Advanced Green Synthesis of 3-Sulfone Methyl-1H-Indole Derivatives for Commercial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally sustainable pathways to access complex heterocyclic scaffolds. Patent CN112094220A introduces a significant breakthrough in the synthesis of 3-sulfone methyl-1H-indole compounds, a structural motif prevalent in numerous bioactive molecules and drug candidates. This technology leverages a copper-catalyzed oxidative decarboxylation strategy that fundamentally shifts the paradigm from multi-step, hazardous processes to a streamlined, one-pot green synthesis. By utilizing readily available 3-indoleacetic acid derivatives and sodium sulfinates, this method eliminates the need for pre-functionalized halides or expensive transition metal complexes often required in traditional cross-coupling reactions. For R&D directors and procurement specialists, this represents a tangible opportunity to enhance supply chain resilience while reducing the environmental footprint of API intermediate manufacturing. The strategic implementation of this patent data allows for the production of high-purity indole sulfones with exceptional efficiency, directly addressing the critical needs of modern drug development pipelines where speed and purity are paramount.
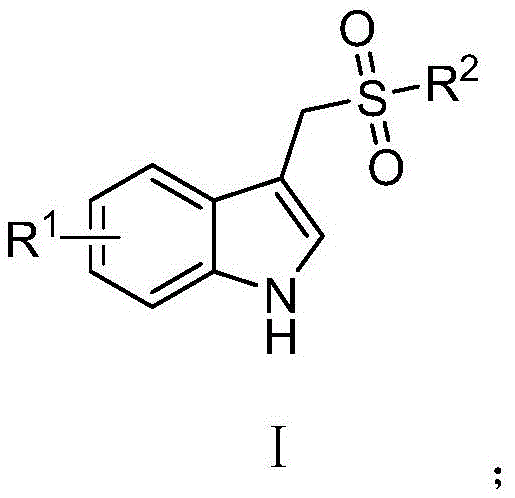
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of sulfone-functionalized indole cores has been plagued by significant synthetic inefficiencies that hinder commercial viability. Traditional routes often rely on the oxidation of thioether or sulfoxide precursors, which necessitates harsh oxidizing agents and generates substantial quantities of sulfur-containing waste streams that are costly to treat. Furthermore, existing methodologies frequently involve multi-step sequences requiring the protection and deprotection of sensitive functional groups, leading to cumulative yield losses and extended production timelines. The reliance on precious metal catalysts such as palladium or rhodium in conventional cross-coupling approaches not only inflates raw material costs but also introduces stringent requirements for residual metal removal to meet pharmaceutical regulatory standards. These legacy processes often suffer from poor atom economy and require large volumes of organic solvents, creating safety hazards and complicating the scale-up process for manufacturing teams. Consequently, the industry has faced persistent challenges in securing a reliable supply of these critical intermediates at a cost structure that supports competitive drug pricing.
The Novel Approach
The methodology disclosed in CN112094220A offers a transformative solution by employing a direct oxidative decarboxylative coupling strategy that bypasses the limitations of prior art. This novel approach utilizes inexpensive and abundant copper salts, specifically copper acetate, to catalyze the reaction between 3-indoleacetic acids and sodium sulfinates under mild thermal conditions. By avoiding the use of toxic oxidants and precious metals, the process inherently reduces the burden on downstream purification and waste management systems. The reaction proceeds efficiently in polar aprotic solvents like DMF or DMSO, which facilitate the solubility of ionic intermediates and promote the catalytic cycle without the need for exotic ligands or additives. This simplification of the reaction profile translates directly into operational excellence, allowing for easier process control and reduced risk of batch failure during commercial production. The ability to achieve high conversion rates in a single step significantly compresses the manufacturing timeline, offering a distinct competitive advantage for companies aiming to accelerate their time-to-market for new therapeutic agents.
Mechanistic Insights into Copper-Catalyzed Oxidative Decarboxylation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific substrate classes. The reaction initiates with the coordination of the copper catalyst to the carboxylate group of the 3-indoleacetic acid derivative, forming a reactive copper-carboxylate complex. Under thermal conditions, this complex undergoes decarboxylation to generate a nucleophilic indolyl-methyl copper species, releasing carbon dioxide as the only byproduct of this step. Simultaneously, the sodium sulfinate interacts with the copper center, facilitating the formation of a sulfur-copper bond that positions the sulfonyl group for transfer. The subsequent reductive elimination step forge the carbon-sulfur bond, releasing the desired 3-sulfone methyl-1H-indole product and regenerating the active copper catalyst species. This catalytic cycle is highly efficient because it avoids the formation of stable off-cycle intermediates that often plague transition metal catalysis. The use of nitrogen protection ensures that the reactive copper species are not quenched by atmospheric oxygen, maintaining the integrity of the catalytic cycle throughout the reaction duration.
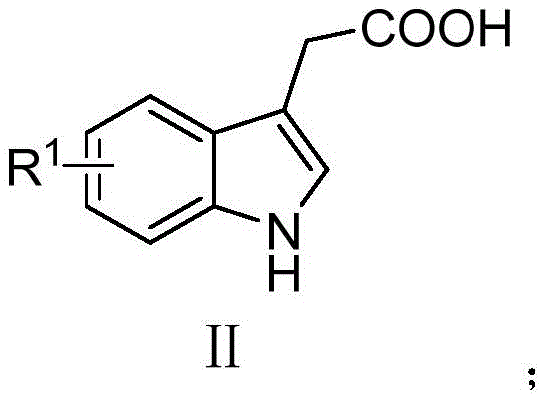
Impurity control is a critical aspect of this synthesis, particularly given the potential for over-oxidation or homocoupling side reactions. The choice of copper acetate as the catalyst plays a pivotal role in minimizing impurity profiles, as it provides a balanced redox potential that favors the desired cross-coupling over competing pathways. The reaction conditions, specifically the temperature range of 100°C to 115°C, are optimized to drive the decarboxylation forward without inducing thermal degradation of the sensitive indole nucleus. Furthermore, the stoichiometry of the sodium sulfinate is carefully controlled to ensure complete consumption of the indole acid while preventing the accumulation of unreacted sulfinate which could complicate isolation. The simplicity of the workup procedure, involving basic extraction and column chromatography, allows for the effective removal of copper residues and inorganic salts. This results in a final product with high chemical purity, meeting the stringent specifications required for pharmaceutical intermediates and reducing the need for extensive recrystallization steps that lower overall yield.
How to Synthesize 3-Sulfone Methyl-1H-Indole Efficiently
Implementing this synthesis route requires precise adherence to the optimized parameters identified in the patent data to ensure reproducibility and safety. The process begins with the charging of the reactor with the substituted 3-indoleacetic acid and the corresponding sodium sulfinate in a molar ratio that favors complete conversion of the limiting reagent. The addition of the copper catalyst and the selection of the appropriate solvent system are critical steps that determine the reaction kinetics and final yield. Operators must maintain an inert atmosphere throughout the heating phase to prevent catalyst deactivation and ensure consistent product quality. While the general procedure is robust, specific substrates may require minor adjustments to temperature or reaction time to accommodate steric or electronic effects. For detailed standard operating procedures and safety guidelines, please refer to the technical documentation provided below which outlines the step-by-step protocol for laboratory and pilot-scale execution.
- Prepare the reaction mixture by combining R1-substituted 3-indoleacetic acid and R2-substituted sodium sulfinate in a reaction vessel.
- Add a copper catalyst, specifically copper acetate, and use N,N-dimethylformamide (DMF) or dimethyl sulfoxide (DMSO) as the solvent under nitrogen protection.
- Heat the reaction mixture to a temperature between 100°C and 115°C, preferably 115°C, to facilitate oxidative decarboxylation and yield the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this green synthesis method offers profound benefits for procurement managers and supply chain leaders focused on cost optimization and risk mitigation. The elimination of expensive precious metal catalysts and the reduction in solvent usage directly contribute to a lower cost of goods sold (COGS), enhancing the margin potential for downstream API production. By simplifying the synthetic route to a single step, the process significantly reduces the operational complexity and the associated labor costs required for multi-step manufacturing. This efficiency gain allows for faster batch turnover, enabling suppliers to respond more agilely to fluctuating market demands and urgent procurement requests. Furthermore, the use of commercially available and stable starting materials ensures a secure supply chain that is less vulnerable to the disruptions often seen with specialized reagents. The environmental compliance inherent in this green chemistry approach also reduces the regulatory burden and potential liabilities associated with hazardous waste disposal, aligning with the sustainability goals of modern pharmaceutical corporations.
- Cost Reduction in Manufacturing: The substitution of precious metal catalysts with abundant copper salts drastically lowers the raw material expenditure per kilogram of product. Additionally, the one-pot nature of the reaction eliminates the costs associated with isolating intermediates, reducing solvent consumption, and minimizing energy usage across multiple heating and cooling cycles. This streamlined process flow translates into substantial cost savings that can be passed down the supply chain, making the final API more economically viable. The reduced need for complex purification steps further lowers the operational overhead, allowing for a more competitive pricing structure in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Sourcing 3-indoleacetic acid derivatives and sodium sulfinates is significantly more straightforward than procuring specialized halogenated indoles or organometallic reagents. These starting materials are commodity chemicals with established global supply networks, reducing the risk of shortages or price volatility. The robustness of the reaction conditions means that production can be scaled across multiple manufacturing sites without requiring highly specialized equipment or expertise. This flexibility ensures continuity of supply even in the face of regional disruptions, providing procurement teams with the confidence to commit to long-term supply agreements. The ability to rapidly scale production from gram to ton scale ensures that clinical trial material and commercial stock can be generated without bottlenecks.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this method facilitate easier regulatory approval and environmental compliance. The reduction in hazardous waste generation simplifies the permitting process for new manufacturing facilities and lowers the cost of waste treatment. The mild reaction conditions and use of standard solvents make the process highly amenable to scale-up in existing stainless steel reactors without the need for costly retrofitting. This scalability ensures that the technology can meet the growing demand for indole-based therapeutics as they progress through clinical pipelines. By adopting this sustainable methodology, companies can enhance their corporate social responsibility profiles while simultaneously improving their operational efficiency and bottom line.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation to ensure accuracy and reliability. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The insights provided here cover catalyst selection, reaction optimization, and substrate compatibility, offering a comprehensive overview for decision-makers.
Q: What is the optimal catalyst for synthesizing 3-sulfone methyl-1H-indole compounds?
A: According to patent CN112094220A, copper acetate (Cu(OAc)2·H2O) demonstrates superior catalytic activity compared to other copper salts like CuO or CuI, achieving yields up to 90% in DMF solvent.
Q: What are the critical reaction conditions for this green synthesis method?
A: The process requires heating the reaction mixture to 115°C under a nitrogen atmosphere. Using polar aprotic solvents like DMF or DMSO is essential for high conversion, while avoiding expensive additives.
Q: Does this method support diverse substrate scopes for drug discovery?
A: Yes, the method tolerates various substituents on both the indole ring (R1) and the sulfinate group (R2), including halogens, alkyls, and alkoxy groups, making it versatile for generating diverse pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Sulfone Methyl-1H-Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality intermediates to drive your drug development programs forward. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch. By leveraging the advanced copper-catalyzed technology described in CN112094220A, we can offer you a supply solution that balances cost-efficiency with uncompromising quality standards. Our dedication to green chemistry aligns with the evolving regulatory landscape, providing you with a sustainable partner for your long-term supply needs.
We invite you to engage with our technical procurement team to discuss how this synthesis route can be tailored to your specific project requirements. Request a Customized Cost-Saving Analysis to understand the potential economic benefits of switching to this streamlined process for your supply chain. We are prepared to provide specific COA data and route feasibility assessments to support your vendor qualification process. Let us help you optimize your sourcing strategy and secure a reliable supply of high-purity indole derivatives for your next breakthrough therapy.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
