Advanced Asymmetric Catalysis for High-Purity Levalbuterol Hydrochloride Manufacturing
Advanced Asymmetric Catalysis for High-Purity Levalbuterol Hydrochloride Manufacturing
The pharmaceutical industry continuously demands more efficient, cost-effective, and environmentally sustainable pathways for producing critical active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is detailed in patent CN113801029A, which discloses a novel preparation method for Levalbuterol Hydrochloride, a potent beta-2 adrenergic receptor agonist used extensively in the treatment of bronchial asthma. This technology represents a paradigm shift from traditional racemic resolution techniques to a sophisticated asymmetric catalytic reduction strategy. By leveraging a specific chiral catalyst system, manufacturers can now achieve product purity exceeding 99.90% with single impurity and isomer levels strictly controlled below 0.05%, meeting the rigorous standards of the United States Pharmacopoeia (USP43-NF). This advancement not only enhances the therapeutic profile of the final drug by eliminating the inactive and potentially side-effect-inducing dextrorotatory isomer but also offers a robust framework for industrial scale-up.
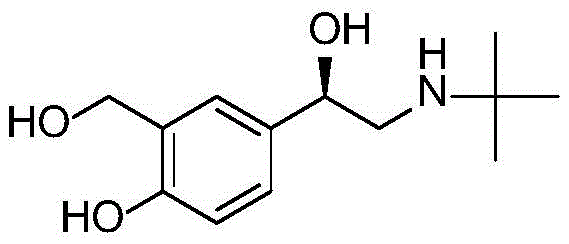
Levalbuterol, chemically known as (R)-alpha-[[ (1,1-dimethylethyl) amino] methyl]-4-hydroxy-1,3-benzenedimethanol, serves as the pharmacologically active enantiomer of salbutamol. The clinical superiority of the single enantiomer over the racemate is well-documented, offering improved bronchodilation with a reduced incidence of systemic side effects such as tremors and palpitations. Consequently, the ability to synthesize this molecule with high optical purity is of paramount importance to global pharmaceutical supply chains. The patented method addresses historical bottlenecks in synthesis by introducing a streamlined route that avoids the use of expensive chiral resolving agents and hazardous high-pressure hydrogenation, positioning it as a highly attractive option for reliable pharmaceutical intermediates supplier networks seeking to optimize their production portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Levalbuterol has been plagued by inefficiencies inherent in chiral resolution and heavy metal catalysis. Traditional approaches, such as those disclosed in British patent GB1298494A and various US patents, rely on the crystallization resolution of racemic salbutamol using agents like D-(+)-dibenzoyl tartaric acid. These methods are fundamentally flawed because they are limited by a maximum theoretical yield of 50%, necessitating the disposal or recycling of the unwanted enantiomer, which drastically inflates production costs and waste generation. Furthermore, alternative synthetic routes reported in literature, such as those utilizing rhodium complexes for asymmetric hydrogen transfer, introduce significant safety and regulatory hurdles. These processes often require high-pressure hydrogen gas and toxic reagents like aluminum trichloride or azido acid, creating dangerous operating environments and complicating the removal of trace heavy metals to meet strict ICH guidelines for elemental impurities.
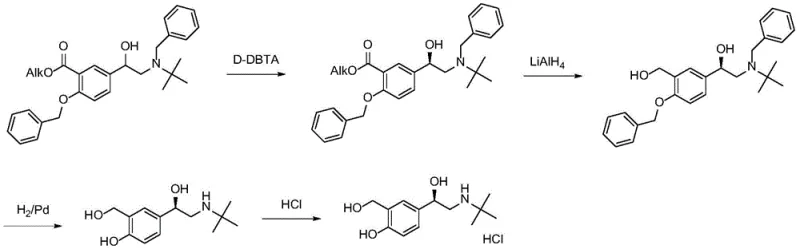
The Novel Approach
In stark contrast to these legacy methods, the invention described in CN113801029A introduces a scientifically superior pathway centered on asymmetric catalytic reduction. This novel approach utilizes a borane reduction system catalyzed by (R)-2-methyl-CBS-oxazaborolidine or similar chiral ligands to establish the critical stereocenter early in the synthesis. This catalytic system allows for the generation of large quantities of chiral product using only a small molar fraction of the chiral catalyst, thereby decoupling production volume from the cost of chiral auxiliaries. The process eliminates the need for noble metals like rhodium and avoids high-pressure reactors, significantly reducing capital expenditure and operational risk. The resulting synthetic route is not only more economical but also inherently greener, producing less hazardous waste and simplifying the purification workflow to achieve the requisite high purity without multiple recrystallization steps.
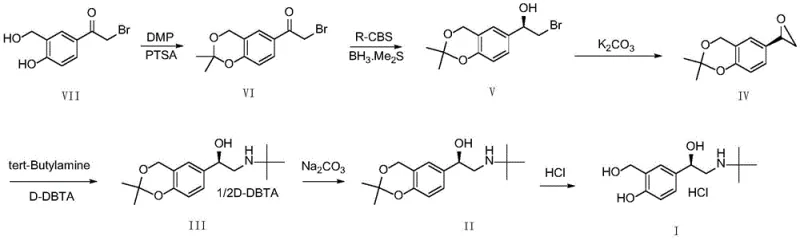
Mechanistic Insights into CBS-Catalyzed Asymmetric Reduction
The cornerstone of this innovative synthesis is the asymmetric reduction step (Step b), where the prochiral ketone intermediate (Compound VI) is converted into the chiral alcohol (Compound V). This transformation is mediated by the Corey-Bakshi-Shibata (CBS) catalyst system, which operates through a highly organized transition state. The borane species coordinates with the oxazaborolidine catalyst to form a reactive complex that delivers hydride to the carbonyl group with exceptional facial selectivity. The patent data highlights that the enantiomeric excess (ee value) of this reaction is exquisitely sensitive to moisture levels; therefore, rigorous control of water content in both the substrate (≤0.3%) and the tetrahydrofuran solvent (≤0.2%) is mandatory. Deviations from these dryness specifications can lead to catalyst decomposition and a erosion of optical purity, underscoring the need for precise process control in a commercial setting. The reaction is typically conducted at a mild temperature of 20±5°C, balancing reaction rate with stereochemical fidelity.
Following the establishment of chirality, the process employs a clever sequence of cyclization and ring-opening to install the amine functionality. The chiral alcohol is first cyclized to an epoxide (Compound IV) using anhydrous potassium carbonate, which activates the leaving group for intramolecular substitution. Subsequently, the epoxide undergoes nucleophilic attack by tert-butylamine. A critical refinement in this patent is the integration of the cyclization and amine addition steps, or the immediate subsequent resolution using D-(+)-dibenzoyl tartaric acid (D-DBTA). This specific acid is identified as uniquely capable of inducing crystallization of the desired intermediate (Compound III), whereas other organic acids fail to precipitate the product effectively. This selective crystallization acts as a powerful purification checkpoint, ensuring that any minor isomers formed during the reduction are removed before the final deprotection and salt formation steps, guaranteeing the final API meets the <0.05% isomer specification.
How to Synthesize Levalbuterol Hydrochloride Efficiently
The synthesis of Levalbuterol Hydrochloride via this patented route involves a logical sequence of six distinct chemical transformations, starting from a protected bromoketone precursor. The process is designed to maximize yield at each stage while maintaining strict control over stereochemistry and impurity profiles. Key operational parameters include the use of acetone for the initial protection step, anhydrous tetrahydrofuran for the critical reduction, and a specific ethyl acetate/water/methanol system for the final salification to prevent degradation. The following guide outlines the standardized protocol derived from the patent examples, providing a roadmap for laboratory and pilot-scale execution.
- Protect the diol precursor (Compound VII) using 2,2-dimethoxypropane and p-toluenesulfonic acid in acetone to form the acetonide intermediate (Compound VI).
- Perform asymmetric reduction on Compound VI using borane dimethyl sulfide and (R)-2-methyl-CBS-oxazaborolidine catalyst in anhydrous THF at 20°C to establish chirality.
- Cyclize the chiral alcohol to an epoxide (Compound IV) using potassium carbonate, followed by ring opening with tert-butylamine and resolution with D-DBTA.
- Hydrolyze the protecting group using sodium carbonate to obtain the free base (Compound II), then finalize with hydrochloric acid salification at 0-5°C to yield Levalbuterol Hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis method offers compelling strategic advantages that extend beyond mere technical feasibility. By transitioning away from resolution-based manufacturing, companies can effectively double their theoretical output from the same amount of raw materials, as the process is not capped by the 50% yield limit of racemic separation. This fundamental shift in stoichiometry translates directly into substantial cost savings in raw material procurement and waste disposal. Furthermore, the elimination of expensive noble metal catalysts like rhodium removes a significant variable cost driver and mitigates supply chain risks associated with the volatility of precious metal markets. The simplified operational requirements, specifically the avoidance of high-pressure hydrogenation, allow for production in standard glass-lined or stainless steel reactors, enhancing facility utilization rates and reducing the need for specialized, high-maintenance equipment.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the catalytic nature of the chirality introduction. Unlike resolution methods that consume stoichiometric amounts of expensive chiral acids, this method uses a sub-stoichiometric amount of catalyst that can be recovered or used in low loadings. Additionally, the removal of heavy metal catalysts eliminates the costly and time-consuming downstream processing steps required to scavenge trace metals to ppm levels, streamlining the purification train and reducing solvent consumption. The overall simplification of the workflow leads to a drastic reduction in manufacturing cycle times and utility costs.
- Enhanced Supply Chain Reliability: The reliance on readily available, commodity-grade reagents such as borane dimethyl sulfide and tert-butylamine ensures a stable supply chain that is less susceptible to geopolitical disruptions compared to specialized chiral resolving agents or rare earth catalysts. The robustness of the reaction conditions, particularly the tolerance for moderate temperatures and atmospheric pressure, facilitates easier technology transfer between manufacturing sites. This flexibility allows for multi-site production strategies, ensuring business continuity and reducing lead time for high-purity pharmaceutical intermediates even during periods of high market demand.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is significantly superior to prior art. The absence of high-pressure hydrogen gas removes the risk of explosion, and the exclusion of toxic reagents like aluminum trichloride reduces the burden on wastewater treatment facilities. The process generates less hazardous waste, aligning with green chemistry principles and facilitating easier regulatory approval in jurisdictions with stringent environmental laws. The scalability is proven by the patent examples which demonstrate successful execution from gram to kilogram scales with consistent purity profiles, indicating a smooth path to commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on critical process parameters and quality outcomes. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: How does this new method improve upon traditional resolution processes?
A: Traditional resolution methods often suffer from theoretical yield limits of less than 50% and require expensive resolving agents. This novel asymmetric catalytic route bypasses resolution, utilizing a catalytic amount of chiral catalyst to achieve high yields and >99.9% purity directly.
Q: What are the critical control parameters for the asymmetric reduction step?
A: Moisture control is paramount. The water content in the substrate must be ≤0.3% and in the THF solvent ≤0.2%. Additionally, maintaining the reaction temperature at 20±5°C during the dropwise addition ensures optimal enantioselectivity and minimizes isomer formation.
Q: Does this process involve hazardous heavy metal catalysts?
A: No. Unlike prior art methods that utilize noble metal rhodium complexes under high-pressure hydrogen, this process employs an organocatalytic borane reduction system. This eliminates heavy metal contamination risks and removes the need for dangerous high-pressure hydrogenation equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Levalbuterol Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of delivering high-quality intermediates that meet the exacting standards of the global pharmaceutical industry. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of Levalbuterol Hydrochloride complies with USP and EP monographs. Our dedication to quality assurance ensures that our clients receive materials that are ready for immediate formulation, minimizing their internal testing burdens and accelerating time-to-market for their final drug products.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and profitability in your API manufacturing operations. Let us be your trusted partner in navigating the complexities of modern pharmaceutical synthesis.
