Scalable Green Synthesis of 3-Sulfone Methyl-1H-Indole Derivatives for Advanced Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing complex heterocyclic scaffolds, particularly those serving as critical building blocks for bioactive molecules. A recent technological breakthrough, documented in Chinese Patent CN112094220B, introduces a highly efficient green synthesis method for 3-sulfone methyl-1H-indole compounds, addressing long-standing challenges in yield and environmental impact. This innovation leverages a copper-catalyzed oxidative decarboxylation strategy, transforming readily available 3-indoleacetic acid derivatives into valuable sulfone-functionalized indoles in a single operational step. The significance of this development cannot be overstated for R&D directors and procurement specialists alike, as it offers a pathway to high-purity intermediates while drastically simplifying the supply chain logistics associated with multi-step syntheses. By utilizing earth-abundant copper catalysts instead of precious metals, the process aligns perfectly with modern sustainability goals without compromising on chemical efficiency.
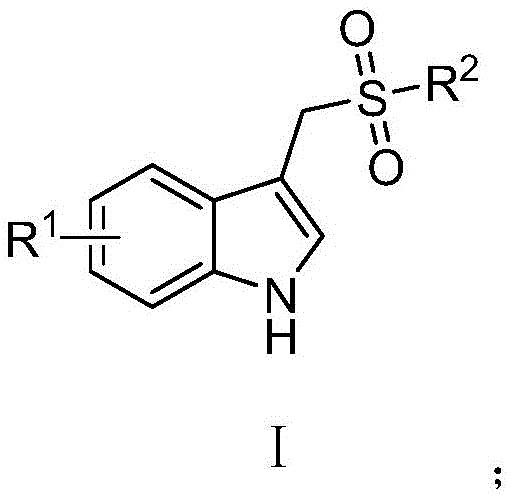
Furthermore, the versatility of this synthetic route allows for broad substrate scope, accommodating various halogen, alkyl, and alkoxy substitutions on the indole ring, as well as diverse aryl and alkyl groups on the sulfone moiety. This structural flexibility is paramount for medicinal chemists exploring structure-activity relationships (SAR) in drug discovery programs targeting inflammation and pain management. The patent explicitly details how this method overcomes the limitations of prior art, which often relied on harsh oxidation conditions or expensive reagents that complicated downstream processing. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such patented technologies allows us to offer clients not just a product, but a strategically optimized manufacturing solution that ensures continuity and cost-effectiveness in the production of next-generation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-sulfone methyl substituted indoles has been fraught with significant technical and economic hurdles that hindered their widespread adoption in large-scale manufacturing. Traditional approaches typically involved the oxidation of 3-thiomethyl or sulfoxymethyl indole precursors, a process that necessitates multiple synthetic steps, each introducing potential yield losses and impurity profiles that are difficult to manage. These multi-step sequences often require stoichiometric amounts of strong oxidizing agents, leading to substantial waste generation and safety concerns regarding exothermic reactions. Moreover, alternative methods reported in literature, such as iron-catalyzed sulfonation in aqueous systems, while greener in theory, often suffer from limited substrate scope and the requirement for specialized ligands or conditions that are difficult to replicate outside of a research laboratory setting. The cumulative effect of these inefficiencies is a dramatic increase in the cost of goods sold (COGS) and extended lead times, creating bottlenecks for procurement managers trying to secure reliable supplies of high-quality intermediates for clinical and commercial batches.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent data utilizes a direct oxidative decarboxylation coupling between substituted 3-indoleacetic acids and sodium sulfinates, streamlining the entire production workflow into a single pot. This approach fundamentally shifts the paradigm from stepwise construction to convergent synthesis, thereby eliminating the need for isolating unstable intermediates and reducing the overall solvent footprint by nearly half compared to traditional routes. The use of simple copper salts as catalysts, specifically copper(II) acetate, replaces expensive and toxic transition metals, offering a dual benefit of cost reduction and improved environmental compliance. Reaction conditions are remarkably mild, operating effectively at temperatures between 100°C and 115°C under a standard nitrogen atmosphere, which minimizes energy consumption and equipment stress. For supply chain heads, this translates to a more resilient manufacturing process that is less susceptible to raw material volatility and regulatory scrutiny regarding heavy metal residues in the final active pharmaceutical ingredients.
Mechanistic Insights into Copper-Catalyzed Oxidative Decarboxylation
The core of this technological advancement lies in the intricate mechanistic pathway facilitated by the copper catalyst, which orchestrates the cleavage of the carbon-carbon bond in the acetic acid side chain while simultaneously forming the new carbon-sulfur bond. The reaction initiates with the coordination of the copper(II) species to the carboxylate group of the 3-indoleacetic acid substrate, forming a reactive bivalent copper complex that lowers the activation energy for decarboxylation. Upon heating, this complex undergoes oxidative decarboxylation to generate a transient radical or cationic species at the 3-position of the indole ring, which is immediately trapped by the nucleophilic sulfur species derived from the sodium sulfinate. This concerted mechanism ensures high regioselectivity, preventing unwanted substitution on the benzene ring of the indole scaffold and maintaining the integrity of sensitive functional groups. The choice of solvent plays a critical role in stabilizing these transition states; polar aprotic solvents like DMF and DMSO are essential for solubilizing the ionic sodium sulfinate reagents and facilitating the electron transfer processes required for the catalytic cycle to turnover efficiently.
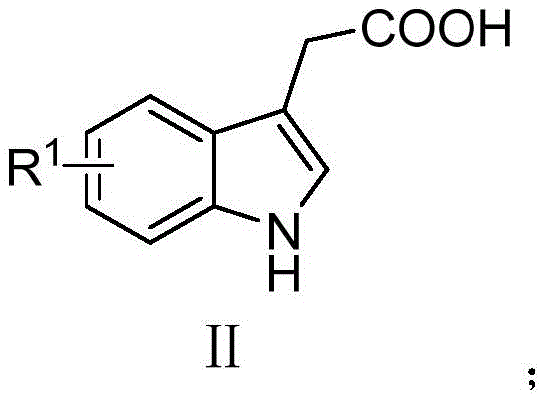
From an impurity control perspective, this mechanism offers distinct advantages by minimizing side reactions such as polymerization or over-oxidation, which are common pitfalls in free-radical chemistries. The patent data highlights that the reaction proceeds cleanly to afford the target 3-sulfone methyl-1H-indole compounds with yields reaching up to 90% under optimized conditions, a testament to the robustness of the catalytic system. Post-reaction workup is equally straightforward, involving simple aqueous extraction and column chromatography, which effectively removes copper residues and unreacted starting materials. This level of purity is crucial for R&D directors who require materials with well-defined impurity profiles for toxicological studies and regulatory filings. The ability to tune the electronic properties of both the indole and the sulfinate components allows for precise modulation of the reaction kinetics, ensuring that even sterically hindered or electronically deactivated substrates can be converted with high efficiency, thereby expanding the chemical space accessible for drug design.
How to Synthesize 3-Sulfone Methyl-1H-Indole Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators must adhere to specific protocols regarding reagent ratios and atmospheric controls to maximize yield and safety. The process begins with the precise weighing of the substituted 3-indoleacetic acid and the corresponding sodium sulfinate, typically in a molar ratio of 1:2 to drive the equilibrium towards product formation. These solids are suspended in anhydrous N,N-dimethylformamide (DMF), and the reaction vessel is purged with nitrogen to exclude oxygen, which could otherwise lead to non-productive oxidation of the sulfinate. Following the addition of the copper(II) acetate catalyst, the mixture is heated to 115°C and stirred for approximately 6 hours, after which conversion is monitored via TLC or HPLC. Detailed standardized synthesis steps follow below to ensure reproducibility and quality control across different batches.
- Combine substituted 3-indoleacetic acid and substituted sodium sulfinate in a reaction vessel under nitrogen protection.
- Add copper(II) acetate catalyst and dissolve the mixture in N,N-dimethylformamide (DMF) or dimethyl sulfoxide (DMSO).
- Heat the reaction mixture to 115°C for 6 hours, then perform extraction and column chromatography to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this copper-catalyzed green synthesis method presents a compelling value proposition that extends far beyond simple chemical transformation. By shifting away from multi-step oxidation routes that rely on volatile sulfur reagents and precious metal catalysts, manufacturers can achieve a significant reduction in raw material costs and inventory complexity. The reliance on commodity chemicals like copper acetate and DMF, which are globally sourced and price-stable, mitigates the risk of supply disruptions that often plague specialty reagent markets. Furthermore, the simplified workup procedure reduces the demand for extensive purification infrastructure, allowing for faster batch turnover and shorter lead times for delivering high-purity intermediates to downstream customers. This operational agility is a critical competitive advantage in the fast-paced pharmaceutical sector, where time-to-market is often the deciding factor in commercial success.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and the reduction of synthetic steps directly translate to lower production costs per kilogram. By avoiding the need for specialized ligands or harsh oxidizing agents, the process minimizes waste disposal fees and safety compliance costs associated with hazardous materials handling. The high atom economy of the decarboxylation reaction ensures that a greater proportion of the input mass is converted into valuable product, reducing the overall material intensity of the manufacturing process. Additionally, the ability to recycle the polar aprotic solvents further enhances the economic viability of the route, making it an attractive option for cost-sensitive generic drug manufacturing.
- Enhanced Supply Chain Reliability: The use of widely available starting materials, such as substituted indole acetic acids and sodium sulfinates, ensures a robust and diversified supply base that is less susceptible to geopolitical or logistical shocks. Unlike proprietary reagents that may be sourced from a single vendor, these commoditized inputs can be procured from multiple qualified suppliers, providing procurement teams with greater negotiating power and security of supply. The mild reaction conditions also reduce the wear and tear on reactor vessels and heating systems, extending equipment lifespan and reducing maintenance downtime. This reliability is essential for maintaining continuous production schedules and meeting the rigorous delivery commitments expected by global pharmaceutical partners.
- Scalability and Environmental Compliance: The green chemistry credentials of this method, characterized by low toxicity reagents and reduced solvent usage, align perfectly with increasingly stringent environmental regulations worldwide. Scaling this process from gram to ton scale does not require fundamental changes to the reaction engineering, as the heat transfer and mixing requirements are manageable with standard industrial equipment. The absence of heavy metal contaminants in the final product simplifies the regulatory approval process for new drug applications, reducing the burden on quality assurance teams. Moreover, the reduced generation of hazardous waste streams lowers the environmental footprint of the facility, supporting corporate sustainability goals and enhancing the brand reputation of the manufacturing organization as a responsible chemical partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this route for their specific projects. Understanding these details is crucial for making informed decisions about process adoption and resource allocation.
Q: What are the primary advantages of this copper-catalyzed method over traditional oxidation routes?
A: This method eliminates the need for multi-step oxidation of thioethers, utilizing a direct one-pot oxidative decarboxylation that significantly reduces solvent consumption and simplifies purification.
Q: Which solvents provide the highest yield for this transformation?
A: Experimental data indicates that polar aprotic solvents such as N,N-dimethylformamide (DMF) and dimethyl sulfoxide (DMSO) are critical for success, yielding up to 90% compared to trace amounts in non-polar solvents.
Q: Is this process suitable for large-scale manufacturing of API intermediates?
A: Yes, the use of inexpensive copper catalysts, mild reaction temperatures (100-115°C), and simple workup procedures makes this route highly scalable and compliant with green chemistry principles for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Sulfone Methyl-1H-Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process expertise and a commitment to quality that spans from milligram to metric ton scales. Our team of expert chemists has extensively analyzed the copper-catalyzed oxidative decarboxylation pathway described in CN112094220B, optimizing it for industrial throughput while maintaining the stringent purity specifications required by global regulatory bodies. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our rigorous QC labs employ state-of-the-art analytical techniques to verify the identity and purity of every batch, guaranteeing that the 3-sulfone methyl-1H-indole derivatives you receive are ready for immediate use in your drug development pipelines.
We invite you to collaborate with us to leverage this green synthesis technology for your next project, unlocking significant efficiencies in your manufacturing operations. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage you to reach out today to discuss your needs,索取 specific COA data, and review our comprehensive route feasibility assessments. Let us help you navigate the complexities of fine chemical sourcing with a partner dedicated to innovation, reliability, and mutual growth in the global pharmaceutical marketplace.
