Advanced Synthesis of Fluorene-Spirofluorene Thiophene Derivatives for High-Performance OLEDs
Introduction to Patent CN102532098B
The rapid evolution of the organic electronics sector demands materials that offer superior thermal stability and luminous efficiency, particularly for next-generation display technologies. Patent CN102532098B, filed in 2014, introduces a groundbreaking class of fluorene and spirofluorene thiophene derivatives designed to meet these rigorous standards. This intellectual property outlines a novel synthetic pathway that fuses the robust thermal properties of fluorene and spirofluorene scaffolds with the excellent charge-transport capabilities of thiophene rings. By integrating these structural units directly through ring fusion rather than simple linkage, the invention achieves a unique molecular architecture that minimizes aggregation and maximizes glass transition temperatures. For R&D directors and procurement specialists in the optoelectronic industry, this technology represents a significant leap forward in creating reliable organic electroluminescent material supplier solutions that can withstand the operational stresses of modern OLED and organic solar cell applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of blue-emitting OLED materials has relied heavily on connecting oligothiophene chains to spirobifluorene cores through single bonds or linkers, as seen in prior art by researchers like Pei Jian and Huang Wei. While these conventional approaches successfully created conjugated systems, they often suffered from inherent drawbacks related to molecular flexibility and solid-state packing. The rotational freedom around the linking bonds could lead to non-radiative decay pathways, reducing fluorescence quantum efficiency. Furthermore, linear oligomers tended to crystallize over time or under thermal stress, leading to phase separation in thin films which drastically shortens device lifespan. The synthesis of these linked structures also frequently required complex cross-coupling reactions that were sensitive to moisture and oxygen, complicating the manufacturing process and increasing the cost reduction in display material manufacturing efforts due to low yields and difficult purification requirements.
The Novel Approach
In stark contrast, the methodology disclosed in CN102532098B employs a direct ring-fusion strategy that fundamentally alters the physical properties of the resulting material. The core innovation lies in the use of a Diels-Alder cycloaddition between 1,4-butynediol and indenocyclopentadienone to construct a rigid bis-benzyl alcohol framework. This scaffold is then elaborated into various fluorene and spirofluorene precursors before undergoing a final sulfurization and aromatization sequence. This approach locks the thiophene ring into the fluorene plane, creating a highly planar and rigid conjugated system that resists crystallization while maintaining high solubility due to strategic substituent placement. The result is a series of compounds, such as diphenyl-9H-fluoren[2,3-c]thiophene and its spiro-analogs, that exhibit exceptional thermal stability and improved optical purity. This structural rigidity effectively prevents the formation of excimers and excited complexes, addressing the primary failure modes of earlier generation materials.
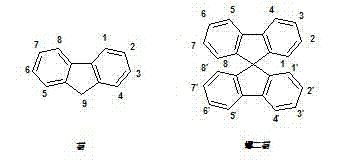
Mechanistic Insights into Diels-Alder Cyclization and Dehydrogenation
The synthetic route described in the patent is a masterclass in constructing complex polycyclic aromatic hydrocarbons through sequential functionalization. The process initiates with a thermal Diels-Alder reaction at 240°C, where 1,3-diphenyl-indenocyclopentadienone acts as the diene and 1,4-butynediol serves as the dienophile. This step is critical as it establishes the central seven-membered ring fused to the fluorene core, setting the stage for subsequent modifications. Following this, the hydroxyl groups are protected as acetals and the ketone is reduced, allowing for the precise introduction of alkyl groups or spiro-units at the 9-position. The conversion of hydroxymethyl groups to bromomethyl groups using tetrabutylammonium bromide (TBAB) and hydrobromic acid is a key activation step, transforming inert alcohols into highly reactive electrophiles ready for nucleophilic attack by sulfur species.
The final transformation involves a two-step cascade that forms the target thiophene ring. First, the bis-benzyl bromide precursors react with sodium sulfide nonahydrate in a THF/ethanol mixture. This nucleophilic substitution cyclizes the side chains to form a saturated sulfur-containing five-membered ring intermediate. The second and final step is an oxidative dehydrogenation catalyzed by palladium on carbon (Pd/C) in refluxing xylene. This step is mechanistically fascinating as it removes four hydrogen atoms to aromatize the thiophene ring, restoring full conjugation across the molecule. The use of heterogeneous Pd/C catalyst is particularly advantageous for commercial scale-up of complex OLED intermediates, as it allows for easy removal of the catalyst via simple filtration, ensuring the final high-purity electronic chemicals are free from metal contamination that could quench luminescence.
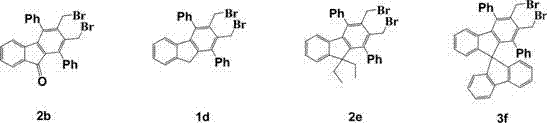
How to Synthesize Diphenyl-9H-fluoren[2,3-c]thiophene Efficiently
The synthesis of these advanced materials requires precise control over reaction conditions to maximize yield and purity. The patent details a robust protocol starting from commercially available ninhydrin and 1,3-diphenylacetone to generate the initial dione, followed by the high-temperature Diels-Alder step. Subsequent protection and reduction steps must be monitored carefully to avoid over-reduction or hydrolysis of the acetal protecting group. The bromination step typically proceeds at 60°C over 72 hours, requiring patience to ensure complete conversion of the diol to the dibromide. Finally, the sulfurization and dehydrogenation steps are performed under nitrogen atmosphere to prevent oxidation of the sensitive thiophene intermediates. For a detailed breakdown of the specific reagent quantities, temperature profiles, and workup procedures, please refer to the standardized synthesis guide below.
- Perform Diels-Alder reaction between 1,3-diphenyl-indenocyclopentadienone and 1,4-butynediol at 240°C to form the bis-benzyl alcohol core.
- Convert the alcohol intermediate into bis-benzyl bromide precursors through protection, reduction, and bromination steps using TBAB and hydrobromic acid.
- Cyclize the bromide precursors with sodium sulfide nonahydrate in THF/EtOH, followed by palladium/carbon catalytic dehydrogenation to aromatize the thiophene ring.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain heads and procurement managers, the transition from complex cross-coupling strategies to this fused-ring synthesis offers tangible logistical and financial benefits. The reliance on bulk commodity chemicals like 1,4-butynediol and sodium sulfide reduces dependency on exotic, high-cost catalysts often required for Suzuki or Stille couplings. Furthermore, the synthetic route avoids the use of toxic tin reagents or expensive phosphine ligands, simplifying waste disposal and regulatory compliance. The modular nature of the synthesis allows for the production of a library of derivatives (fluorene, 9,9-diethylfluorene, spirobifluorene) from a common intermediate, streamlining inventory management and reducing lead time for high-purity electronic chemicals. This flexibility ensures that manufacturers can rapidly respond to changing market demands for specific emission colors or stability profiles without retooling entire production lines.
- Cost Reduction in Manufacturing: The elimination of precious metal cross-coupling catalysts and the use of heterogeneous Pd/C for the final step significantly lowers the raw material cost per kilogram. Additionally, the high yields observed in the protection (85%) and reduction (83%) steps minimize material loss, contributing to substantial cost savings in organic electroluminescent material manufacturing. The simplified purification process, primarily relying on standard column chromatography and recrystallization, further reduces processing time and solvent consumption compared to multi-step oligomer syntheses.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials such as indenocyclopentadienone precursors and common solvents like THF and xylene, the risk of supply chain disruption is markedly reduced. The synthesis does not depend on single-source specialty reagents, allowing procurement teams to source inputs from multiple vendors. This diversification strengthens the supply chain against geopolitical or logistical shocks, ensuring consistent delivery of critical OLED intermediates to downstream device manufacturers.
- Scalability and Environmental Compliance: The reaction conditions are mild enough for large-scale implementation, with the highest temperature (240°C) confined to a solvent-free melt step that is easily managed in industrial reactors. The aqueous workups and standard organic extractions align with existing chemical infrastructure, facilitating a smooth transition from pilot plant to full commercial production. Moreover, the absence of heavy metal residues in the final product simplifies environmental reporting and meets the stringent purity specifications required for high-end display applications.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and performance of these fluorene-thiophene derivatives. These answers are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on the practical aspects of adopting this technology. Understanding these nuances is essential for R&D teams evaluating the material for specific device architectures.
Q: What are the key advantages of this fluorene-thiophene fusion method over traditional oligomer substitution?
A: Unlike traditional methods that attach oligothiophenes to spirobifluorenes via linkage, this patent utilizes a direct ring-fusion strategy via Diels-Alder reaction. This creates a more rigid, fully conjugated system that prevents crystallization and improves thermal stability, crucial for long-life OLED devices.
Q: How does the patent address the solubility issues common in high-molecular-weight OLED materials?
A: The synthesis introduces specific structural modifications, such as ethyl groups or spiro-configurations at the 9-position of the fluorene core. These bulky substituents disrupt planar stacking, significantly enhancing solubility in common organic solvents like chloroform and THF, which facilitates solution-processing techniques.
Q: Is the palladium/carbon dehydrogenation step scalable for industrial production?
A: Yes, the process uses heterogeneous catalysis (Pd/C) which is easily separable via filtration. The reaction conditions involve standard reflux in xylene, avoiding extreme pressures or temperatures, making it highly amenable to scale-up from gram-scale lab synthesis to multi-kilogram commercial batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorene Thiophene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance organic semiconductors play in the future of display and energy technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of CN102532098B can be translated into reliable industrial supply. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of fluorene and spirofluorene thiophene derivatives meets the exacting standards of the global optoelectronics market. Our commitment to quality ensures that your downstream OLED devices achieve maximum efficiency and longevity.
We invite you to collaborate with us to optimize your material sourcing strategy. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your next-generation product development. Let us be your partner in driving innovation through superior chemical synthesis and supply chain excellence.
