Advanced Synthesis of Tetrasubstituted Biphenyl Ligands for Asymmetric Catalysis and Commercial Scale-Up
The landscape of asymmetric catalysis is continuously evolving, driven by the demand for highly efficient chiral ligands that can facilitate the production of single-enantiomer pharmaceuticals. Patent CN101709023A introduces a significant breakthrough in this domain with the disclosure of a novel 2,2',6,6'-tetrahydroxy-3,3',5,5'-tetrasubstituted biphenyl ligand and its streamlined synthesis method. Unlike traditional chiral ligands that often require complex resolution processes, this invention provides an achiral compound that serves as a versatile precursor. Upon coordination with metals, it forms bimetallic axis chiral compounds with a single configuration, offering exceptional reactivity and stereoselectivity in various asymmetric reactions. This technological advancement addresses critical pain points in the fine chemical industry, particularly regarding the cost and complexity associated with producing high-performance catalytic systems for drug discovery and manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of axially chiral ligands, such as the widely known BINAP, has been fraught with challenges related to stereochemical control and synthetic efficiency. Conventional methods often rely on the resolution of racemic mixtures, which inherently limits the maximum theoretical yield to 50% unless dynamic kinetic resolution is employed, a process that adds significant operational complexity and cost. Furthermore, many existing routes involve harsh reaction conditions or expensive chiral auxiliaries that are difficult to recover and reuse. The structural rigidity required for effective axial chirality often necessitates multi-step syntheses with low overall atom economy. For procurement managers and supply chain heads, these inefficiencies translate into higher raw material costs, longer lead times, and increased waste disposal burdens, making the sourcing of high-quality chiral ligands a persistent bottleneck in the pharmaceutical supply chain.
The Novel Approach
The methodology outlined in CN101709023A presents a paradigm shift by utilizing an achiral biphenyl scaffold that acquires chirality only upon metal complexation. This approach eliminates the immediate need for chiral resolution during the ligand synthesis phase. The core structure, represented by general formula (1), features a 2,2',6,6'-tetrahydroxy substitution pattern that provides four coordination centers, enabling the formation of stable bimetallic complexes. 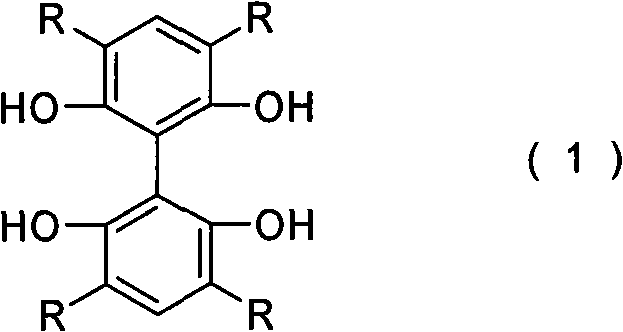 The versatility of this system is enhanced by the variable R groups, which can be alkyl, aryl, cycloalkyl, aralkyl, or halogen substituents, allowing for fine-tuning of the steric and electronic properties of the resulting catalyst. By starting from readily available achiral materials like 2,2',6,6'-tetramethoxybiphenyl, the process significantly reduces the barrier to entry for producing these high-value intermediates, offering a more direct and economically viable pathway for industrial applications.
The versatility of this system is enhanced by the variable R groups, which can be alkyl, aryl, cycloalkyl, aralkyl, or halogen substituents, allowing for fine-tuning of the steric and electronic properties of the resulting catalyst. By starting from readily available achiral materials like 2,2',6,6'-tetramethoxybiphenyl, the process significantly reduces the barrier to entry for producing these high-value intermediates, offering a more direct and economically viable pathway for industrial applications.
Mechanistic Insights into Suzuki Coupling and Demethylation Strategies
The synthesis strategy detailed in the patent relies heavily on robust carbon-carbon bond-forming reactions, specifically the Suzuki-Miyaura coupling, to install the diverse substituent groups at the 3,3',5,5' positions. In Method A, the process begins with the bromination of 2,2',6,6'-tetramethoxybiphenyl to generate a tetrahalogenated intermediate, specifically 2,2',6,6'-tetramethoxy-3,3',5,5'-tetrabromobiphenyl. This intermediate then undergoes a quadruple Suzuki coupling with aryl boronic acids (R-B(OH)2) in the presence of palladium catalysts such as palladium acetate or tetrakis(triphenylphosphine)palladium. 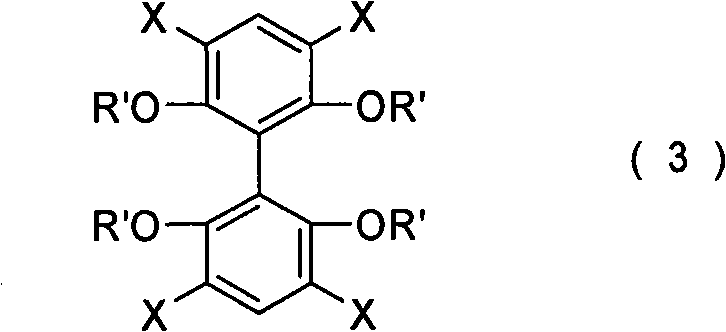 The reaction conditions are optimized for high conversion, utilizing bases like potassium carbonate or saturated sodium bicarbonate in solvents such as DMF or toluene at temperatures ranging from 50°C to 150°C. This step is crucial for introducing the steric bulk necessary for inducing axial chirality in the final metal complex, and the patent reports yields as high as 86% for this transformation, demonstrating excellent efficiency.
The reaction conditions are optimized for high conversion, utilizing bases like potassium carbonate or saturated sodium bicarbonate in solvents such as DMF or toluene at temperatures ranging from 50°C to 150°C. This step is crucial for introducing the steric bulk necessary for inducing axial chirality in the final metal complex, and the patent reports yields as high as 86% for this transformation, demonstrating excellent efficiency.
Following the construction of the substituted biphenyl skeleton, the final critical step involves the deprotection of the methoxy groups to reveal the active hydroxyl functionalities. This is achieved through a demethylation reaction using strong Lewis acids, with boron tribromide (BBr3) being the preferred reagent. The reaction is typically conducted in dichloromethane at cryogenic temperatures, starting at -78°C to control exothermicity and prevent side reactions, before gradually warming to room temperature. 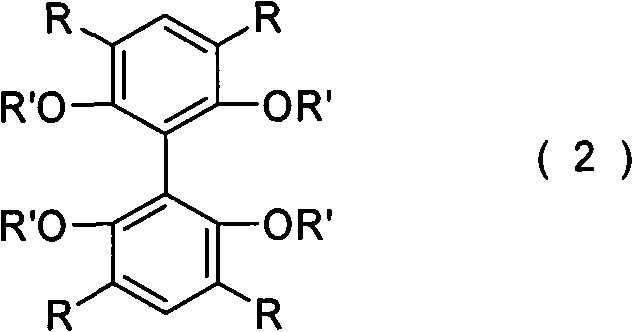 This careful temperature control ensures the integrity of the biphenyl backbone while efficiently cleaving the methyl ethers to yield the target 2,2',6,6'-tetrahydroxy product. Examples in the patent demonstrate that this demethylation step can proceed with yields exceeding 95%, ensuring that the valuable intermediate generated in the previous steps is not lost. The resulting ligand possesses four hydroxyl groups capable of coordinating with metal centers, forming the basis for highly active bimetallic catalytic systems used in reactions like hetero-Diels-Alder and carbonylene transformations.
This careful temperature control ensures the integrity of the biphenyl backbone while efficiently cleaving the methyl ethers to yield the target 2,2',6,6'-tetrahydroxy product. Examples in the patent demonstrate that this demethylation step can proceed with yields exceeding 95%, ensuring that the valuable intermediate generated in the previous steps is not lost. The resulting ligand possesses four hydroxyl groups capable of coordinating with metal centers, forming the basis for highly active bimetallic catalytic systems used in reactions like hetero-Diels-Alder and carbonylene transformations.
How to Synthesize 2,2',6,6'-tetrahydroxy-3,3',5,5'-tetrasubstituted biphenyl Efficiently
The synthesis of these advanced ligands requires precise control over reaction parameters to ensure high purity and reproducibility, which are essential for their application in sensitive asymmetric catalysis. The patent provides multiple pathways (Methods A, B, and C), with Method A being particularly suitable for large-scale production due to the reliability of Suzuki coupling chemistry. The process involves sequential bromination, cross-coupling, and demethylation, each step optimized to minimize impurities and maximize throughput. For R&D teams looking to implement this technology, understanding the stoichiometry and purification techniques described in the examples is vital. The detailed standardized synthesis steps见下方的指南。
- Bromination of 2,2',6,6'-tetramethoxybiphenyl using liquid bromine and acetic acid to form the tetrabromo intermediate.
- Suzuki coupling reaction between the tetrabromo intermediate and aryl boronic acids using palladium catalysts to introduce substituents.
- Demethylation of the tetraalkoxy intermediate using boron tribromide (BBr3) at low temperatures to yield the final tetrahydroxy ligand.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers substantial benefits for organizations aiming to optimize their supply chains for chiral intermediates. The reliance on well-established reactions like Suzuki coupling means that the process can be easily transferred to existing manufacturing facilities without the need for specialized or exotic equipment. This compatibility with standard chemical infrastructure significantly reduces capital expenditure risks and accelerates the timeline from laboratory development to commercial production. Furthermore, the high yields reported across the synthetic steps indicate a process with excellent atom economy, which directly correlates to reduced raw material consumption and lower waste generation, aligning with modern green chemistry initiatives and sustainability goals.
- Cost Reduction in Manufacturing: The elimination of chiral resolution steps in the early stages of synthesis represents a major cost-saving opportunity. Traditional methods often discard half of the material during resolution, whereas this achiral route utilizes the full mass of the starting materials effectively. Additionally, the use of commercially available boronic acids and simple demethylation reagents avoids the need for custom-synthesized chiral building blocks, which are typically expensive and have long lead times. The high yields observed in the patent examples suggest that the overall cost of goods sold (COGS) for these ligands can be significantly lower than comparable chiral ligands produced via resolution.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as 2,2',6,6'-tetramethoxybiphenyl and various aryl boronic acids, are commodity chemicals available from multiple global suppliers. This diversification of the supply base mitigates the risk of shortages that often plague specialized chiral reagents. By decoupling the ligand synthesis from chiral pool dependencies, manufacturers can maintain consistent production schedules even when specific chiral feedstocks are constrained. This stability is crucial for pharmaceutical companies that require uninterrupted supply of key intermediates to meet regulatory filing deadlines and market demand.
- Scalability and Environmental Compliance: The synthetic route is designed with scalability in mind, utilizing solvents and reagents that are manageable on a multi-kilogram to ton scale. The ability to perform the coupling reactions in common solvents like toluene or DMF simplifies solvent recovery and recycling processes. Moreover, the high selectivity of the reactions minimizes the formation of difficult-to-remove byproducts, reducing the burden on downstream purification and waste treatment systems. This operational simplicity facilitates a smoother scale-up process, allowing suppliers to rapidly increase capacity to meet surging demand for high-performance catalytic ligands without compromising on quality or environmental compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these tetrasubstituted biphenyl ligands. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this technology into your current R&D pipeline or manufacturing operations.
Q: What is the primary advantage of this ligand synthesis method?
A: The method produces an achiral ligand precursor that can be converted into a single-configuration bimetallic axis chiral compound upon metal coordination, simplifying the synthesis compared to resolving racemic mixtures.
Q: What are the typical reaction conditions for the demethylation step?
A: The demethylation is preferably conducted using boron tribromide (BBr3) in dichloromethane, starting at -78°C and gradually warming to room temperature overnight.
Q: Can this process be scaled for industrial production?
A: Yes, the use of robust reactions like Suzuki coupling and standard demethylation protocols, along with high yields reported in examples (up to 97%), indicates strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2',6,6'-tetrahydroxy-3,3',5,5'-tetrasubstituted biphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodologies described in CN101709023A for the next generation of asymmetric catalysts. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of high-purity intermediates. Our state-of-the-art facilities are equipped to handle the specific requirements of this synthesis, including low-temperature demethylation processes and palladium-catalyzed coupling reactions, all underpinned by our rigorous QC labs and stringent purity specifications. We are committed to delivering products that meet the exacting standards required for pharmaceutical and fine chemical applications.
We invite you to collaborate with us to leverage this advanced technology for your specific catalytic needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your project requirements, demonstrating how our optimized processes can reduce your overall production costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your journey from concept to commercial reality with confidence and precision.
