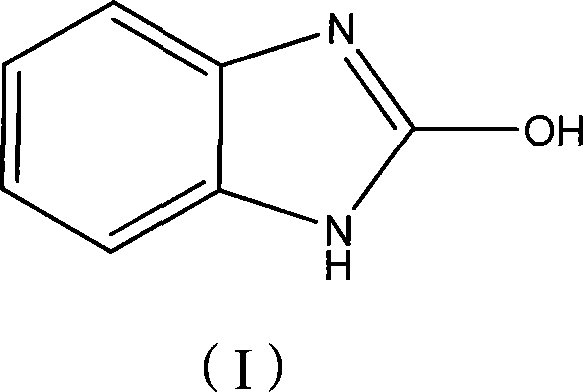
Advanced Synthesis of 2-Hydroxybenzimidazole: Technical Breakthroughs for Commercial Scale-Up
Advanced Synthesis of 2-Hydroxybenzimidazole: Technical Breakthroughs for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for producing critical heterocyclic intermediates. A pivotal advancement in this domain is documented in patent CN101209996A, which outlines a superior chemical synthesis method for 2-hydroxybenzimidazole. This compound serves as a vital building block for various active pharmaceutical ingredients (APIs), including antihistamines like mizolastine, and acts as a key precursor for organic pigments. The traditional reliance on harsh thermal conditions and high-boiling solvents has long plagued manufacturers with efficiency losses and safety concerns. However, the innovative approach detailed in this patent utilizes bis(trichloromethyl)carbonate (BTC) as a carbonyl source in the presence of organic bases, enabling the reaction to proceed under remarkably mild conditions ranging from -10°C to 80°C. This shift not only enhances the safety profile of the operation but also significantly improves the purity and yield of the final product, making it an attractive option for reliable pharmaceutical intermediate suppliers aiming to optimize their production lines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the development of the BTC-mediated cyclization, the industrial standard for synthesizing 2-hydroxybenzimidazole involved the condensation of o-phenylenediamine with urea. This legacy process necessitates the use of high-boiling solvents such as n-amyl alcohol, dimethylformamide (DMF), or ethylene glycol to achieve the requisite activation energy for cyclization. Consequently, the reaction must be maintained at elevated temperatures for extended periods, often involving prolonged reflux. Such operational parameters introduce significant安全隐患 (safety hazards), including the risk of thermal runaway and the degradation of heat-sensitive intermediates. Furthermore, the removal of high-boiling solvents post-reaction is energy-intensive and technically challenging, often requiring high-vacuum distillation which increases production costs. The yields obtained through these conventional thermal methods are frequently described as merely moderate, and the harsh conditions can lead to the formation of complex impurity profiles that are difficult to purge, thereby compromising the quality required for high-purity pharmaceutical intermediate applications.
The Novel Approach
In stark contrast to the thermal intensity of urea-based methods, the novel approach utilizing bis(trichloromethyl)carbonate represents a paradigm shift towards milder, more controlled chemistry. By employing BTC as an efficient phosgene equivalent, the reaction can be initiated and sustained at temperatures as low as -10°C and up to 80°C, with optimal results often observed between -5°C and 30°C. This drastic reduction in thermal demand eliminates the need for energy-heavy reflux setups and allows for the use of volatile, easily recoverable solvents like dichloromethane or chloroform. The reaction mechanism proceeds smoothly through a carbamoylation intermediate which rapidly cyclizes in the presence of organic bases such as pyridine or triethylamine. This pathway not only accelerates the reaction kinetics, reducing total processing time to between 1 and 13 hours, but also ensures a cleaner reaction profile. The ability to operate under such温和 (mild) conditions translates directly into enhanced process safety and operational simplicity, providing a distinct competitive advantage for manufacturers focused on cost reduction in API manufacturing.
Mechanistic Insights into BTC-Mediated Cyclization
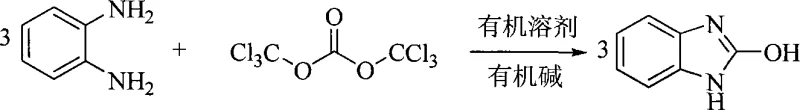
The core of this synthetic breakthrough lies in the reactivity of bis(trichloromethyl)carbonate (BTC), which serves as a solid, stable substitute for gaseous phosgene. In the presence of an organic base, BTC decomposes in situ to generate reactive carbonyl species that attack the nucleophilic amine groups of o-phenylenediamine. This initial step forms a urea-like intermediate, which subsequently undergoes an intramolecular cyclization to close the imidazole ring. The choice of organic base is critical; bases like pyridine or triethylamine not only neutralize the hydrochloric acid byproduct generated during the decomposition of BTC but also catalyze the cyclization step by deprotonating the intermediate. The stoichiometry is carefully balanced, typically employing a molar ratio of o-phenylenediamine to BTC ranging from 1:0.34 to 1:0.8, ensuring that the carbonyl source is not in excessive surplus which could lead to side reactions. This precise control over the reaction environment prevents the formation of polymeric byproducts or over-chlorinated species, which are common pitfalls in less controlled halogenation processes. The result is a highly selective transformation that preserves the integrity of the aromatic system while efficiently constructing the heterocyclic core.
Impurity control is another significant advantage of this mechanistic pathway. In traditional high-temperature syntheses, thermal stress often leads to the decomposition of the starting material or the product, generating tarry residues and colored impurities that are difficult to remove. The low-temperature nature of the BTC method mitigates these thermal degradation pathways almost entirely. Furthermore, the use of chlorinated solvents like dichloromethane facilitates excellent solubility of the reactants while allowing for easy crystallization or precipitation of the product upon cooling or solvent exchange. The work-up procedure involves simple filtration and washing with water and organic solvents, which effectively removes residual salts and unreacted amines. This streamlined purification process ensures that the final 2-hydroxybenzimidazole achieves purity levels exceeding 98%, meeting the stringent specifications required for downstream pharmaceutical synthesis. For R&D teams, understanding this mechanism provides a roadmap for further optimization, such as fine-tuning the base-to-substrate ratio to maximize yield without compromising purity.
How to Synthesize 2-Hydroxybenzimidazole Efficiently
Implementing this synthesis route requires careful attention to reagent addition rates and temperature control to manage the exothermic nature of the BTC decomposition. The general protocol involves dissolving o-phenylenediamine and the chosen organic base in a dry organic solvent, followed by the slow, dropwise addition of the BTC solution to maintain the reaction temperature within the optimal window of -5°C to 30°C. Once the addition is complete, the mixture is stirred for a defined period, typically 3 to 6 hours, to ensure complete conversion. The reaction progress can be monitored via TLC or HPLC to determine the exact endpoint. Upon completion, the reaction mass is cooled to induce precipitation of the product, which is then isolated by filtration. The crude solid is washed thoroughly to remove any adhering mother liquor and salts, then dried under vacuum to yield the final white to off-white powder. Detailed standardized synthesis steps and specific parameter optimizations for different scales are outlined in the guide below.
- Dissolve o-phenylenediamine and an organic base (such as pyridine or triethylamine) in a suitable organic solvent like dichloromethane or chloroform.
- Control the reaction temperature between -10°C and 80°C, preferably -5°C to 30°C, while slowly adding bis(trichloromethyl)carbonate dropwise.
- Stir the mixture for 1 to 13 hours, then cool, filter, wash the filter cake with solvent and water, and dry to obtain the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this BTC-mediated synthesis offers substantial strategic benefits beyond mere technical feasibility. The primary driver for adoption is the potential for significant cost reduction in pharmaceutical intermediate manufacturing. By eliminating the need for high-boiling, difficult-to-remove solvents like DMF or ethylene glycol, manufacturers can drastically reduce energy costs associated with solvent recovery and distillation. Furthermore, the reaction times are considerably shorter compared to traditional reflux methods, increasing the throughput of existing reactor vessels and improving overall asset utilization. The raw materials, specifically o-phenylenediamine and bis(trichloromethyl)carbonate, are commodity chemicals with stable global supply chains, ensuring consistent availability and pricing power. This stability is crucial for long-term contract planning and mitigates the risk of supply disruptions that can plague more exotic reagent-dependent processes.
- Cost Reduction in Manufacturing: The economic advantages of this process are multifaceted, stemming primarily from the reduction in energy consumption and waste treatment costs. Since the reaction operates at near-ambient or mildly elevated temperatures, the demand for steam or heating oil is minimized, directly lowering utility expenses. Additionally, the use of volatile solvents like dichloromethane allows for efficient recovery and recycling through standard distillation columns, reducing the volume of fresh solvent required per batch. The high selectivity of the reaction means fewer byproducts are formed, which simplifies the wastewater treatment process and lowers the burden on environmental compliance systems. These cumulative efficiencies translate into a lower cost of goods sold (COGS), allowing suppliers to offer more competitive pricing to their downstream clients without sacrificing margin.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available, non-proprietary raw materials. Unlike specialized catalysts or reagents that may have single-source suppliers, o-phenylenediamine and BTC are produced by multiple chemical manufacturers globally. This diversification of the supply base reduces the risk of bottlenecks and ensures that production schedules can be maintained even if one supplier faces issues. Moreover, the simplicity of the process equipment—requiring only standard glass-lined or stainless steel reactors with basic cooling capabilities—means that the technology can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs). This flexibility is invaluable for scaling production to meet surging market demand or for establishing redundant production capacities in different geographic regions to mitigate logistical risks.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often introduces unforeseen challenges, particularly regarding heat transfer and mixing. However, the mild exothermic profile of this BTC cyclization makes it inherently safer and easier to scale. The ability to control the reaction temperature effectively prevents hot spots that could lead to runaway reactions, a critical safety consideration for large-scale operations. From an environmental perspective, the process generates significantly less hazardous waste compared to older methods. The reduction in heavy metal catalysts (which are not required here) and high-boiling organic solvents simplifies the disposal of waste streams and aids in meeting increasingly strict environmental regulations. This alignment with green chemistry principles not only reduces regulatory risk but also enhances the corporate sustainability profile of the manufacturer, a factor that is becoming increasingly important to global pharmaceutical buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. They are derived from the specific technical disclosures and beneficial effects highlighted in the patent literature, providing clarity for stakeholders evaluating this technology for adoption. Understanding these nuances is essential for making informed decisions about process integration and supplier selection.
Q: What are the primary advantages of using bis(trichloromethyl)carbonate over urea for this synthesis?
A: Using bis(trichloromethyl)carbonate allows for significantly milder reaction conditions (-10°C to 80°C) compared to the high-temperature reflux required by traditional urea methods. This reduces energy consumption, minimizes safety risks associated with prolonged heating, and typically results in higher yields and purity profiles suitable for pharmaceutical applications.
Q: Which organic solvents are most effective for this cyclization process?
A: The patent identifies chlorinated solvents such as dichloromethane, chloroform, and 1,2-dichloroethane as highly effective. Additionally, ethers like 2-methyltetrahydrofuran and aromatics like toluene can be used. Dichloromethane is often preferred for its ability to dissolve reactants well while maintaining a low boiling point for easy removal post-reaction.
Q: How does this method impact the environmental footprint of production?
A: This method generates less waste ('three wastes') compared to older techniques that utilize high-boiling solvents like DMF or ethylene glycol. The milder conditions reduce energy usage, and the use of volatile organic solvents allows for easier recovery and recycling, aligning with modern green chemistry principles for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydroxybenzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We understand the critical importance of stringent purity specifications in the pharmaceutical sector, which is why our rigorous QC labs employ advanced analytical techniques to verify every batch of 2-hydroxybenzimidazole against the highest industry standards. By leveraging the advanced BTC-mediated synthesis method, we are able to deliver products that not only meet but often exceed the purity requirements necessary for sensitive downstream API synthesis, providing our partners with a reliable foundation for their own manufacturing success.
We invite global pharmaceutical and agrochemical companies to collaborate with us to optimize their supply chains. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. Whether you require pilot-scale quantities for clinical trials or multi-ton volumes for commercial launch, NINGBO INNO PHARMCHEM is equipped to support your growth with consistent quality, competitive pricing, and unwavering supply chain reliability.
