Advanced Synthesis of Minodronic Acid Intermediates: Enhancing Purity and Commercial Scalability
Introduction to Advanced Minodronic Acid Synthesis
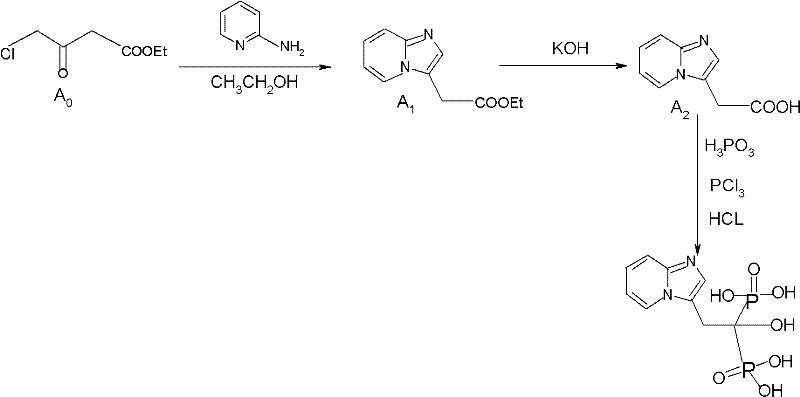
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical therapeutic agents, particularly those addressing chronic conditions like osteoporosis. Patent CN102153585A presents a transformative approach to the synthesis of Minodronic Acid, a potent bisphosphonate, and its key intermediates. This intellectual property addresses long-standing inefficiencies in the legacy production methods, which were plagued by hazardous reagents and poor atom economy. By re-engineering the synthetic route, the inventors have established a protocol that not only enhances the safety profile of the manufacturing environment but also drastically improves the overall yield and purity of the final active pharmaceutical ingredient (API). For stakeholders in the global supply chain, this patent represents a pivotal shift towards greener, more cost-effective chemical manufacturing.
The core innovation lies in the strategic replacement of toxic and expensive starting materials with benign, commercially abundant alternatives. Specifically, the transition from sodium cyanide-based chemistry to a potassium hydroxide-mediated pathway eliminates a major regulatory and safety bottleneck. Furthermore, the optimization of reaction conditions allows for operations at moderate temperatures, removing the energy-intensive requirement for cryogenic cooling. This technical breakthrough ensures a more reliable supply of high-purity Minodronic Acid intermediates, directly benefiting procurement strategies and production planning for downstream drug manufacturers.
![Chemical structure of Minodronic Acid showing the imidazo[1,2-a]pyridine core and bisphosphonic acid groups](/insights/img/minodronic-acid-intermediate-synthesis-pharma-supplier-20260306102209-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Minodronic Acid relied on synthetic routes that were inherently dangerous and economically inefficient. Traditional methodologies frequently necessitated the use of sodium cyanide, a hypertoxic chemical that imposes severe handling restrictions, complex waste disposal protocols, and significant liability risks for manufacturing facilities. Additionally, these legacy processes often required extremely low reaction temperatures, sometimes reaching as low as -80°C, which demands specialized cryogenic equipment and results in excessive energy consumption. The reliance on expensive halogenated precursors, such as 4-ethyl bromoacetoacetate, further inflated the raw material costs, while the multi-step nature of the old synthesis (often involving 5 to 6 steps) led to cumulative yield losses, with reported molar yields hovering around a mere 38.9%.
The Novel Approach
In stark contrast, the methodology disclosed in CN102153585A streamlines the synthesis into a concise three-step sequence that prioritizes safety and efficiency. The new route ingeniously substitutes the toxic cyanide reagent with potassium hydroxide, a common and manageable base, thereby simplifying the operational workflow and reducing environmental impact. By switching the alkylating agent to 4-chloroacetyl acetacetic ester, the process leverages a cheaper and more readily available raw material without compromising reaction kinetics. The elimination of deep-freeze conditions in favor of moderate heating (40-110°C) significantly lowers the barrier to entry for scale-up, allowing standard reactor setups to be utilized effectively. This holistic redesign has propelled the molar yield to an impressive 83.2%, more than doubling the output compared to conventional techniques.
Mechanistic Insights into Imidazo[1,2-a]pyridine Formation and Phosphonation
The chemical elegance of this synthesis begins with the cyclization of 2-aminopyridine and 4-chloroacetyl acetacetic ester to form the imidazo[1,2-a]pyridine core (Intermediate A1). This condensation reaction is facilitated by the nucleophilic attack of the pyridine nitrogen on the alpha-carbon of the chloroacetyl group, followed by intramolecular cyclization. The choice of solvent plays a critical role here; the patent highlights the efficacy of ethanol or ethyl acetate, which not only dissolve the reactants effectively but also facilitate the removal of byproducts during the workup phase. The subsequent hydrolysis step converts the ester moiety into a carboxylic acid (Intermediate A2) using aqueous KOH. This saponification is conducted under mild thermal conditions, ensuring the stability of the sensitive heterocyclic ring while achieving complete conversion, a feat difficult to accomplish with the harsher conditions required by older cyanide-based routes.
The final transformation involves the introduction of the bisphosphonic acid functionality, a crucial structural motif for the drug's biological activity in inhibiting bone resorption. This is achieved through a reaction with phosphorous acid and phosphorus trichloride in a chlorobenzene medium. The mechanism likely involves the formation of a phosphonate intermediate which is subsequently hydrolyzed under acidic conditions (6N HCl) to yield the free phosphonic acid groups. Rigorous control of pH during the isolation of Intermediate A2 (adjusting to pH 2-3) is essential to precipitate the desired product while keeping impurities in the solution. The final crystallization steps, utilizing ethanol and ethyl acetate washes, are designed to strip away residual inorganic salts and organic impurities, ensuring the final API meets stringent purity specifications required for clinical applications.
How to Synthesize Minodronic Acid Efficiently
Implementing this optimized synthesis requires precise adherence to the stoichiometric ratios and thermal profiles outlined in the patent data. The process begins with the careful addition of the chloroacetyl ester to the aminopyridine solution under nitrogen protection to prevent oxidation. Following the isolation of the first intermediate, the hydrolysis step must be monitored via TLC to ensure complete conversion before acidification. The final phosphonation stage demands strict temperature control during the addition of phosphorus trichloride to manage exothermicity, followed by a prolonged reflux to ensure full incorporation of the phosphorus groups. For a detailed, step-by-step breakdown of the exact quantities, timing, and purification techniques validated in the examples, please refer to the standardized guide below.
- Cyclization of 2-aminopyridine with 4-chloroacetyl acetacetic ester in ethanol to form the imidazo[1,2-a]pyridine intermediate (A1).
- Hydrolysis of the ester intermediate using potassium hydroxide (KOH) to obtain the carboxylic acid derivative (A2), replacing toxic sodium cyanide methods.
- Phosphonation reaction using phosphorous acid and phosphorus trichloride in chlorobenzene, followed by acid hydrolysis and crystallization to yield pure Minodronic Acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers compelling strategic advantages that extend beyond simple unit cost calculations. The primary value proposition lies in the drastic simplification of the supply chain risk profile. By eliminating sodium cyanide, manufacturers remove the need for specialized hazardous material handling certifications and the associated high costs of toxic waste remediation. This shift not only reduces the direct operational expenditure but also mitigates the risk of production stoppages due to regulatory compliance issues or supply disruptions of controlled substances. The ability to source raw materials like 4-chloroacetyl acetacetic ester from the broader commodity chemical market, rather than relying on niche, expensive bromo-derivatives, further stabilizes the supply chain against price volatility.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by both raw material arbitrage and yield enhancement. Substituting the expensive bromo-ester with a chloro-ester significantly lowers the bill of materials, while the jump in yield from roughly 39% to over 83% effectively halves the raw material cost per kilogram of finished product. Furthermore, the reduction in synthetic steps from five or six down to three decreases labor hours, utility consumption, and solvent usage, leading to substantial overall cost savings in the manufacturing of this pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: The robustness of the new method ensures a more consistent and reliable supply of Minodronic Acid intermediates. The use of common solvents like ethanol and ethyl acetate, as opposed to hazardous dioxane, simplifies logistics and storage requirements. Additionally, the avoidance of extreme low-temperature reactions means that production is less susceptible to equipment failures or energy supply fluctuations, guaranteeing shorter lead times and improved on-time delivery performance for downstream API manufacturers.
- Scalability and Environmental Compliance: From a sustainability perspective, this route aligns perfectly with modern green chemistry principles. The absence of heavy metal catalysts and toxic cyanide simplifies the effluent treatment process, reducing the environmental footprint of the facility. The mild reaction conditions and high throughput make the process highly scalable, allowing for seamless transition from pilot plant batches to multi-ton commercial production without the engineering complexities associated with cryogenic reactors or high-pressure toxic gas handling.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms historical precedents. Understanding these nuances is critical for technical teams evaluating the feasibility of adopting this route for their own production lines or for procurement teams negotiating supply contracts based on these improved specifications.
Q: How does the new synthesis method improve safety compared to traditional routes?
A: The novel method completely eliminates the use of highly toxic sodium cyanide (NaCN) and avoids extreme low-temperature reactions (-80°C), significantly reducing operational hazards and waste treatment costs.
Q: What represents the primary cost-saving driver in this manufacturing process?
A: Cost efficiency is achieved by substituting expensive 4-ethyl bromoacetoacetate with the more economical 4-chloroacetyl acetacetic ester, alongside a dramatic increase in molar yield from approximately 39% to over 83%.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes common solvents like ethanol and ethyl acetate instead of hazardous dioxane, operates at moderate temperatures (40-110°C), and involves fewer steps, making it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Minodronic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthesis routes in the competitive landscape of pharmaceutical intermediates. Our technical team has extensively analyzed the innovations presented in CN102153585A and possesses the expertise to implement this advanced chemistry at an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this high-yield, low-toxicity process are fully realized in tangible product output. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Minodronic Acid intermediate we deliver adheres to the highest international standards.
We invite global partners to leverage our technical capabilities to optimize their supply chains and reduce manufacturing costs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your osteoporosis treatment portfolio.
