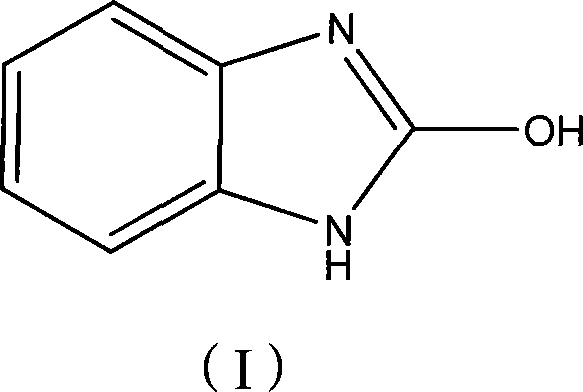
Advanced Manufacturing of 2-Hydroxybenzimidazole: A Safe and Scalable Triphosgene Route for Global Markets
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes for critical heterocyclic intermediates, and the production of 2-hydroxybenzimidazole stands as a prime example of process evolution. Patent CN101209996B introduces a refined chemical synthesis method that utilizes o-phenylenediamine and bis(trichloromethyl) carbonate, commonly known as triphosgene, as the primary raw materials. This innovation represents a significant departure from legacy methodologies, shifting the paradigm from harsh, high-temperature conditions to a controlled, mild environment ranging from -10°C to 80°C. By employing organic bases as acid scavengers within standard organic solvents, this process achieves reaction times between 1 and 13 hours, delivering a product that serves as a pivotal precursor for active pharmaceutical ingredients like mizolastine. The strategic adoption of this technology offers manufacturers a pathway to enhance both operational safety and economic efficiency without compromising on the structural integrity of the final molecule.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 2-hydroxybenzimidazole has been plagued by significant technical hurdles that impact both safety and yield consistency. Traditional protocols often relied on the condensation of o-phenylenediamine with urea in high-boiling solvents such as pentyl alcohol, dimethylformamide (DMF), or ethylene glycol. These processes necessitated prolonged refluxing at elevated temperatures, creating potential safety hazards associated with thermal runaway and the handling of hot, viscous reaction masses. Furthermore, alternative routes involving ethyl o-aminobenzoate and hydroxylamine under the influence of benzenesulfonyl chloride were characterized by excessive step counts and lower overall yields. These legacy methods not only suffered from moderate efficiency but also generated substantial environmental pollution, making them increasingly untenable in a regulatory landscape that demands greener chemistry and stricter waste management protocols.
The Novel Approach
In stark contrast to these outdated techniques, the novel approach detailed in the patent leverages the reactivity of bis(trichloromethyl) carbonate to facilitate a rapid and clean cyclization. This method operates under remarkably mild conditions, typically between -5°C and 30°C when optimized, which drastically reduces the energy input required for heating and cooling cycles. The use of triphosgene acts as a safe, solid substitute for phosgene gas, allowing for precise stoichiometric control during the addition phase. By selecting appropriate organic bases like pyridine or triethylamine, the reaction efficiently neutralizes the hydrogen chloride byproduct in situ, driving the equilibrium towards the desired 2-hydroxybenzimidazole formation. This streamlined process eliminates the need for complex purification steps often required to remove high-boiling solvents, thereby simplifying the isolation of the filter cake and enhancing the overall throughput of the manufacturing line.
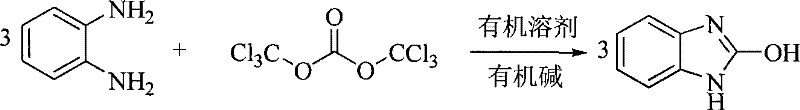
Mechanistic Insights into Triphosgene-Mediated Cyclization
The core of this synthetic breakthrough lies in the mechanistic interaction between the diamine and the carbonyl source provided by triphosgene. Upon dissolution in an organic solvent, the organic base activates the system by coordinating with the electrophilic centers, while the o-phenylenediamine acts as a nucleophile. As the triphosgene solution is added dropwise, it decomposes in situ to release phosgene equivalents which immediately react with the amine groups. This initial acylation forms an unstable intermediate that rapidly undergoes intramolecular cyclization to close the imidazole ring. The presence of the organic base is critical here, as it sequesters the hydrochloric acid generated during the substitution, preventing the protonation of the amine nucleophiles which would otherwise halt the reaction progress. This delicate balance ensures that the cyclization proceeds smoothly to form the stable aromatic benzimidazole system without the formation of polymeric byproducts or open-chain urea derivatives.
From an impurity control perspective, the mild temperature profile of this reaction is instrumental in maintaining high product purity, often exceeding 98% as demonstrated in specific embodiments. High-temperature methods often promote side reactions such as oxidation of the amine or decomposition of the solvent, leading to colored impurities that are difficult to remove. By keeping the reaction temperature low, specifically within the preferred range of -5°C to 30°C, the kinetic energy of the molecules is managed to favor the desired cyclization pathway over competing degradation routes. Furthermore, the choice of solvent plays a pivotal role; halogenated solvents like dichloromethane provide excellent solubility for the reactants while remaining inert to the reaction conditions, ensuring that the final filter cake consists primarily of the target molecule with minimal solvent inclusion or adduct formation.
How to Synthesize 2-Hydroxybenzimidazole Efficiently
To implement this synthesis effectively, operators must adhere to a precise sequence of mixing and temperature control to maximize yield and safety. The process begins with the dissolution of the diamine and base, followed by the controlled addition of the triphosgene solution, ensuring that the exotherm is managed effectively. Detailed standardized operating procedures regarding specific molar ratios and washing protocols are essential for reproducibility on a commercial scale. For the complete technical workflow and specific parameter optimization, please refer to the structured guide below.
- Dissolve o-phenylenediamine and an organic base (such as pyridine or triethylamine) in a suitable organic solvent like dichloromethane or chloroform.
- Cool the mixture to a temperature between -10°C and 30°C, then slowly add a solution of bis(trichloromethyl) carbonate (triphosgene) while maintaining stirring.
- Continue stirring the reaction mixture for 1 to 13 hours, then filter the resulting precipitate, wash with solvent and water, and dry to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this triphosgene-based methodology offers tangible benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the supply chain for raw materials; o-phenylenediamine and triphosgene are commodity chemicals available from multiple global sources, reducing the risk of single-supplier dependency. Additionally, the elimination of high-boiling solvents like DMF or ethylene glycol simplifies the recovery and recycling infrastructure, as lower boiling point solvents like dichloromethane can be distilled and reused with significantly less energy expenditure. This operational efficiency translates directly into a leaner cost structure, as the facility can achieve higher batch turnover rates without the lengthy cooldown and cleaning cycles associated with traditional high-temperature reflux processes.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the removal of expensive and energy-intensive unit operations. By avoiding prolonged heating at high temperatures, the facility sees a substantial reduction in utility costs associated with steam and cooling water. Furthermore, the high selectivity of the reaction minimizes the loss of valuable starting materials to side products, ensuring that the theoretical yield is closely approached in practice. The simplicity of the work-up procedure, which involves basic filtration and washing rather than complex distillation or chromatography, further lowers the labor and equipment maintenance costs per kilogram of produced intermediate.
- Enhanced Supply Chain Reliability: The robustness of this chemical route ensures consistent delivery schedules, a critical factor for downstream API manufacturers. Because the reaction conditions are mild and forgiving, the risk of batch failure due to thermal excursions or equipment malfunction is significantly mitigated. The use of stable, solid triphosgene instead of gaseous phosgene also simplifies logistics and storage requirements, removing the need for specialized high-pressure containment systems. This reliability allows supply chain planners to maintain lower safety stocks while confidently meeting the just-in-time delivery demands of international pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is straightforward due to the absence of extreme physical parameters. The reaction does not require specialized high-pressure reactors or exotic metallurgy, allowing it to be run in standard glass-lined or stainless steel vessels found in most multipurpose chemical plants. From an environmental standpoint, the reduction in "three wastes" is a major compliance advantage; the process generates less wastewater and avoids the creation of difficult-to-treat organic sludge. This aligns perfectly with modern green chemistry initiatives, facilitating easier permitting and reducing the long-term liability associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on operational parameters and quality expectations. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this method for large-scale production.
Q: What are the primary advantages of using triphosgene over urea for synthesizing 2-hydroxybenzimidazole?
A: The use of triphosgene allows for significantly milder reaction conditions compared to the traditional urea method, which requires high-boiling solvents and prolonged refluxing. This new approach minimizes thermal degradation risks, improves operational safety, and typically yields higher purity products with simplified downstream processing.
Q: Which organic solvents are most effective for this cyclization reaction?
A: According to the patented process, halogenated solvents such as methylene dichloride (DCM) and trichloromethane (chloroform) are highly preferred due to their ability to dissolve reactants effectively while maintaining mild reaction temperatures. Ether-based solvents like 2-methyltetrahydrofuran are also viable alternatives that offer good solubility and ease of removal.
Q: How does this method impact the environmental footprint of production?
A: This synthesis route generates significantly less 'three wastes' (wastewater, waste gas, and solid waste) compared to older methods involving ethyl o-aminobenzoate or high-temperature urea condensation. The milder conditions reduce energy consumption, and the use of recoverable organic solvents contributes to a more sustainable manufacturing profile.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydroxybenzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex heterocycles requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We utilize stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-hydroxybenzimidazole meets the exacting standards required for pharmaceutical intermediates, minimizing the risk of downstream processing issues for our partners.
We invite global partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. Our technical procurement team is ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can support your supply chain goals. Contact us today to discuss how we can collaborate to secure a stable, high-quality supply of this critical building block for your next generation of therapeutic agents.
