Scalable Synthesis of Levalbuterol Hydrochloride: A Breakthrough in Chiral Pharmaceutical Intermediates
Scalable Synthesis of Levalbuterol Hydrochloride: A Breakthrough in Chiral Pharmaceutical Intermediates
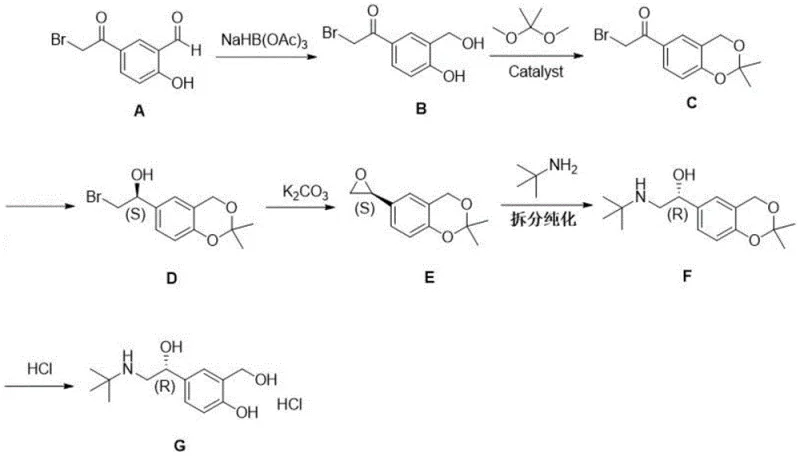
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective routes for high-value active pharmaceutical ingredients (APIs) and their intermediates. Patent CN114409552A introduces a significant advancement in the preparation of Levalbuterol Hydrochloride, the pharmacologically active (R)-enantiomer of salbutamol. Unlike traditional methods that rely on the resolution of racemates or expensive transition metal catalysis, this invention utilizes a streamlined sequence starting from 5-(2-bromoacetyl)-2-hydroxybenzaldehyde. The process integrates aldehyde reduction, acetonide protection, asymmetric carbonyl reduction, epoxidation, amination, and chiral resolution followed by deprotection. By operating under mild conditions and avoiding complex purification techniques like column chromatography, this methodology offers a compelling solution for manufacturers aiming to enhance supply chain reliability and reduce production costs in the competitive respiratory drug market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Levalbuterol Hydrochloride has been plagued by inefficiencies that hinder large-scale commercial viability. One common approach involves the chemical resolution of racemic salbutamol using chiral reagents. However, literature indicates that such methods often suffer from poor raw material utilization, with total yields after purification dropping as low as 28.5%. This inherent inefficiency makes the process economically unattractive for mass production. Another prevalent strategy employs asymmetric epoxidation of styrene derivatives using chiral borneol-based beta-diketone iron complexes. While this achieves high enantioselectivity, it relies on expensive and potentially polluting iron catalysts. Furthermore, these routes typically necessitate column chromatography for purification, a unit operation that is notoriously difficult to scale, consumes vast amounts of solvents, and creates significant bottlenecks in continuous manufacturing environments.
The Novel Approach
The methodology disclosed in patent CN114409552A fundamentally restructures the synthetic pathway to overcome these historical barriers. By initiating the synthesis with 5-(2-bromoacetyl)-2-hydroxybenzaldehyde, the route strategically installs the chiral center through asymmetric carbonyl reduction rather than late-stage resolution or epoxidation. This shift allows for the use of milder reagents and avoids the need for heavy metal catalysts that pose environmental and regulatory challenges. Crucially, the entire process is designed to achieve high purity (>99%) through crystallization and slurry washing, completely bypassing the need for column chromatography. This simplification not only drastically reduces solvent consumption and waste generation but also significantly lowers the equipment requirements, making it an ideal candidate for reliable pharmaceutical intermediates supplier operations seeking to optimize their manufacturing footprint.
Mechanistic Insights into Borane-Mediated Asymmetric Carbonyl Reduction
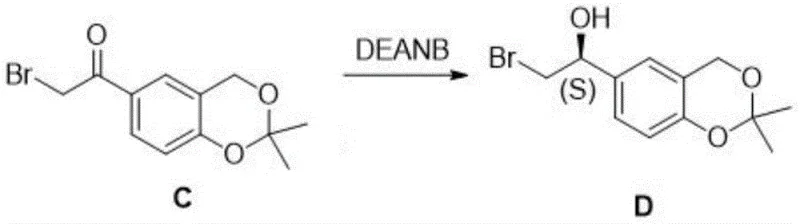
The cornerstone of this synthetic strategy is the stereoselective reduction of the ketone intermediate (Compound C) to the chiral alcohol (Compound D). This transformation is achieved using a borane complex, specifically N,N-diethylaniline borane (DEANB), in the presence of a chiral ligand, (1R,2S)-(+)-cis-1-amino-2-indanol. The mechanism involves the formation of a chiral borane species where the ligand coordinates to the boron atom, creating a sterically defined environment. When the ketone substrate approaches this complex, hydride transfer occurs preferentially from one face of the carbonyl group, dictated by the chiral pocket formed by the indanol ligand. This ensures the formation of the desired (S)-configured alcohol intermediate with high optical purity. Performing this reaction at low temperatures (0~10°C) further enhances stereocontrol by minimizing non-selective background reduction pathways.
Following the establishment of chirality, the synthesis proceeds through an intramolecular cyclization to form an epoxide intermediate (Compound E) using potassium carbonate. This step is critical as it activates the side chain for nucleophilic attack by tert-butylamine. The subsequent ring-opening reaction inverts the configuration at the chiral center, converting the (S)-epoxide into the desired (R)-amino alcohol framework found in the final Levalbuterol structure. The final purification involves forming a salt with D-(+)-dibenzoyl tartaric acid (D-DBTA). This diastereomeric salt formation leverages solubility differences to remove any minor enantiomeric impurities generated during the earlier steps, ensuring the final API meets stringent purity specifications without the need for preparative HPLC or silica gel chromatography.
How to Synthesize Levalbuterol Hydrochloride Efficiently
Implementing this synthesis requires precise control over reaction parameters, particularly temperature and stoichiometry during the asymmetric reduction and resolution steps. The process begins with the selective reduction of the aldehyde moiety in the starting material, followed by protection of the resulting diol to prevent side reactions during the subsequent ketone reduction. The critical asymmetric reduction step must be conducted under inert atmosphere to maintain the activity of the borane complex. Following the construction of the chiral backbone, the epoxide formation and amine coupling are performed under basic and thermal conditions respectively. The final stages involve chiral resolution via salt formation and acidic deprotection to reveal the free phenolic groups. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized guide below.
- Reduce 5-(2-bromoacetyl)-2-hydroxybenzaldehyde using sodium triacetoxyborohydride to form the benzyl alcohol intermediate.
- Protect the diol system using 2,2-dimethoxypropane and an acid catalyst to form the acetonide derivative.
- Perform asymmetric reduction of the ketone using a borane complex and chiral ligand to establish the (S)-configuration.
- Cyclize the bromo-alcohol to an epoxide using potassium carbonate, followed by ring-opening with tert-butylamine.
- Resolve the racemic amine using D-(+)-dibenzoyl tartaric acid and finally deprotect with HCl to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthesis route offers substantial strategic benefits beyond mere technical feasibility. The elimination of column chromatography is a primary driver for cost reduction in API manufacturing. Chromatographic purification is capital intensive, requiring large columns, significant volumes of high-purity solvents, and extensive labor for fraction collection and analysis. By replacing this with crystallization and filtration, the process dramatically simplifies the downstream processing workflow. This reduction in complexity translates directly to lower operating expenses and a smaller environmental footprint, aligning with modern green chemistry initiatives and reducing the burden on waste treatment facilities.
- Cost Reduction in Manufacturing: The avoidance of expensive transition metal catalysts, such as the iron complexes used in older asymmetric epoxidation routes, removes a significant variable cost from the bill of materials. Furthermore, the use of readily available reagents like sodium triacetoxyborohydride and common organic solvents ensures stable pricing and availability. The simplified purification train reduces energy consumption associated with solvent recovery and distillation, leading to comprehensive operational savings that enhance the overall margin profile of the final product.
- Enhanced Supply Chain Reliability: Dependence on specialized, custom-synthesized catalysts can introduce fragility into the supply chain. This new method relies on commodity chemicals and standard chiral ligands that are widely sourced from multiple global suppliers. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages. Additionally, the robustness of the reaction conditions, which tolerate mild temperatures and standard pressure, reduces the likelihood of batch failures due to equipment malfunction or thermal runaway, ensuring consistent delivery schedules for downstream formulation partners.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial mass production, with equipment requirements that fit within standard multipurpose reactor suites. The absence of heavy metal residues simplifies the regulatory filing process, as there is no need for extensive validation of metal clearance steps. This facilitates faster time-to-market for generic versions of Levalbuterol. Moreover, the reduced solvent intensity and lack of toxic metal waste streamline environmental compliance reporting and lower the costs associated with hazardous waste disposal, making the facility more sustainable and compliant with increasingly strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on purity profiles, scalability, and process safety for potential manufacturing partners.
Q: How does this new synthesis route improve upon traditional resolution methods?
A: Traditional resolution of racemic salbutamol often suffers from low theoretical yields (max 50%) and requires extensive purification. This novel route constructs the chiral center early via asymmetric reduction, avoiding the 50% yield loss inherent in late-stage resolution and eliminating the need for column chromatography.
Q: Is column chromatography required for purification in this process?
A: No. A key advantage of this patented method is that it achieves over 99% purity through crystallization and slurry purification steps alone. This eliminates the bottleneck of column chromatography, making the process highly suitable for industrial scale-up.
Q: What represents the critical chiral control step in this synthesis?
A: The critical step is the asymmetric reduction of the ketone intermediate using a borane-amine complex coordinated with a chiral ligand, specifically (1R,2S)-(+)-cis-1-amino-2-indanol. This ensures high enantioselectivity early in the synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Levalbuterol Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we understand that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands engineering excellence and rigorous quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant and finally to full-scale manufacturing is seamless. We are committed to meeting stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify chiral purity and impurity profiles at every stage of the synthesis. Our infrastructure is designed to handle the specific solvent systems and temperature controls required for the asymmetric reduction and resolution steps outlined in this novel process.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data from our pilot batches and conduct comprehensive route feasibility assessments to demonstrate how this optimized synthesis can enhance your product competitiveness. Let us partner with you to deliver high-quality Levalbuterol Hydrochloride that meets the demanding standards of the global respiratory therapy market.
