Advanced Synthesis of Pyrazole Amide Compounds for Scalable Agrochemical Production
Advanced Synthesis of Pyrazole Amide Compounds for Scalable Agrochemical Production
The global agrochemical industry faces a persistent challenge in combating pest resistance, necessitating the continuous development of novel insecticidal agents with improved efficacy and environmental profiles. Patent CN110845472B introduces a significant technological breakthrough in the preparation of pyrazole amide compounds, a class of molecules exhibiting potent insecticidal activity. This patent details a refined synthetic route that addresses critical bottlenecks in traditional manufacturing, specifically targeting the issues of low yield and complex purification that have historically hindered the cost-effective production of these valuable intermediates. By shifting the synthetic strategy towards a direct hydrolysis of a pyrazole amide nitrile precursor, the disclosed method achieves a substantial improvement in process efficiency. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, this technology represents a pivotal advancement in securing a stable supply chain for next-generation crop protection agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those described in CN104447688, often rely on oxidative pathways to generate the terminal primary amide functionality required for biological activity. These conventional routes are fraught with inefficiencies, primarily stemming from the formation of numerous byproducts during the oxidation step. For instance, comparative examples in the patent data illustrate the use of hydrogen peroxide and potassium hydroxide in ethanol, a harsh oxidative environment that frequently leads to over-oxidation or incomplete conversion. The resulting crude reaction mixtures are notoriously difficult to purify, often requiring labor-intensive column chromatography to isolate the target molecule, which is economically unviable for industrial scale-up. Furthermore, the overall yields in these traditional processes are disappointingly low, with some comparative examples reporting total yields as low as 14% over two steps, leading to significant wastage of expensive starting materials and increased production costs.
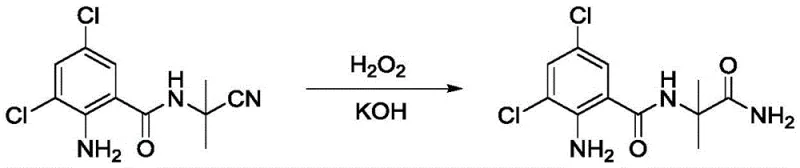
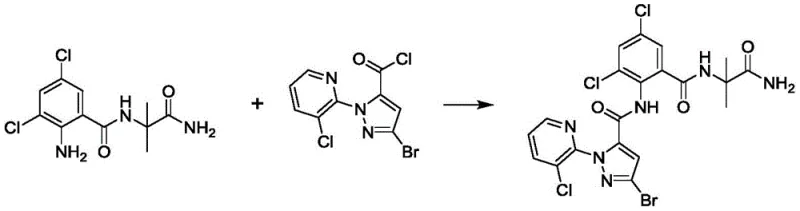
The Novel Approach
In stark contrast to the oxidative struggles of the past, the novel approach presented in patent CN110845472B employs a strategic hydrolysis of a stable nitrile intermediate. This method bypasses the unpredictable nature of oxidation by utilizing a robust nucleophilic substitution followed by a controlled acid-catalyzed hydrolysis. The core innovation lies in the conversion of the pyrazole amide nitrile compound (Formula II) directly into the target pyrazole amide (Formula I) using aqueous acid. This shift in chemistry fundamentally alters the impurity profile, drastically reducing the formation of side products. The process flow is streamlined, moving from a complex multi-step purification to a simple precipitation and filtration workflow. This not only enhances the purity of the final product but also significantly shortens the manufacturing cycle time, offering a compelling value proposition for cost reduction in pesticide manufacturing.
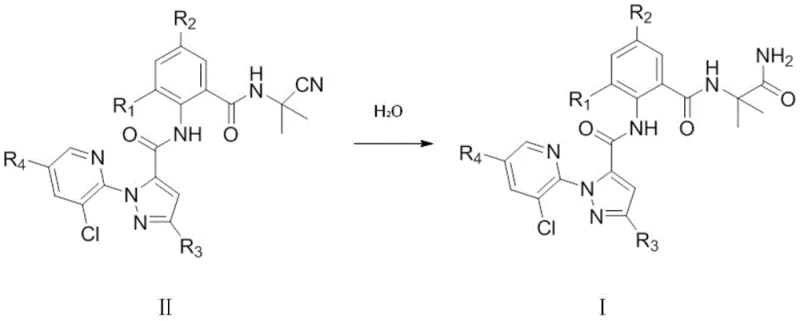
Mechanistic Insights into Acid-Catalyzed Nitrile Hydrolysis
The success of this synthetic route hinges on the precise control of the hydrolysis mechanism. The transformation of the cyano group (-CN) in Formula II to the primary amide (-CONH2) in Formula I is achieved under mildly acidic conditions, typically using 30-40 wt% sulfuric acid or concentrated hydrochloric acid. The reaction temperature is critically maintained between 35°C and 40°C. This specific thermal window is essential; it provides sufficient energy to drive the hydration of the nitrile while preventing the thermal degradation of the sensitive amide bonds elsewhere in the molecule. Mechanistically, the acid protonates the nitrogen of the nitrile group, increasing its electrophilicity and facilitating nucleophilic attack by water molecules. This leads to the formation of an imidic acid intermediate, which tautomerizes to the stable primary amide.
A crucial aspect of this mechanism is the suppression of competing dehydration and cyclization reactions. In many similar chemical environments, the proximity of the amine and carbonyl groups can lead to intramolecular cyclization, forming unwanted heterocyclic byproducts. However, the specific electronic environment created by the substituents (R1, R2, R3, R4) and the controlled acidic conditions stabilize the electron cloud around the reaction center. This stabilization effectively blocks the pathway for dehydration, ensuring that the reaction proceeds selectively towards the desired amide. The result is a reaction mixture with a remarkably clean profile, allowing for high-purity isolation without the need for chromatographic separation, a feature that is highly attractive for commercial scale-up of complex agrochemical intermediates.
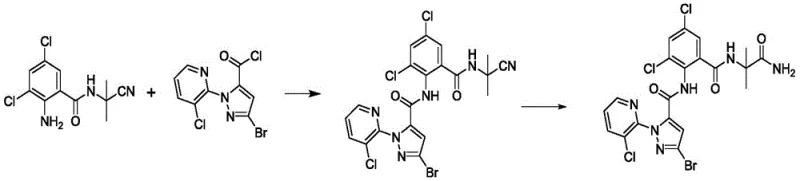
How to Synthesize Pyrazole Amide Compounds Efficiently
The synthesis of these high-value intermediates is designed for operational simplicity and robustness. The process begins with the preparation of the nitrile precursor via the coupling of a substituted 2-aminobenzamide with a pyrazole carbonyl chloride in a suitable organic solvent such as acetonitrile or toluene. Once the intermediate is secured, the critical hydrolysis step is executed under the optimized acidic conditions described previously. The detailed standardized synthetic steps, including specific reagent ratios, mixing protocols, and safety parameters, are outlined in the guide below to ensure reproducibility and safety in your pilot plant operations.
- Synthesize the pyrazole amido nitrile intermediate (Formula II) by reacting a substituted 2-aminobenzamide derivative (Formula III) with a pyrazole carbonyl chloride (Formula IV) in an organic solvent under reflux conditions.
- Perform the key hydrolysis reaction by treating the nitrile intermediate with a concentrated acidic solution (30-40% HCl or H2SO4) at a controlled temperature of 35-40°C to convert the cyano group to a primary amide.
- Isolate the final product by cooling the reaction mixture to 0-10°C to induce precipitation, followed by filtration and drying to obtain the target pyrazole amide compound with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible benefits that extend beyond mere chemical yield. The elimination of complex purification steps translates directly into reduced operational expenditures and a more resilient supply chain. By avoiding the use of expensive silica gel and large volumes of eluents required for column chromatography, the process significantly lowers the consumption of consumables and solvents. This reduction in material usage not only cuts costs but also simplifies waste management, aligning with increasingly stringent environmental regulations. The streamlined workflow ensures that production batches can be turned around more quickly, enhancing the responsiveness of the supply chain to market demands.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the drastic simplification of the downstream processing. Traditional methods often incur high costs due to low yields and the necessity for chromatographic purification, which is both time-consuming and resource-intensive. By achieving high conversion rates and enabling isolation via simple crystallization or precipitation, this method removes the most expensive unit operations from the production line. The high atom economy of the hydrolysis reaction further ensures that raw material utilization is maximized, reducing the cost per kilogram of the final active ingredient precursor.
- Enhanced Supply Chain Reliability: The robustness of the hydrolysis reaction contributes to a more predictable manufacturing schedule. Unlike oxidative reactions that can be sensitive to trace impurities or slight variations in temperature, this acid-catalyzed process is forgiving and consistent. This reliability minimizes the risk of batch failures, ensuring a steady flow of materials to downstream formulation plants. Furthermore, the starting materials (Formula III and IV) are chemically stable and readily available, reducing the risk of supply disruptions associated with exotic or unstable reagents.
- Scalability and Environmental Compliance: The process is inherently designed for scale. The use of common industrial acids and the absence of heavy metal catalysts or hazardous oxidants make the technology easier to license and operate in large-scale reactors. The simplified workup generates less hazardous waste, lowering the burden on wastewater treatment facilities. This environmental compatibility is a key factor for long-term sustainability, allowing manufacturers to maintain compliance with global green chemistry initiatives while scaling production from pilot kilograms to multi-ton commercial volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyrazole amide synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, offering clarity on process capabilities and product quality standards.
Q: How does the novel hydrolysis method improve purity compared to prior art?
A: The novel method utilizes a direct acidic hydrolysis of the nitrile intermediate at mild temperatures (35-40°C), which significantly suppresses side reactions such as dehydration and cyclization that commonly plague oxidative methods, resulting in a much cleaner impurity profile.
Q: What are the critical reaction conditions for the hydrolysis step?
A: The hydrolysis is optimally conducted in a strong acidic environment, specifically using 30-40 wt% sulfuric acid or concentrated hydrochloric acid, maintaining a strict temperature range of 35-40°C to ensure complete conversion without degrading the sensitive amide structure.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is highly scalable because it eliminates complex purification steps like column chromatography; the final product is isolated simply by cooling and precipitation, making it ideal for multi-ton production campaigns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazole Amide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the competitive agrochemical landscape. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN110845472B and is fully equipped to leverage these advancements for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. Our state-of-the-art facilities are designed to handle the specific requirements of acid-catalyzed hydrolysis and precipitation workflows, guaranteeing stringent purity specifications and rigorous QC labs testing for every batch we produce.
We invite you to collaborate with us to optimize your supply chain for these essential insecticide intermediates. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our mastery of this advanced synthesis method can drive value and efficiency for your organization.
