Advanced Synthesis of Tetrahydroxy Biphenyl Ligands for High-Purity Pharmaceutical Intermediates
Advanced Synthesis of Tetrahydroxy Biphenyl Ligands for High-Purity Pharmaceutical Intermediates
The rapid evolution of the chiral drug industry relies heavily on the development of sophisticated asymmetric synthesis methodologies, particularly those capable of generating single enantiomers with high atomic efficiency. Patent CN101709023B introduces a groundbreaking class of 2,2',6,6'-tetrahydroxy-3,3',5,5'-tetrasubstituted biphenyl ligands that serve as critical precursors for advanced catalytic systems. Unlike traditional ligands, these novel compounds are achiral in their base form but can be readily converted into bimetallic axis chiral compounds with a single configuration through the introduction of external chirality. This technological breakthrough addresses the persistent challenges in designing high-selectivity catalysts, offering a streamlined pathway for producing complex pharmaceutical intermediates with exceptional stereocontrol.
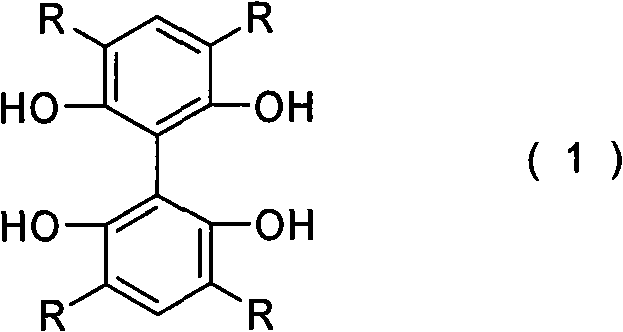
For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, understanding the structural versatility of these ligands is paramount. The general formula (1) depicted above illustrates the core scaffold where R can represent various substituents such as phenyl, p-trifluoromethylphenyl, or p-tert-butylphenyl groups. This modularity allows for fine-tuning the steric and electronic properties of the resulting catalyst, thereby optimizing performance in diverse asymmetric reactions like hetero-Diels-Alder or carbonylene processes. The ability to synthesize these structures via multiple robust pathways ensures supply chain resilience and cost-effectiveness in large-scale manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of biphenyl-based ligands has been plagued by inefficient coupling strategies and harsh reaction conditions that compromise overall yield and purity. Traditional methods often rely on direct oxidative coupling or less selective substitution reactions that generate significant amounts of regioisomeric impurities, necessitating costly and time-consuming purification steps. Furthermore, many conventional routes struggle to introduce bulky aryl substituents at the 3,3',5,5' positions without causing steric hindrance that halts the reaction prematurely. These limitations result in inconsistent batch-to-batch quality and elevated production costs, which are unacceptable for the rigorous standards required in high-purity OLED material or API intermediate manufacturing.
The Novel Approach
The patented methodology overcomes these hurdles by employing a strategic combination of Suzuki-Miyaura cross-coupling and Ullmann coupling reactions, followed by a highly efficient demethylation step. This approach allows for the precise installation of diverse aryl groups onto the biphenyl backbone under mild conditions, significantly reducing the formation of unwanted byproducts. By utilizing stable intermediates such as 2,2',6,6'-tetraalkoxy-3,3',5,5'-tetrahalobiphenyls, the process ensures high conversion rates and simplifies the isolation of the final product. This novel route not only enhances the chemical purity of the ligands but also facilitates the commercial scale-up of complex polymer additives and specialty chemicals by providing a reproducible and scalable synthetic framework.
Mechanistic Insights into Suzuki Coupling and Demethylation Strategies
The core of this synthesis lies in the palladium-catalyzed Suzuki coupling reaction, which enables the formation of carbon-carbon bonds between the halogenated biphenyl core and various boronic acid derivatives. In Method A, for instance, 2,2',6,6'-tetramethoxy-3,3',5,5'-tetrabromobiphenyl reacts with arylboronic acids in the presence of palladium acetate and triphenylphosphine. The catalytic cycle involves oxidative addition of the aryl bromide to the palladium center, transmetallation with the boronic acid species activated by a base such as potassium carbonate, and finally reductive elimination to forge the new biaryl bond. This mechanism is highly tolerant of functional groups, allowing for the introduction of electron-withdrawing groups like trifluoromethyl or bulky groups like tert-butyl without significant loss in efficiency.
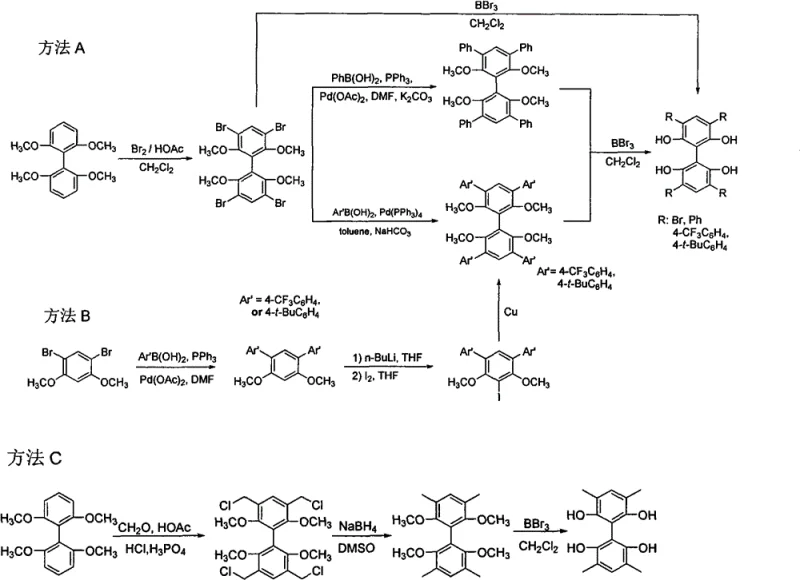
Following the construction of the substituted skeleton, the final critical transformation is the demethylation of the methoxy groups to reveal the active hydroxyl functionalities. As illustrated in the reaction schemes, this is typically achieved using boron tribromide (BBr3) in dichloromethane at low temperatures, often starting at -78°C and warming to room temperature. The Lewis acidic nature of BBr3 coordinates with the oxygen atoms of the methoxy groups, facilitating the cleavage of the methyl-oxygen bond to generate the free phenol. Experimental data from the patent indicates that this step proceeds with remarkable efficiency, achieving yields as high as 95% to 100%, which is crucial for minimizing waste and maximizing the economic viability of the process for cost reduction in electronic chemical manufacturing.
How to Synthesize 2,2',6,6'-tetrahydroxy-3,3',5,5'-tetrasubstituted biphenyl Efficiently
The synthesis of these high-value ligands requires precise control over reaction parameters to ensure optimal yield and stereochemical integrity. The process generally begins with the bromination of the starting biphenyl material, followed by the coupling steps described previously, and concludes with the deprotection sequence. Operators must maintain strict anhydrous conditions during the coupling phases to prevent catalyst deactivation and carefully monitor temperature profiles during the exothermic demethylation reaction. Detailed standardized synthetic steps see the guide below for specific stoichiometric ratios and workup procedures that guarantee reproducibility.
- Perform bromination of 2,2',6,6'-tetramethoxybiphenyl using liquid bromine and acetic acid to obtain the tetrabromo intermediate.
- Execute Suzuki coupling reaction with appropriate boronic acids using palladium catalysts to introduce aryl substituents.
- Conduct demethylation using boron tribromide (BBr3) in dichloromethane at low temperatures to yield the final tetrahydroxy ligand.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this synthesis technology offers substantial strategic benefits by streamlining the supply chain for critical chiral building blocks. The reliance on commercially available starting materials such as tetramethoxybiphenyl and common boronic acids reduces dependency on exotic reagents, thereby mitigating supply risks and stabilizing raw material costs. Moreover, the high yields reported in the patent examples suggest that the process is atom-economical, leading to significant cost reduction in API manufacturing by minimizing the volume of waste solvents and reagents that require disposal. This efficiency translates directly into a more competitive pricing structure for downstream customers seeking reliable agrochemical intermediate suppliers.
- Cost Reduction in Manufacturing: The elimination of complex resolution steps and the use of robust palladium catalysts that can be potentially recovered or used in low loadings contribute to a leaner manufacturing cost structure. By avoiding the generation of racemic mixtures that require tedious separation, the process inherently saves on energy consumption and chromatography media, driving down the overall cost of goods sold. Additionally, the high conversion rates in the final demethylation step mean that less starting material is wasted, further enhancing the economic profile of the production line.
- Enhanced Supply Chain Reliability: The versatility of the synthetic route, which offers three distinct methods (A, B, and C) to reach the target molecule, provides a built-in contingency plan for supply chain disruptions. If a specific boronic acid or coupling reagent faces availability issues, alternative pathways utilizing different precursors can be activated to maintain continuous production flow. This flexibility ensures reducing lead time for high-purity pharmaceutical intermediates and guarantees consistent delivery schedules even in volatile market conditions.
- Scalability and Environmental Compliance: The reactions described operate under relatively mild conditions and utilize standard organic solvents like dichloromethane and toluene, which are well-understood in terms of handling and recycling at an industrial scale. The high selectivity of the Suzuki coupling minimizes the formation of hazardous byproducts, simplifying wastewater treatment and aligning with stringent environmental regulations. This ease of scale-up from gram to kilogram quantities supports the commercial expansion of capacity without requiring massive capital investment in specialized reactor infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the application and handling of these biphenyl ligands in industrial settings. Understanding the nuances of storage, stability, and catalytic performance is essential for integrating these materials into existing process workflows effectively. The answers provided are derived directly from the technical specifications and experimental data outlined in the patent documentation.
Q: What are the primary advantages of this ligand synthesis method?
A: The method offers high reaction activity and stereoselectivity, utilizing robust Suzuki and Ullmann coupling reactions that are scalable for commercial production.
Q: Can this ligand be used for bimetallic catalytic systems?
A: Yes, the ligand contains four coordination centers, making it ideal for constructing bimetallic axis chiral compounds with single configurations.
Q: What are the typical yields for the demethylation step?
A: Experimental data indicates extremely high yields, often reaching up to 98-100% in the final demethylation step using boron tribromide.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2',6,6'-tetrahydroxy-3,3',5,5'-tetrasubstituted biphenyl Supplier
As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory discovery to full-scale manufacturing. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of ligand meets the exacting standards required for asymmetric catalysis in the pharmaceutical sector. We understand the critical nature of chiral purity and are committed to delivering materials that enable your team to achieve superior enantioselectivity in your final drug products.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. By collaborating with us, you gain access to specific COA data and route feasibility assessments that will empower your decision-making process. Let us help you optimize your supply chain and accelerate your time-to-market with our advanced synthesis capabilities and dedicated customer support.
