Advanced Fluorene Thiophene Derivatives for High-Performance OLED and Solar Cell Manufacturing
The rapid evolution of the organic electronics sector demands materials that combine exceptional luminous efficiency with robust thermal stability, a challenge addressed directly by the innovations disclosed in patent CN102532098B. This pivotal intellectual property introduces a novel class of fluorene and spirofluorene thiophene derivatives, engineered to overcome the limitations of traditional organic light-emitting diode (OLED) materials. By integrating the rigid fluorene and spirofluorene structural units with thiophene rings, the invention achieves a synergistic effect that enhances charge transport properties while maintaining high fluorescence quantum efficiency. For R&D directors and procurement specialists in the electronic chemical industry, this represents a significant opportunity to secure high-purity OLED material supplies that are both technically superior and commercially viable. The synthesis pathway described leverages well-established organic transformations, ensuring that the transition from laboratory discovery to commercial scale-up of complex polymer additives and small molecule emitters is seamless and reliable.
Furthermore, the strategic combination of these heterocyclic systems addresses the critical issue of morphological stability in thin-film devices, which is often a bottleneck in the lifespan of next-generation displays. The patent details a comprehensive preparation method that starts from readily accessible raw materials like 1,4-butynediol and indenocyclopentadienone, utilizing a Diels-Alder reaction as the foundational step. This approach not only simplifies the synthetic route but also minimizes the generation of hazardous waste, aligning with modern green chemistry principles that supply chain heads prioritize. As the global demand for high-resolution, energy-efficient displays continues to surge, securing a reliable agrochemical intermediate supplier or electronic chemical partner who understands these nuanced synthetic pathways becomes paramount for maintaining competitive advantage in the marketplace.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing organic electroluminescent materials often suffer from complex multi-step sequences that involve expensive transition metal catalysts and harsh reaction conditions, leading to inconsistent batch-to-batch quality. Many conventional routes rely on cross-coupling reactions that require rigorous purification to remove trace metal residues, which can act as quenching sites and drastically reduce the efficiency of the final OLED device. Additionally, the thermal stability of standard fluorene derivatives can be compromised during device operation, leading to crystallization within the emissive layer and subsequent device failure over time. These technical shortcomings translate into significant commercial risks, including increased manufacturing costs due to low yields and extended lead times for high-purity electronic chemical batches. Procurement managers frequently encounter supply disruptions when relying on suppliers who cannot consistently meet the stringent purity specifications required for advanced display technologies without incurring prohibitive costs.
The Novel Approach
In contrast, the methodology outlined in patent CN102532098B offers a streamlined and efficient pathway that circumvents these historical inefficiencies by employing a robust Diels-Alder cycloaddition followed by selective functionalization. This novel approach allows for the precise introduction of ethyl and spirofluorene groups, which sterically hinder molecular packing and effectively prevent the formation of excimers that degrade color purity. The use of sodium sulfide nonahydrate for ring closure and palladium/carbon for dehydrogenation represents a cost-effective strategy that avoids the need for exotic ligands or ultra-high vacuum conditions. 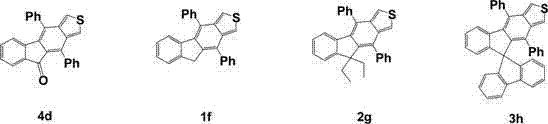 This structural versatility enables the fine-tuning of emission wavelengths and energy levels, making these derivatives highly adaptable for various layers within an OLED stack. For supply chain stakeholders, this translates to a more resilient sourcing strategy where cost reduction in electronic chemical manufacturing is achieved through simplified processing rather than compromising on material performance standards.
This structural versatility enables the fine-tuning of emission wavelengths and energy levels, making these derivatives highly adaptable for various layers within an OLED stack. For supply chain stakeholders, this translates to a more resilient sourcing strategy where cost reduction in electronic chemical manufacturing is achieved through simplified processing rather than compromising on material performance standards.
Mechanistic Insights into Diels-Alder Cycloaddition and Thiophene Fusion
The core of this synthetic innovation lies in the initial Diels-Alder reaction between 1,3-diphenyl-indene[a]pentadiene-2,8-dione and 1,4-butynediol, which constructs the fundamental carbon skeleton with high atom economy. This pericyclic reaction proceeds under thermal conditions to yield a bis-benzyl alcohol compound, establishing the necessary stereochemistry for subsequent transformations without the need for chiral auxiliaries. Following this, a series of protection and reduction steps convert the carbonyl groups into methylene or tertiary alcohol functionalities, setting the stage for the critical ring-closing events. The mechanistic precision required here ensures that impurities such as regioisomers are minimized, which is crucial for achieving the high-purity OLED material standards demanded by top-tier display manufacturers. Understanding this mechanism allows process chemists to optimize reaction parameters such as temperature and solvent polarity to maximize yield and minimize downstream purification burdens.
The final stages of the synthesis involve the nucleophilic substitution of bromomethyl groups with sulfur species followed by oxidative aromatization, a sequence that effectively fuses the thiophene ring onto the fluorene core. 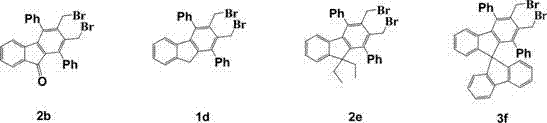 This cyclization is facilitated by the specific reactivity of the bisbenzyl bromide precursors, which react cleanly with sodium sulfide to form the sulfur-containing five-membered intermediate. The subsequent palladium/carbon catalyzed dehydrogenation removes excess hydrogen to restore aromaticity, completing the conjugated system essential for charge transport. This two-step finishing sequence is particularly advantageous because it avoids the use of toxic phosphorus reagents often found in alternative thiophene synthesis methods. For R&D teams, this mechanistic clarity provides a solid foundation for troubleshooting and scaling, ensuring that the commercial scale-up of complex polymer additives remains predictable and controllable throughout the production lifecycle.
This cyclization is facilitated by the specific reactivity of the bisbenzyl bromide precursors, which react cleanly with sodium sulfide to form the sulfur-containing five-membered intermediate. The subsequent palladium/carbon catalyzed dehydrogenation removes excess hydrogen to restore aromaticity, completing the conjugated system essential for charge transport. This two-step finishing sequence is particularly advantageous because it avoids the use of toxic phosphorus reagents often found in alternative thiophene synthesis methods. For R&D teams, this mechanistic clarity provides a solid foundation for troubleshooting and scaling, ensuring that the commercial scale-up of complex polymer additives remains predictable and controllable throughout the production lifecycle.
How to Synthesize Fluorene Thiophene Derivatives Efficiently
Executing this synthesis requires careful attention to the stoichiometry of the Diels-Alder reactants and the precise control of the bromination conditions to ensure complete conversion of the hydroxymethyl groups. The process begins with the thermal cycloaddition to form the bicyclic ketone, followed by acetal protection to safeguard the reactive centers during the reduction phase. Subsequent lithiation and alkylation steps introduce the necessary ethyl or spiro-groups, which are critical for the final material's amorphous characteristics. The detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios and temperature profiles required to replicate the high yields reported in the patent documentation. Adhering to these protocols ensures that the resulting fluorene thiophene derivatives meet the rigorous quality benchmarks necessary for integration into high-performance optoelectronic devices.
- Perform Diels-Alder cycloaddition between 1,3-diphenyl-indene[a]pentadiene-2,8-dione and 1,4-butynediol to form the bis-benzyl alcohol intermediate.
- Execute protection, reduction, and bromination steps to generate specific bisbenzyl bromide precursor compounds tailored for fluorene or spirofluorene structures.
- React precursors with sodium sulfide nonahydrate followed by palladium/carbon catalytic dehydrogenation to finalize the thiophene ring fusion.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers substantial benefits for organizations looking to optimize their supply chain reliability and reduce overall manufacturing expenditures. The reliance on commercially available starting materials such as 1,4-butynediol and common solvents like tetrahydrofuran and ethanol eliminates the dependency on specialized, single-source reagents that often create bottlenecks in production schedules. This accessibility ensures that procurement managers can negotiate better pricing and secure longer-term contracts with multiple vendors, thereby mitigating the risk of supply disruptions that can halt entire production lines. Furthermore, the simplified purification processes associated with this method reduce the consumption of chromatography media and solvents, contributing to significant cost savings in waste management and operational overhead.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of heterogeneous palladium/carbon for the final dehydrogenation step drastically simplify the workup procedure, removing the need for costly metal scavenging resins. This streamlining of the downstream processing directly lowers the cost of goods sold, allowing for more competitive pricing in the volatile electronic chemical market without sacrificing margin. Additionally, the high yields observed in the intermediate steps, such as the protection and bromination reactions, mean that less raw material is wasted, further enhancing the economic efficiency of the overall process. These factors combine to create a robust financial case for switching to this newer, more efficient synthetic methodology.
- Enhanced Supply Chain Reliability: By utilizing a synthesis route that depends on bulk commodity chemicals rather than bespoke intermediates, companies can significantly reduce lead time for high-purity electronic chemical deliveries. The robustness of the Diels-Alder reaction and the subsequent steps ensures consistent output even when scaling from kilogram to tonnage quantities, providing supply chain heads with the predictability needed for accurate demand forecasting. This reliability is crucial for maintaining continuous production of OLED panels, where any delay in material delivery can result in massive downstream losses. Consequently, partnering with a supplier capable of executing this specific chemistry ensures a steady flow of critical components.
- Scalability and Environmental Compliance: The process is inherently scalable due to the absence of extremely sensitive reagents that require cryogenic conditions or inert atmosphere gloveboxes for every step. The use of standard reflux conditions and common organic solvents facilitates easy technology transfer from pilot plants to full-scale commercial reactors, accelerating time-to-market for new display technologies. Moreover, the reduced generation of heavy metal waste aligns with increasingly stringent environmental regulations, minimizing the compliance burden and potential fines associated with hazardous waste disposal. This environmental compatibility enhances the corporate sustainability profile of manufacturers adopting this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorene thiophene synthesis technology in industrial settings. These answers are derived directly from the experimental data and beneficial effects described in the patent specification, providing clarity on performance expectations and operational requirements. Understanding these details is essential for technical teams evaluating the feasibility of integrating these materials into existing production workflows. The responses highlight the balance between high performance and practical manufacturability that defines this innovation.
Q: What are the primary applications of these fluorene thiophene derivatives?
A: These derivatives are specifically designed for organic electroluminescent devices (OLEDs) and organic small molecule solar cells, offering improved luminous performance and thermal stability.
Q: How does the spirofluorene structure benefit OLED manufacturing?
A: The spirofluorene structure prevents molecular crystallization in the solid state due to its amorphous helical configuration, significantly enhancing the optical purity and operational stability of the display material.
Q: Is the synthesis route scalable for commercial production?
A: Yes, the method utilizes commercially available reagents and standard vacuum line techniques, avoiding exotic catalysts, which facilitates straightforward scale-up from laboratory to industrial manufacturing volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorene Thiophene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from R&D to mass manufacturing is smooth and efficient. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of verifying the structural integrity and optical properties of every batch of fluorene thiophene derivatives we produce. We understand the critical nature of supply continuity in the electronic materials sector and have built our operations to guarantee consistent quality and timely delivery for our global partners. Our commitment to technical excellence means we can adapt the patented synthesis route to meet your specific volume requirements while maintaining the highest standards of safety and environmental stewardship.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current material sourcing strategy. By collaborating with us, you can access specific COA data and route feasibility assessments that demonstrate exactly how this technology can enhance your product lineup. Let us help you secure a competitive edge in the OLED market with materials that deliver superior performance and reliability. Reach out today to discuss how we can support your long-term growth objectives with our advanced chemical manufacturing capabilities.
