factors combine to create a leaner, more agile manufacturing process that enhances supply chain reliability and reduces lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The primary economic benefit stems from the telescoped nature of the reaction, which consolidates multiple synthetic steps into a single pot, thereby reducing labor hours, solvent consumption, and waste disposal fees. By avoiding the isolation of unstable acid chloride intermediates, the process minimizes material losses due to hydrolysis or decomposition, further improving the overall mass balance. The use of commodity chemicals like trifluoroacetic anhydride and polyphosphoric acid, which are readily available in bulk quantities, ensures stable pricing and protects against the volatility often seen with specialized organometallic catalysts. Consequently, the total cost of production is significantly lowered, providing a competitive margin advantage in the global marketplace for generic pharmaceutical ingredients.
- Enhanced Supply Chain Reliability: From a logistics perspective, the simplified reagent list and moderate reaction conditions make this process highly resilient to supply disruptions. Unlike cryogenic processes that are vulnerable to equipment failure or power fluctuations, this method is robust and forgiving, ensuring consistent output even in varied manufacturing environments. The flexibility in solvent selection allows production sites to switch between acetonitrile, dichloromethane, or toluene based on regional availability and regulatory restrictions, preventing production halts due to solvent shortages. This adaptability ensures a continuous flow of materials to downstream formulation partners, securing the supply chain against external shocks and maintaining service levels for key customers.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is markedly superior to legacy methods, facilitating easier regulatory approval and community acceptance for manufacturing sites. The reduction in hazardous waste generation, particularly the avoidance of aluminum salts and phosphorus oxychloride byproducts, simplifies effluent treatment and lowers the cost of environmental compliance. The process is inherently scalable, as demonstrated by the patent examples which show consistent yields across different scales, making it suitable for commercial scale-up of complex pharmaceutical intermediates from pilot plant to multi-ton production. This scalability ensures that the technology can meet growing global demand for Ramosetron without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route, derived directly from the experimental data and claims within the patent documentation. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer and for procurement professionals assessing the long-term viability of the supply source.
Q: How does the TFAA/PPA system improve upon traditional POCl3 methods?
A: The TFAA/PPA system allows for direct condensation without forming unstable acid chloride intermediates, significantly reducing reaction steps and avoiding the harsh conditions associated with phosphoryl chloride.
Q: What is the optical purity achievable with this synthetic route?
A: By utilizing optically active (R)-acid starting materials directly, the process maintains high stereochemical integrity, achieving optical rotation values consistent with high-purity standards without the need for post-synthesis resolution.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction operates at moderate temperatures (0-110°C) and uses common inert solvents like acetonitrile or dichloromethane, making it highly scalable and safer than cryogenic aluminum alkyl methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ramosetron Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant manufacturing pathways for high-value therapeutic agents like Ramosetron. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this advanced TFAA/PPA catalyzed process are fully realized at an industrial level. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of API intermediate meets the highest international standards. Our commitment to process excellence means that we can deliver materials with consistent optical rotation and assay purity, supporting your regulatory filings and clinical trials with unwavering reliability.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this direct condensation route for your operations. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless integration of high-quality Ramosetron intermediates into your production schedule.
