Optimizing Pazopanib Intermediate Production: A Technical Breakthrough in Safe Manufacturing
Optimizing Pazopanib Intermediate Production: A Technical Breakthrough in Safe Manufacturing
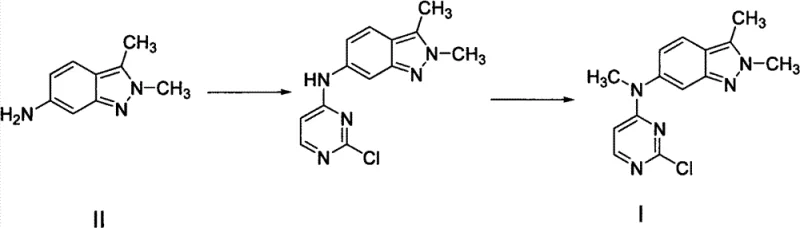
The pharmaceutical industry constantly seeks robust, scalable, and safe pathways for the synthesis of complex oncology agents like Pazopanib Hydrochloride. A pivotal advancement in this domain is detailed in patent CN103373963A, which discloses a novel method for preparing the key intermediate 2,3-dimethyl-N-(2-chloropyrimidin-4-yl)-N-methyl-2H-indazol-6-amine. This technical insight report analyzes the strategic shift from hazardous traditional methylation protocols to a safer, cost-effective reductive amination strategy. By leveraging intermediates III and IIIa, manufacturers can achieve high-purity outputs while mitigating significant supply chain risks associated with volatile alkylating agents. For R&D directors and procurement leaders, understanding this synthetic evolution is critical for securing a reliable pharmaceutical intermediate supplier capable of delivering consistent quality at commercial scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-methylated indazole derivatives relied heavily on direct alkylation strategies that pose substantial operational and environmental challenges. As illustrated in prior art routes, the transformation of the primary amine precursor often necessitates the use of methyl iodide as the methylating source. Methyl iodide is not only highly toxic and volatile but also presents severe handling risks in large-scale reactors, requiring specialized containment infrastructure. Furthermore, these conventional methods frequently employ cesium carbonate as the base to drive the reaction, a reagent that is prohibitively expensive compared to standard inorganic bases. The combination of hazardous reagents and high-cost inputs creates a bottleneck for cost reduction in API manufacturing, forcing producers to absorb significant overheads for safety compliance and waste disposal.
The Novel Approach
In stark contrast, the methodology described in CN103373963A introduces a paradigm shift by utilizing a two-step reductive amination sequence followed by nucleophilic substitution. Instead of direct methylation, the process first condenses the amine precursor with paraformaldehyde in the presence of an alkali metal, forming an imine intermediate (IIIa). This is subsequently reduced using common hydride sources like sodium borohydride to yield the N-methyl amine (III). This approach completely circumvents the need for methyl iodide, replacing it with inexpensive, solid paraformaldehyde and readily available alkali metals like sodium or potassium. The final coupling with 2,4-dichloropyrimidine utilizes mild bases such as sodium bicarbonate, drastically lowering raw material costs. This novel pathway not only enhances safety profiles but also streamlines the purification process, making it ideal for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Reductive Amination and Coupling
The core of this technological breakthrough lies in the efficient generation of the N-methyl moiety via a Schiff base mechanism. In the first stage, the primary amine of compound II reacts with paraformaldehyde under basic conditions in a protic solvent such as methanol or ethanol. The alkali metal facilitates the formation of the imine intermediate IIIa, a transient species that is immediately subjected to reduction. By employing sodium borohydride or lithium aluminum hydride at temperatures ranging from 50°C to 100°C, the C=N bond is selectively reduced to a C-N single bond without affecting other sensitive functional groups on the indazole ring. This selectivity is paramount for maintaining high product purity and minimizing the formation of over-alkylated byproducts or polymeric impurities that often plague direct alkylation reactions.
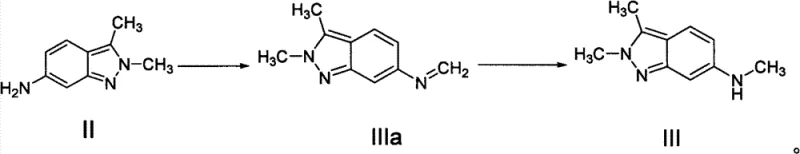
Following the successful formation of intermediate III, the final step involves a nucleophilic aromatic substitution with 2,4-dichloropyrimidine. The secondary amine acts as a nucleophile, attacking the electron-deficient pyrimidine ring. The use of weak inorganic bases like sodium bicarbonate or sodium carbonate in polar aprotic or alcoholic solvents ensures that the reaction proceeds smoothly at moderate temperatures (50°C to 100°C). This mild condition prevents the hydrolysis of the chloro-pyrimidine moiety, a common side reaction in harsher alkaline environments. The result is a highly crystalline product with HPLC purity exceeding 99%, demonstrating the robustness of the impurity control mechanism inherent in this specific reaction design. Such mechanistic precision is essential for meeting the stringent regulatory requirements of global oncology drug markets.
How to Synthesize N-Methyl Indazole Derivatives Efficiently
Implementing this synthesis requires precise control over stoichiometry and reaction parameters to maximize yield and safety. The process begins with the careful addition of alkali metals to protic solvents to generate the active alkoxide species in situ, followed by the introduction of the amine and paraformaldehyde. The subsequent reduction step must be managed to control exotherms, ensuring the safe evolution of hydrogen gas. Finally, the coupling reaction benefits from optimized solvent mixtures, such as THF and ethanol, to balance solubility and reaction kinetics. For detailed operational parameters, temperature gradients, and workup procedures, please refer to the standardized synthesis guide below.
- Condense 2,3-dimethyl-2H-indazol-6-amine with paraformaldehyde in a protic solvent using an alkali metal catalyst.
- Reduce the resulting imine intermediate using sodium borohydride or lithium aluminum hydride to form the N-methyl amine.
- Perform nucleophilic substitution with 2,4-dichloropyrimidine in the presence of a mild inorganic base to yield the final intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this novel synthetic route offers transformative benefits that extend beyond simple yield improvements. By eliminating the dependency on methyl iodide, manufacturers remove a significant regulatory hurdle and reduce the need for specialized hazardous material storage and transport logistics. This simplification directly translates to enhanced supply chain reliability, as the sourcing of paraformaldehyde and alkali metals is far more stable and geographically diverse than that of specialized alkylating agents. Furthermore, the substitution of expensive cesium carbonate with commodity chemicals like sodium bicarbonate results in substantial cost savings in raw material procurement, allowing for more competitive pricing structures in the final API market without compromising on quality standards.
- Cost Reduction in Manufacturing: The replacement of high-cost reagents with commodity chemicals fundamentally alters the cost structure of the intermediate. Cesium carbonate is a premium reagent with significant price volatility, whereas sodium bicarbonate and sodium borohydride are produced at massive global scales with stable pricing. Additionally, the avoidance of methyl iodide reduces the capital expenditure required for corrosion-resistant reactor linings and advanced scrubbing systems. These factors combine to lower the overall cost of goods sold (COGS), providing a distinct margin advantage for manufacturers adopting this technology in their production portfolios.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical metric for procurement managers managing long-term API contracts. The reagents utilized in this novel pathway, including methanol, ethanol, and paraformaldehyde, are bulk chemicals with established, redundant supply chains globally. This diversity minimizes the risk of production stoppages due to single-source supplier failures. Moreover, the improved safety profile reduces the likelihood of regulatory shutdowns or transportation delays associated with hazardous cargo, ensuring a consistent and predictable flow of materials to downstream formulation sites.
- Scalability and Environmental Compliance: The environmental footprint of pharmaceutical manufacturing is increasingly scrutinized by regulators and investors alike. This process generates significantly less hazardous waste compared to traditional halogenated alkylation routes. The solvents used are primarily alcohols, which are easier to recover and recycle than chlorinated alternatives. The absence of heavy metal catalysts or toxic alkylating agents simplifies wastewater treatment protocols, facilitating easier compliance with strict environmental discharge standards. This green chemistry alignment supports sustainable manufacturing goals and future-proofs the production facility against tightening environmental regulations.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its implementation. The following questions address common concerns regarding process safety, scalability, and quality control. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering a transparent view of the technology's capabilities and limitations in an industrial setting.
Q: Why is the new reductive amination route safer than traditional methylation?
A: The novel route eliminates the use of methyl iodide, a hazardous alkylating agent, and replaces expensive cesium carbonate with common alkali metals and bicarbonates, significantly improving operational safety and reducing toxic waste.
Q: What are the critical quality parameters for this intermediate?
A: High purity (HPLC >99%) is achieved through controlled recrystallization steps using ethyl acetate or mixed ether/toluene systems, ensuring minimal impurity carryover into the final API.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the use of commercially available solvents like ethanol and methanol, along with stable reagents like paraformaldehyde and sodium borohydride, makes the process highly scalable and robust for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pazopanib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient synthetic routes is essential for the long-term viability of oncology drug production. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN103373963A are fully realized in practical manufacturing environments. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of intermediate meets the exacting standards required for FDA-approved drug substances. Our commitment to quality assurance ensures that your supply chain remains uninterrupted and compliant.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your pazopanib projects. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this novel route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive edge in the global pharmaceutical market through superior chemical engineering.
