Advanced Manufacturing of Sodium Picosulfate: A Technical Breakthrough in API Intermediate Production
Advanced Manufacturing of Sodium Picosulfate: A Technical Breakthrough in API Intermediate Production
The pharmaceutical industry constantly seeks robust synthetic routes that balance high purity with operational safety, particularly for widely used active pharmaceutical ingredients like Sodium Picosulfate. Patent CN113387877B introduces a transformative preparation method that addresses longstanding challenges in the synthesis of this potent laxative agent. The disclosed technology replaces hazardous reagents and complex purification steps with a streamlined, acid-catalyzed condensation followed by a clean sulfonation process using sulfamic acid. This innovation not only enhances the chemical profile of the final product by minimizing isomeric and inorganic impurities but also offers a scalable pathway suitable for industrial manufacturing. For R&D directors and procurement specialists, understanding the mechanistic advantages of this route is critical for securing a reliable supply chain of high-quality API intermediates.
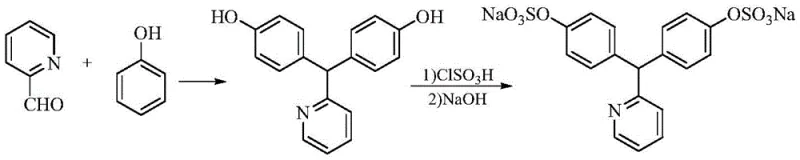
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Sodium Picosulfate has been plagued by significant technical hurdles that impact both yield and environmental compliance. Traditional routes often rely on concentrated sulfuric acid for the initial condensation of phenol and pyridine-2-carboxaldehyde, a condition that promotes the formation of difficult-to-remove isomeric impurities, specifically the 2,4'-(pyridin-2-ylmethylene)-bisphenol derivative. Furthermore, alternative pathways utilizing 2,6-dichlorophenol necessitate a dechlorination step involving nickel-aluminum alloys, which introduces severe safety risks due to the exothermic nature of the reaction and the handling of pyrophoric materials. The subsequent sulfonation using chlorosulfonic acid generates substantial quantities of inorganic salts, necessitating energy-intensive and yield-reducing desalting procedures to meet pharmacopoeial standards for chloride and sulfate residues.
The Novel Approach
In stark contrast, the methodology outlined in CN113387877B employs a sophisticated hydrochloric acid and acetic acid mixture to catalyze the condensation reaction, effectively suppressing ortho-substitution and limiting isomer formation to negligible levels. This is followed by a sulfonation step utilizing sulfamic acid in the presence of organic catalysts such as morpholine or N-methylpyrrolidone, which facilitates smooth esterification without the aggressive side reactions associated with chlorosulfonic acid. By eliminating the need for hazardous metal reductions and minimizing inorganic salt generation, this novel approach simplifies the downstream processing significantly. The result is a process that not only improves the overall yield of the intermediate and final product but also drastically reduces the environmental footprint and operational complexity typically associated with legacy manufacturing protocols.
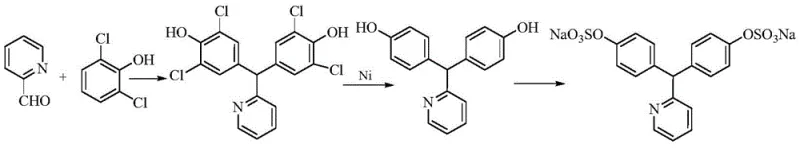
Mechanistic Insights into Acid-Catalyzed Condensation and Sulfamic Acid Sulfonation
The core of this technological advancement lies in the precise control of reaction kinetics and thermodynamics during the condensation phase. By optimizing the molar ratio of phenol to pyridine-2-carboxaldehyde and maintaining a reaction temperature between 40-50°C within the specific HCl-acetic acid matrix, the electrophilic aromatic substitution is directed preferentially to the para-position. This selectivity is paramount because the structural similarity between the desired 4,4'-isomer and the unwanted 2,4'-isomer makes separation via crystallization extremely challenging. The new solvent system modulates the acidity to a level that activates the aldehyde sufficiently for reaction while preventing the excessive protonation that leads to random ortho-attack, thereby ensuring the isomeric impurity content remains consistently below 0.5%, a threshold difficult to achieve with sulfuric acid catalysis.
Furthermore, the choice of sulfamic acid as the sulfonating agent fundamentally alters the impurity profile of the final API. Unlike chlorosulfonic acid, which decomposes to release hydrogen chloride and generate sulfate salts upon neutralization, sulfamic acid reacts cleanly to form the sulfate ester with minimal byproduct formation. This chemical elegance prevents the generation of oxidation impurities that typically arise during the high-temperature distillation steps required to remove water in traditional processes. The structural integrity of the molecule is preserved, avoiding the formation of N-oxide derivatives or other degradation products that compromise stability. Consequently, the final product exhibits superior purity with monosubstituted impurities controlled to less than 0.1%, meeting the rigorous demands of global regulatory bodies without the need for extensive chromatographic purification.
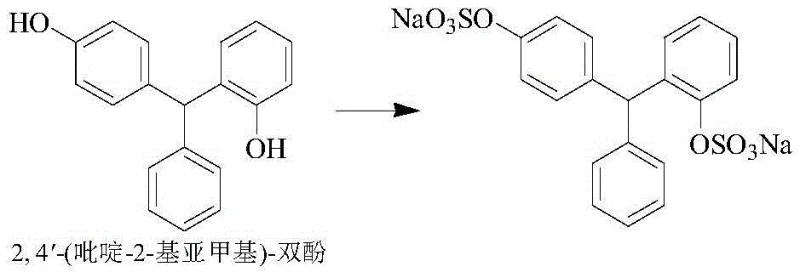
How to Synthesize Sodium Picosulfate Efficiently
The synthesis protocol described in the patent provides a clear roadmap for producing pharmaceutical-grade Sodium Picosulfate with enhanced efficiency and safety. The process begins with the careful preparation of the reaction medium, where the specific ratio of hydrochloric acid to acetic acid is critical for maximizing the yield of the bisphenol intermediate. Following the isolation and recrystallization of this intermediate, the sulfonation step requires precise temperature control and the addition of specific organic catalysts to drive the reaction to completion without degradation. The detailed标准化 synthesis steps见下方的指南 ensure that operators can replicate the high purity and yield metrics reported in the patent examples, facilitating a smooth transition from laboratory scale to commercial production.
- Condense phenol and pyridine-2-carboxaldehyde using a hydrochloric acid and acetic acid mixture at 40-50°C to form the bisphenol intermediate.
- React the purified intermediate with sulfamic acid in the presence of organic catalysts like morpholine or N-methylpyrrolidone.
- Neutralize with alkali, filter, and recrystallize from aqueous ethanol to obtain high-purity Sodium Picosulfate with minimal inorganic residues.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates into tangible strategic benefits regarding cost stability and supply continuity. The elimination of hazardous reagents like nickel-aluminum alloys and chlorosulfonic acid reduces the regulatory burden and insurance costs associated with handling dangerous chemicals, leading to a more resilient manufacturing operation. Additionally, the simplified workup procedure, which avoids complex desalting and repeated recrystallization cycles, shortens the overall production cycle time. This efficiency gain allows for faster throughput and better responsiveness to market demand fluctuations, ensuring that customers receive their orders without the delays often caused by bottlenecked purification stages in traditional facilities.
- Cost Reduction in Manufacturing: The shift to sulfamic acid eliminates the massive generation of inorganic waste salts, which significantly lowers the cost of waste treatment and disposal. By removing the need for expensive and yield-losing desalting steps, the overall material utilization is improved, directly impacting the cost of goods sold. Furthermore, the higher yield of the intermediate means less raw material is required per kilogram of final product, providing a clear economic advantage over legacy methods that suffer from poor atom economy and extensive purification losses.
- Enhanced Supply Chain Reliability: The reliance on readily available and stable starting materials like phenol and pyridine-2-carboxaldehyde ensures a robust supply chain that is less susceptible to the volatility seen with specialized halogenated precursors. The mild reaction conditions reduce equipment corrosion and maintenance downtime, allowing for continuous operation over longer periods. This operational stability guarantees a consistent supply of high-purity intermediates, mitigating the risk of stockouts that can disrupt downstream API formulation and drug product manufacturing schedules.
- Scalability and Environmental Compliance: The process is inherently safer and more environmentally friendly, aligning with modern green chemistry principles and strict environmental regulations. The absence of heavy metal catalysts and the reduction of acidic wastewater simplify the effluent treatment process, making it easier to obtain and maintain environmental permits for large-scale production. This compliance advantage future-proofs the manufacturing site against tightening regulatory frameworks, ensuring long-term viability and uninterrupted service to global pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and quality parameters of this advanced synthesis method. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, offering clarity on how this process outperforms conventional techniques in terms of impurity control and operational safety. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios.
Q: How does the new method control isomeric impurities compared to traditional sulfuric acid catalysis?
A: By utilizing a hydrochloric acid and acetic acid system instead of concentrated sulfuric acid, the process significantly suppresses the formation of the 2,4'-isomer impurity, reducing its content to below 0.5% compared to over 2.0% in conventional methods.
Q: What are the advantages of using sulfamic acid over chlorosulfonic acid for sulfonation?
A: Sulfamic acid acts as a cleaner sulfonating agent that minimizes the generation of inorganic salts like chlorides and sulfates. This eliminates the need for complex and yield-reducing repeated desalting steps required when using chlorosulfonic acid.
Q: Does this process avoid the safety risks associated with nickel-aluminum alloy reduction?
A: Yes, unlike routes starting with dichlorophenol which require dangerous dechlorination using nickel-aluminum alloys, this direct condensation route operates under mild acidic conditions without hazardous metal reductions, enhancing operational safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sodium Picosulfate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying cutting-edge synthetic methodologies to deliver superior pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN113387877B are fully realized in practical, large-scale operations. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch of Sodium Picosulfate meets the highest international standards for isomer content and inorganic residues.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your product quality while driving down overall production costs.
